LNCaP spheroid morphology and doubling time
Small cell clusters shaped sooner or later after 25,000 LNCaP cells had been seeded in ultra-low attachment 96-well plates. The cell clusters step by step aggregated over the course of the following two days to create a three-dimensional spheroid construction. The spheroids consolidated to a imply dimension of 1000 μm after ten days (Fig. 1a).
LNCaP spheroid morphology and doubling time. (a) Microscopic morphology of a spheroid inside ten days after seeding in 96 nicely ULA plates (scale bar = 500 μm). (b) Doubling time (hours) in LNCaP monolayer and spheroids with growing preliminary cell quantity (5,000; 10,000; 25,000). (Field plots min to max; n = 7; **** ≙ p < 0.0001; ** ≙ p < 0.01). (c) Microscopic integrity of a spheroid pre- and post-injection with a 20 G needle (scale bar = 500 μm). (d) Hematoxylin and Eosin stain of a 6-days maturated LNCaP spheroid from initially 25,000 seeded cells (white scale bar = 200 μm; black scale bar = 50 μm).
The doubling time of LNCaP monolayers was 60 h. Nonetheless, the typical technology time in spheroids considerably elevated with a better preliminary variety of cells (Fig. 1b). Doubling time of spheroids with an preliminary cell variety of 5,000 LNCaP was 77 h in distinction to 97 h of spheroids with an preliminary cell variety of 10,000 (p < 0.01). The biggest spheroids, which initially developed from 25,000 cells, exhibited the longest technology time of 150 h, which considerably differed from spheroids developed from 10,000 cells (p < 0.0001).
In preparation for the deliberate orthotopic in-vivo implantation, we assessed the integrity of preformed LNCaP spheroids earlier than and after aspiration in and injection by means of a 20 G needle. Six-day matured spheroids initially cultivated from 25,000 cells maintained their morphology microscopically and histologically when aspirated and injected with a 20 G needle, not like spheroids with greater cell numbers (Fig. 1c,d).
Cell rely and viability in LNCaP spheroids
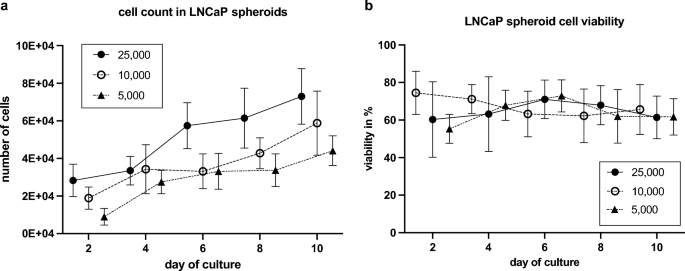
LNCaP spheroids confirmed regular development over a interval of ten days. A number of spheroids skilled a lower in whole cell rely two days after preliminary cultivation. All spheroids reached their most whole cell rely after ten days. The ultimate variety of LNCaP cells elevated relying on the preliminary cell rely. Spheroids initiated with 25,000 cells had a median cell rely of 77,000 after ten days, whereas these with 10,000 cells elevated to 59,000 after ten days. The smallest spheroids beginning with 5,000 LNCaP cells reached a complete of 44,000 cells after 10 days (Fig. 2a).
Cell rely and viability of LNCaP spheroids. (a) Cell rely in LNCaP spheroids with totally different preliminary cell numbers (5,000; 10,000; 25,000) inside ten days (imply with SD; n = 7). (b) Cell viability in LNCaP spheroids with totally different preliminary cell numbers (5,000; 10,000; 25,000) inside ten days (imply with SD; n = 7).
Cell viability in all three spheroid teams ranged from 50 to 85% (Fig. 2b). Spheroids with an preliminary rely of 25,000 and 5,000 cells achieved the best viability (70%) inside ten days. Spheroids with an preliminary rely of 10,000 cells confirmed a slight lower in viability, dropping from a median of 75–65%.
Orthotopic tumor cell engraftment and improvement of metastases
In few circumstances, using a big injection cannula resulted in perforation of the prostatic capsule impeding protected implantation of the LNCaP spheroids within the left anterior prostate lobe. In these circumstances, injection was efficiently carried out within the contralateral (proper) anterior prostate lobe.
4 of eight mice conventionally transplanted with LNCaP single cells from monolayer tradition developed tumors. Lung and lymph node metastases emerged from two of those tumors. 4 out of eight mice implanted with spheroids confirmed tumors, three of which resulted in lung and lymph node metastases (Desk 1).
Sonographic tumor quantity and PSA measurements
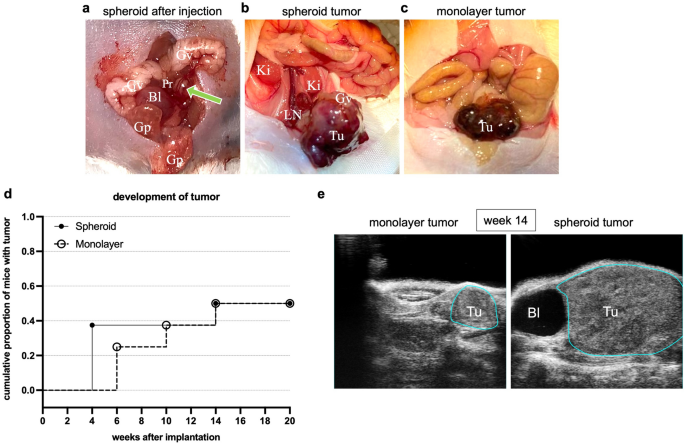
Profitable intraprostatic implantation of a LNCaP spheroid was macroscopically confirmed (Fig. 3a). Utilizing high-resolution small animal ultrasonography, the gut, testicles, epididymis, vasa deferentia, and urinary bladder had been recognized and differentiated from the prostate. Ultrasound visualized native tumor development in all tumor-bearing mice, as exemplary depicted in Fig. 3e. At post-mortem, the sonographic tumor development was macroscopically verified (Fig. 3b,c).
Orthotopic tumor development. (a) Intraoperative situs after profitable intraprostatic implantation of a LNCaP spheroid (marked with inexperienced arrow). (b,c) Intraoperative situs after 20 weeks with formation of a tumor (Tu) after LNCaP spheroid and LNCaP monolayer implantation (tenfold magnification). The glandulae vesiculosae (Gv), the glandulae praeputiales (Gp), the urinary bladder (Bl), the kidneys (Ki) and the left anterior prostate lobe (Pr) are seen. After twenty weeks, the implanted spheroid proliferated into a big tumor (Tu) with formation of lymph node metastases (LN). (d) Cumulative proportion of mice with tumor improvement inside 20 weeks after implantation of a spheroid or single cell suspension from monolayer tradition. (e) Excessive-resolution ultrasonography photos of a monolayer and spheroid tumor (Tu) at week 14. The tumors are marked with a blue line. Bl = bladder.
Three mice developed tumors 4 weeks after spheroid injection, whereas no tumors had been detected within the monolayer injection group presently level (Fig. 3d). A complete of three monolayer tumors weren’t detectable till ten weeks after monolayer single cell injection (Fig. 3d). An extra mouse in every group developed a tumor at fourteen weeks (Fig. 3d).
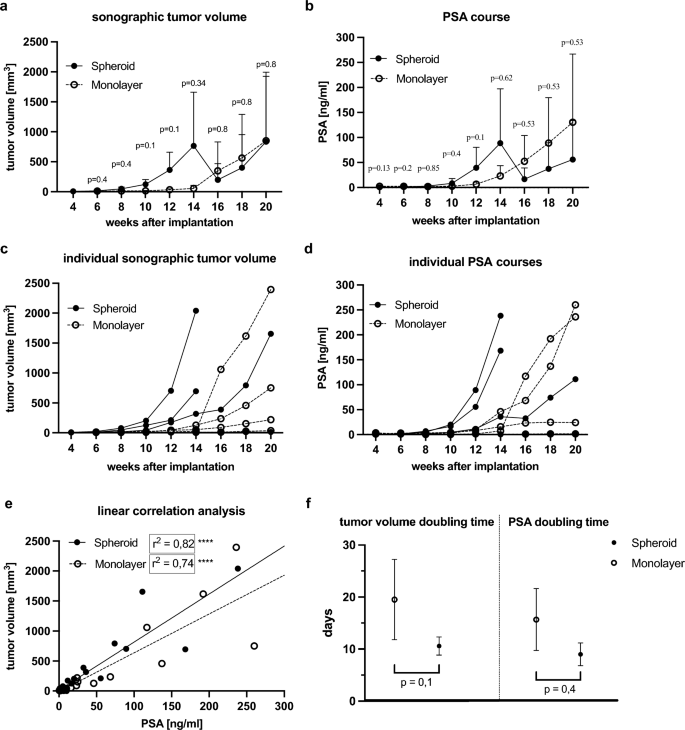
At fourteen weeks, the imply major tumor quantity of LNCaP spheroid tumors (766 mm3) was almost 14 occasions greater than that of LNCaP monolayer tumors (55 mm3, p = 0.34) (Fig. 4a,c). The tumors grew to a imply dimension of 835 mm3 (spheroid) and 850 mm3 (monolayer) after twenty weeks (Fig. 4a,c). The spheroid tumors ranged in dimension from 5.82 mm3 to 2041 mm3, whereas the monolayer tumors ranged from 7.71 mm3 to 2395 mm3. The preliminary tumors within the spheroid group had been 5 mm3 to 10 mm3 in dimension. Two of those tumors grew quickly, reaching roughly 700 mm3 and 2041 mm3 after solely fourteen weeks (Fig. 4c). 4 weeks later, the third early detected tumor had a tumor dimension larger than 700 mm3.
Growth of major tumor quantity and serum PSA. (a) Sonographic tumor quantity after orthotopic implantation of a spheroid (preliminary cell quantity 25,000) and injection of a cell-number matched single cell suspension from monolayer tradition (40,000 cells per injection) within the span of 20 weeks (imply with SD). (b) PSA course of mice inside 20 weeks after implantation (imply with SD). (c) Particular person sonographic tumor volumes. (d) Particular person PSA programs. (e) Linear correlation evaluation of tumor quantity and PSA in spheroid and monolayer tumors (**** ≙ p < 0.0001). (f) Tumor quantity doubling occasions and PSA doubling occasions in days in spheroid and monolayer tumors (imply with SD).
The earliest monolayer tumors had been detected at six weeks with tumor volumes of 8 mm3 and 13 mm3 and at ten weeks with a dimension of 9 mm3. These tumors grew slower than spheroid tumors and reached a dimension of 750 mm3 and 220 mm3 after twenty weeks. The third tumor, with an preliminary quantity of 9 mm3, proliferated quickly from a quantity of roughly 30 mm3 at week fourteen to roughly 1000 mm3 two weeks later. This tumor progressed to grow to be the most important one, measuring 2395 mm3 (Fig. 4c).
After fourteen weeks, one mouse in every group developed a prostate tumor with a quantity of roughly 10 mm3. Nonetheless, these tumors elevated to solely 14 mm3 and 35 mm3 at week 20.
PSA ranges in spheroid tumor bearing mice ranged from 0.11 ng/mL to 238.29 ng/mL, whereas PSA ranges in monolayer tumor bearing mice ranged from 0.27 ng/mL to 260 ng/mL. At week fourteen, imply PSA ranges had been greater after intraprostatic spheroid implantation (88.75 ng/mL) than after single cell injection (23.15 ng/mL, p = 0.62) (Fig. 4b). The best PSA stage in spheroid tumors (238 ng/mL) occurred at fourteen weeks, whereas the best PSA stage in monolayer tumors (260 ng/mL) was measured at twenty weeks (Fig. 4d). The sudden lower in tumor quantity and PSA ranges at week fourteen within the spheroid mice was because of the untimely sacrifice of two mice with the most important tumors to that time (humane finish level), as illustrated within the particular person PSA and tumor quantity improvement (Fig. 4c,d).
The tumor quantity of LNCaP spheroids doubled on common after 10.5 days, whereas monolayer prostate tumors confirmed a doubling of tumor quantity after 19.5 days (p = 0.1) (Fig. 4f). PSA doubling time averaged 9 days in spheroid mice in comparison with 16 days in monolayer mice (p = 0.4) (Fig. 4f).
Linear correlation evaluation revealed a big constructive correlation between sonographic tumor quantity and PSA (r2 = 0.82 and r2 = 0.74, respectively, and p < 0.0001) validating serum PSA as a legitimate non-invasive measure of tumor burden in our orthotopic mannequin (Fig. 4e).
Histological und immunohistochemical analysis
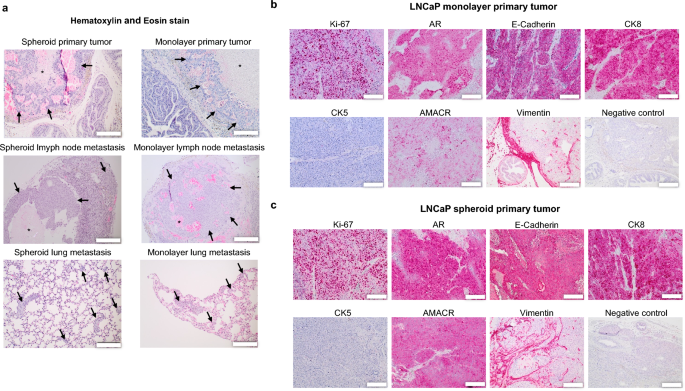
Histological analysis of the first tumors revealed a stable tumor mass with central areas of necrosis and intratumoral hemorrhages (Fig. 5a). The pleomorphic tumor cells exhibited enlarged outstanding nucleoli, a excessive nuclear-to-cytoplasmic ratio and quite a few mitotic figures. Within the tumor periphery, infiltrative tumor development was noticed, and a pseudofibrotic capsule was shaped in some circumstances. In smaller major tumors, the mouse prostate glands had been usually seen, whereas in bigger tumors, they had been fully displaced. Lung metastases offered as a number of tumor cell nests consisting of some cells, a few of which organized into tubular or spherical buildings. The biggest pulmonary metastases had been roughly 200 μm in diameter (Fig. 6a) consisting of huge pleomorphic tumor cells with outstanding nuclei. The lymph node metastases had been characterised by a stable cohesive tumor mass and necrotic zones. The histoarchitecture of the cortex, paracortex, and medulla remained nicely differentiated typically, and the lymph node capsule with marginal sinus was intact. Tumor cells mirrored once more the histological options of the first tumors.
Histology and immunohistochemistry. (a) Histological analysis (H&E staining) of spheroid and monolayer major tumors and metastases. Tumors and metastases are labeled with arrows. Central necroses is labeled with asterisks (*). Scale bar = 200 μm. (b,c) Immunohistochemical expression of Ki-67, AR, E-Cadherin, CK8, CK5, AMACR and Vimentin in LNCaP monolayer and spheroid major tumors. Scale bar = 200 μm.
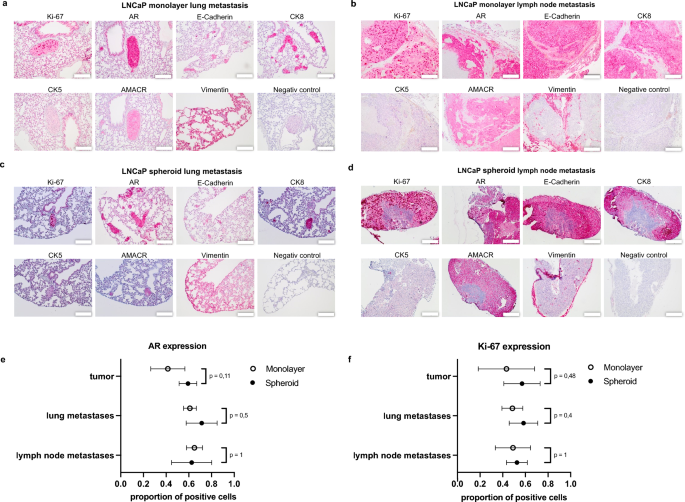
Immunohistochemistry of metastases and quantification of AR and Ki-67 expression in major tumor and metastases. Immunohistochemical expression of Ki-67, AR, E-Cadherin, CK8, CK5, AMACR and Vimentin in LNCaP monolayer lung metastases (a), monolayer lymph node metastases (b), spheroid lung metastases (c) and spheroid lymph node metastases (d). Scale bar = 200 μm. (e) Proportion of LNCaP cells with nuclear AR expression in major tumors, lung and lymph node metastases of monolayer and spheroid tumors (imply with SD). (f) Proportion of Ki-67 expressing LNCaP cells in major tumors, lung and lymph node metastases of monolayer and spheroid tumors (imply with SD).
The first tumors exhibited nuclear staining for Ki-67 and the androgen receptor (Fig. 5b and c). Androgen receptor expression was additionally seen within the cytoplasm. Moreover, the attribute expression of cytokeratin 8 as luminal epithelial marker was recognized, whereas CK5 was unfavorable (Fig. 5b,c). Furthermore, sturdy cytoplasmic expression of AMACR and E-cadherin was noticed (Fig. 5b,c). The lung and lymph node metastases displayed the identical marker expressions as the first tumors (Fig. 6).
The proportion of tumor cells expressing nuclear AR was greater in spheroid major tumors (over 60%) than in monolayer major tumors (45%, p = 0.11) (Fig. 6e). In lung metastases of monolayer tumors, 60% of the tumor cells confirmed sturdy nuclear staining of the AR (Fig. 6e) in distinction to 70% in lung metastases from spheroid tumors (p = 0.5) (Fig. 6e). Lymph node metastases of spheroid tumors exhibited roughly 65% AR-positive cells, which was just like the monolayer lymph node metastases (Fig. 6e).
The expression of the proliferation marker Ki-67 in spheroids was extra pronounced within the peripheral proliferation zone. The expression in spheroid major tumors was on common 20% greater than in monolayer major tumors (p = 0.48) (Fig. 6f). The proportion of constructive nuclear staining was extra pronounced in lung metastases of the spheroids (60%) in comparison with monolayer lung metastases (50%, p = 0.4) (Fig. 6f). In lymph node metastases, round 50% of the tumor cells in each teams expressed Ki-67 (Fig. 6f).