Introduction
Diarrhea represents a typical situation in most cancers sufferers present process chemotherapy that may severely affect the standard of life and therapy outcomes. Chemotherapy-associated diarrhea is a fancy situation requiring a correct understanding of its underlying mechanisms and efficient methods for prevention and administration (1).
Diarrhea in most cancers sufferers is attributable to varied components, primarily triggered by the aggressive nature of most cancers and the uncomfortable side effects of therapeutic interventions equivalent to chemotherapy. The gastrointestinal mucosa, a important barrier defending the digestive system, turns into inclined to break by therapies that disrupt regular mobile processes. Chemotherapy-induced diarrhea, a typical manifestation, is characterised by the poisonous results of anticancer medication on quickly dividing cells throughout the intestinal lining. Moreover, alterations within the intestine microbiota, irritation, and the discharge of varied signaling molecules additional contribute to the disruption of physiological bowel capabilities (1).
Using probiotics in stopping and managing diarrhea relies on each theoretical issues and the outcomes of quite a few scientific trials (2–6). Lactic acid micro organism play a pivotal function in addressing dysbiosis by competing for substrates with pathogenic micro organism, producing bacteriocins, and enhancing transepithelial resistance (7). Their enzymatic exercise influences the activation or deactivation of metabolites answerable for inducing diarrhea (8). Furthermore, the manufacturing of short-chain fatty acids, important for the well-being of intestinal mucosal cells, additional contributes to the anti-diarrheal results of probiotics (9, 10).
Irinotecan, a topoisomerase I inhibitor extensively used within the therapy of varied cancers, together with colorectal most cancers, has been related to a better incidence of diarrhea in comparison with different chemotherapeutic brokers (11). This aspect impact not solely poses discomfort to sufferers however may additionally result in dose reductions or interruptions, compromising the efficacy of the therapy. The incidence of irinotecan-induced diarrhea ranges extensively, encompassing 60-90%, with extreme diarrhea affecting 20-40% of sufferers. This gastrointestinal complication assumes important significance within the panorama of morbidity and mortality related to irinotecan-based chemotherapy. Recognized predisposing components embrace age exceeding 65 years, an Japanese Cooperative Oncology Group efficiency standing (ECOG PS) of ≥1, and a historical past of abdominopelvic radiation (11, 12).
The mechanism of irinotecan-induced diarrhea is mediated by its metabolite SN-38, which is glucuronidated within the liver and subsequently excreted into the gut. Throughout the intestinal lumen, bacterial beta-D-glucuronidase deconjugates SN-38, initiating a cascade of occasions that inflict direct injury to the intestinal mucosa, leading to malabsorption of water and electrolytes, finally culminating within the onset of diarrhea (12). Understanding the intricate mechanisms of irinotecan-induced diarrhea is crucial for devising focused interventions to reinforce the general administration of this chemotherapy-related aspect impact (13–16). Sure probiotic micro organism have demonstrated the potential to decrease the exercise of intestinal beta-D-glucuronidase (14, 15). This means a possible avenue for the applying of those micro organism in stopping diarrhea in sufferers present process irinotecan-based remedy (Determine 1).
Beforehand, we performed two scientific trials targeted on stopping irinotecan-induced diarrhea in metastatic colorectal most cancers sufferers (17, 18). Within the pilot research, which included 46 sufferers who acquired the probiotic system Colon Dophilus™ or placebo, we noticed a decreased diarrhea incidence within the probiotic arm with no grade 3/4 diarrhea (17). Primarily based on these outcomes, we carried out a part III trial in the identical affected person inhabitants. On this trial, sufferers acquired a mix of Bifidobacterium BB-12 and Lactobacillus rhamnosus GG, LGG (18). The outcomes of this trial didn’t verify the effectivity of probiotics within the prevention of irinotecan-induced diarrhea; nonetheless, subgroup evaluation steered their effectivity in sufferers with colostomy. These trials utilized totally different probiotic formulation extensively obtainable for sufferers with out prescription. The selection of formulation was decided primarily by their availability for investigator-initiated trials from pharmaceutical corporations. Whereas Colon Dophilus™ is extra complicated and accommodates 10 totally different probiotic strains, the probiotic system Probio-Tec® BG-Vcap-6.5 consists of two strains and has been extra extensively studied in varied scientific eventualities.
The statistical energy of subgroup evaluation, particularly in underrepresented subgroups, is proscribed in single trials. Profiting from similar eligibility standards and a really related statistical design of those two scientific trials, we carried out pooled evaluation aiming to find out the effectiveness of the probiotics within the prophylaxis of irinotecan-induced diarrhea in metastatic colorectal most cancers (CRC) sufferers and figuring out particular subgroups that would profit from preventive administration of probiotics throughout irinotecan-based chemotherapy. Apart from having larger statistical energy for the first endpoint, the dataset of this pooled evaluation has elevated the variety of sufferers in a number of particular subgroups in comparison with particular person earlier trials, which permits extra sturdy testing, enhances the power to detect heterogeneity, and improves the generalizability of research outcomes.
Sufferers and strategies
This mixed evaluation included two research; 46 sufferers with CRC enrolled within the Probio-SK-003 (NCT01410955) between January 2011 and December 2013, beginning a brand new line of irinotecan-based remedy (17) and 233 sufferers of Probio-SK-005 research (NCT02819960) randomized from March 2016 to Might 2022 with similar eligibility standards as earlier trial (18).
Eligibility standards
Each trials had the identical eligibility standards (17, 18). Eligible individuals have been grownup sufferers with histologically confirmed colorectal most cancers beginning a brand new line of chemotherapy primarily based on irinotecan with ECOG PS 0-1 at research entry. Exclusion standards comprised impossibility to take oral remedy, energetic an infection handled by antibiotic remedy, ileostomy or jejunostomy, hypersensitivity to review drug, and any concurrent malignancy aside from non-melanoma pores and skin most cancers, no different most cancers previously 5 years.
Trial design
Each trials have been multi-centered, double-blinded scientific research performed to judge the effectiveness of oral probiotic dietary supplements in comparison with a placebo in stopping extreme diarrhea in sufferers with colorectal most cancers who have been beginning a brand new spherical of chemotherapy therapy involving irinotecan. Sufferers have been randomly assigned to obtain both the probiotic complement or the placebo, with an equal variety of sufferers in every group. The randomization course of was centralized, the place every affected person was given a singular identification quantity and acquired a corresponding container with the assigned therapy. These containers, indistinguishable from one another, have been labeled with sequential numbers assigned randomly to protect blinding. All researchers, statisticians, and sufferers remained unaware of which therapy every affected person acquired till the ultimate end result evaluation.
Therapy
In Probio-SK-003, the probiotic system Colon Dophilus™ (produced by Harmoniom Worldwide, Inc., Mirabel, Canada) was administered orally at a dose of three×1cps per day for 12 weeks and every capsule contained 10×109 CFU of micro organism. Whereas, in Probio-SK-005, the probiotic system Probio-Tec® BG-Vcap-6.5 (produced by Chr. Hansen A/S, Hoersholm, Denmark) containing 2.7×109 CFU was administered orally at a dose of 3×1 cps per day for six weeks. No premedication or affected person monitoring after probiotic supplementation was required in each trials. The probiotic system may be taken after meals or snacks to cut back abdomen upset. The probiotic system may be taken after meals or snacks to cut back abdomen upset. The capsule ought to be swallowed entire or opened, and the content material combined with a small quantity of meals in case of issues with swallowing. Probiotic system Colon Dophilus™ contained Bifidobacterium breve HA-129 (25%), Bifidobacterium bifidum HA-132 (20%), Bifidobacterium longum HA-135 (14.5%), Lactobacillus rhamnosus HA-111 (8%), Lactobacillus acidophilus HA-122 (8%), Lactobacillus casei HA-108 (8%), Lactobacillus plantarum HA-119 (8%), Streptococcus thermophilus HA-110 (6%), Lactobacillus brevis HA-112 (2%), Bifidobacterium infantis HA-116 (0.5%). Probio-Tec BG-Vcap-6.5® contained Bifidobacterium BB-12 (50%) and Lactobacillus rhamnosus GG, LGG (50%).
Period of remedy
In Probio-SK-003, the probiotic system was administered throughout irinotecan-based chemotherapy for 12 weeks, whereas in Probio-SK-005, probiotic supplementation lasted for six weeks. As a result of totally different durations of research therapies, solely the primary 6 weeks of remedy have been used for the evaluation.
In each trials, sufferers may additionally discontinue protocol remedy within the case of intercurrent sickness, affecting the sufferers’ security in investigator judgment, the power to ship therapy or the first research endpoints, and/or by affected person request.
Concomitant remedy
Sufferers acquired full supportive care in the course of the research, together with transfusion of blood and blood merchandise, antibiotic therapy, anti-emetics, antidiarrheal brokers, analgesics, erythropoietin, or bisphosphonates, when applicable.
Therapy analysis
The scientific evaluation encompassed varied components equivalent to demographic data, birthdate, ethnicity, gender, and medical background. This included an in depth account of cancer-specific historical past, encompassing the date of analysis, main tumor sort together with histology findings, previous surgical and/or radiological therapies (together with dates and particular organ/anatomic areas focused), present most cancers stage, earlier systemic therapies, persistent uncomfortable side effects from prior therapies, any historical past of further malignancies, and important medical occasions throughout the final six months. The evaluation of opposed results, together with diarrhea and enterocolitis, was performed in keeping with the NCI Frequent Terminology Standards for Opposed Occasions Model 4.1 (CTCAE) (18). Sufferers maintained diaries to file every day stool frequency and consistency, in addition to the usage of antidiarrheal medicines all through the research. Nevertheless, analysis of sufferers’ compliance with the prescribed research medicines was not carried out (17, 18).
Statistical evaluation
Information evaluation adopted the pre-specified plan for statistical evaluation. The sufferers’ attributes have been summarized by presenting the median (vary) for steady variables and frequency (share) for categorical variables. The Kolmogorov-Smirnov check was utilized to evaluate the distribution’s normality. If the information adopted a traditional distribution, pattern means have been examined utilizing both the Pupil t-test or evaluation of variance (ANOVA), with changes like Bonferroni’s or Tamhane’s primarily based on variance homogeneity. For non-normally distributed information, the nonparametric Mann-Whitney U or Kruskal-Wallis H check was utilized. Fisher’s precise check or Chi-square check was employed for categorical information. Occasion-free survival, particularly regarding diarrhea, was decided using Kaplan-Meier strategies, and in contrast between research arms utilizing the log-rank check. The information have been computed from the initiation of probiotic administration (day 1) till the occasion or the top of the research, at which level the information have been censored. All offered p-values are two-sided, with associations thought-about important if the p-value was 0.05 or decrease. The statistical analyses have been performed utilizing NCSS 2022 statistical software program (Hintze J, 2022, Kaysville, UT, USA).
Outcomes
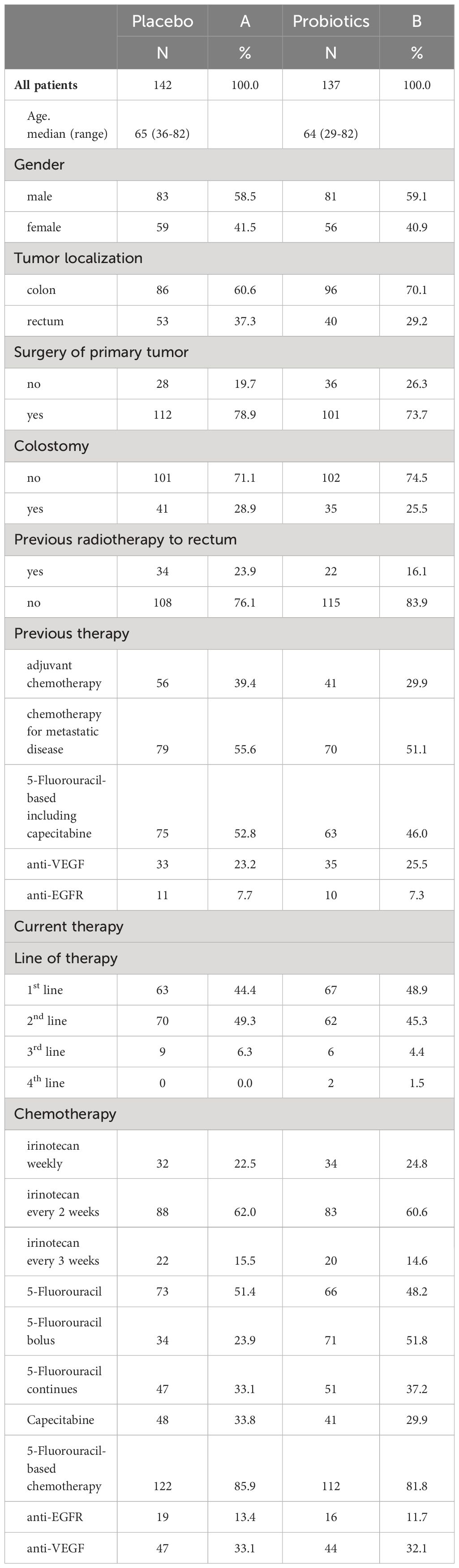
Affected person traits and chemotherapy protocols will be present in Desk 1. There have been disparities noticed between the teams receiving totally different therapies. The probiotic arm had a better proportion of sufferers with colon most cancers in comparison with rectal most cancers, which was in keeping with earlier radiation remedy patterns for rectal most cancers. The placebo arm had barely extra sufferers receiving adjuvant remedy, whereas the probiotic arm had a better variety of sufferers handled with first-line chemotherapy. Colostomy was barely extra prevalent within the placebo arm. The distribution of irinotecan regimens and different therapies, together with 5-FU-based, anti-EGFR, and anti-VEGF remedy, was balanced throughout each arms.
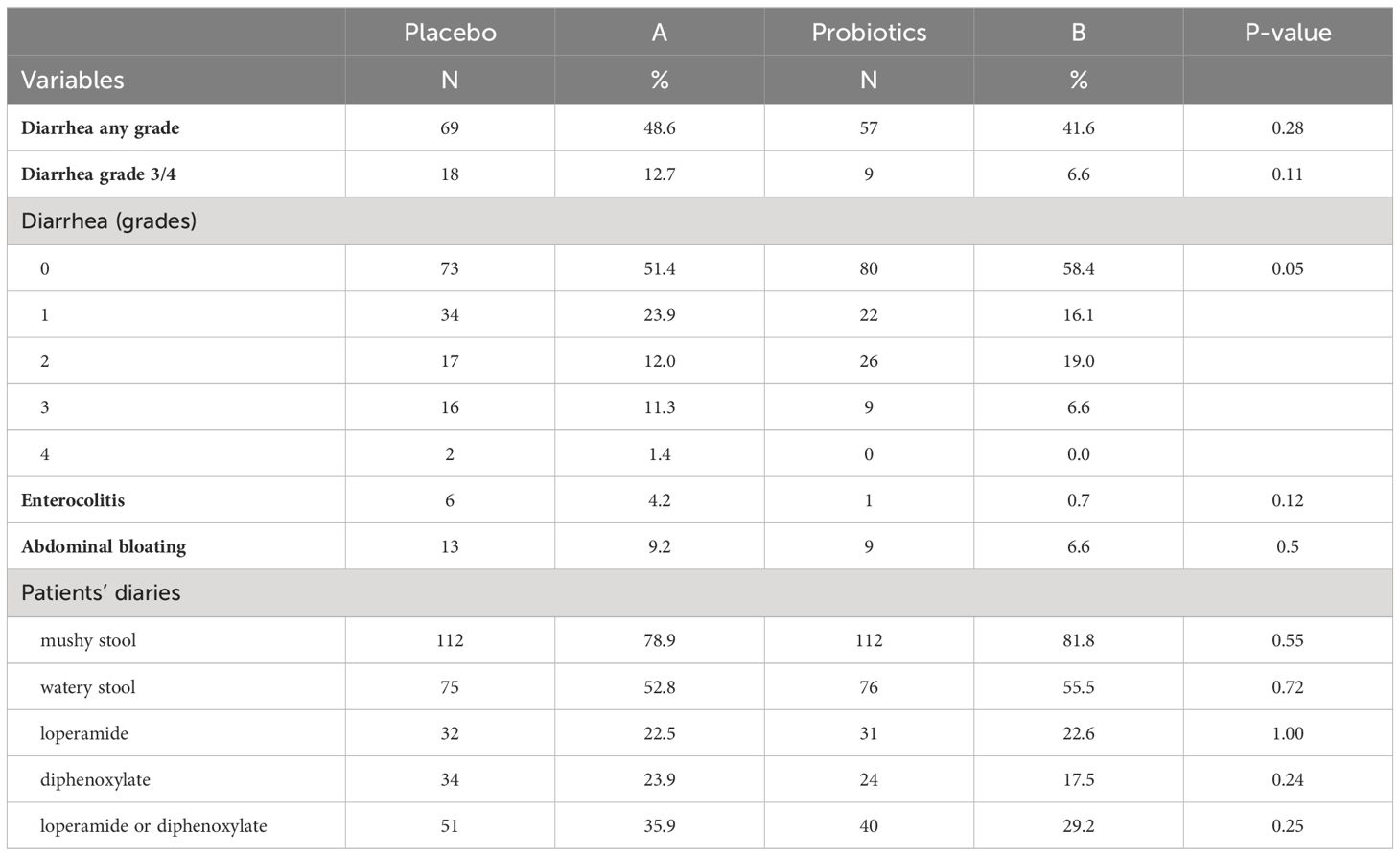
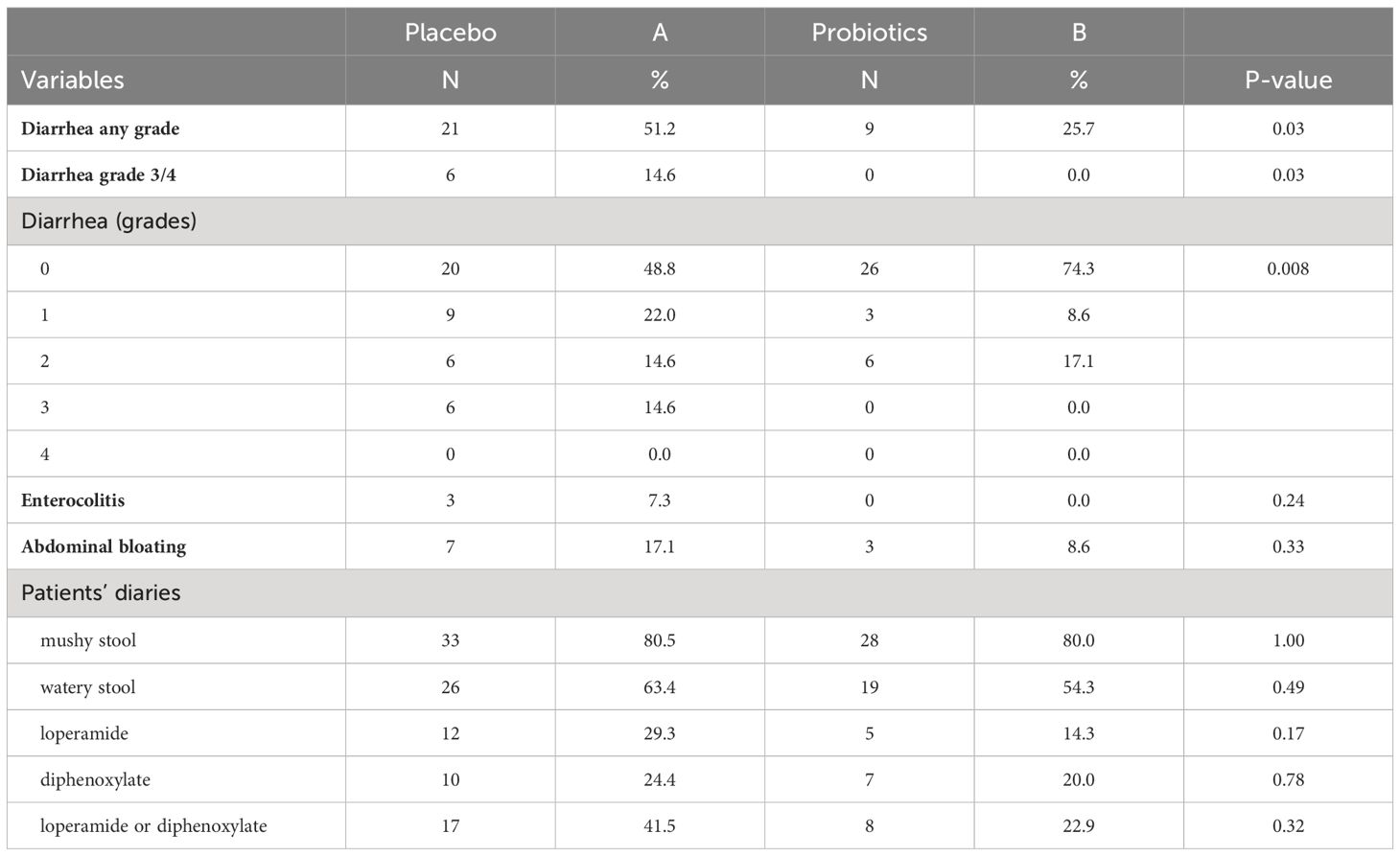
Completely 279 sufferers have been randomized (placebo 142, probiotics 137). Administration of probiotics didn’t considerably scale back the incidence of grade 3/4 diarrhea in comparison with placebo (placebo 12.7% vs. probiotics 6.6%, p = 0.11) (Determine 2A). Neither the general incidence of diarrhea (placebo 48.6% vs. probiotics 41.6%, p = 0.28) nor the incidence of enterocolitis (placebo 4.2% vs. probiotics 0.7%, p = 0.12) was totally different within the placebo vs. probiotic arm (Desk 2). Nevertheless, subgroup evaluation revealed that sufferers with a colostomy who acquired a placebo had a considerably larger incidence of any diarrhea (placebo 51.2% vs. probiotics 25.7%, p = 0.028) and grade 3/4 diarrhea (placebo 14.6% vs. probiotics 0.0%, p = 0.03) in comparison with the probiotic arm. Furthermore, sufferers with colostomy had no enterocolitis in comparison with 7.3% of sufferers in a placebo arm (Desk 3; Determine 2B). Sufferers within the probiotic arm wanted numerically much less salvage remedy (loperamide) in distinction to the placebo arm (placebo 29.3% vs. probiotics 14.3%, p = 0.17). We didn’t observe any an infection attributable to probiotic strains on this research.
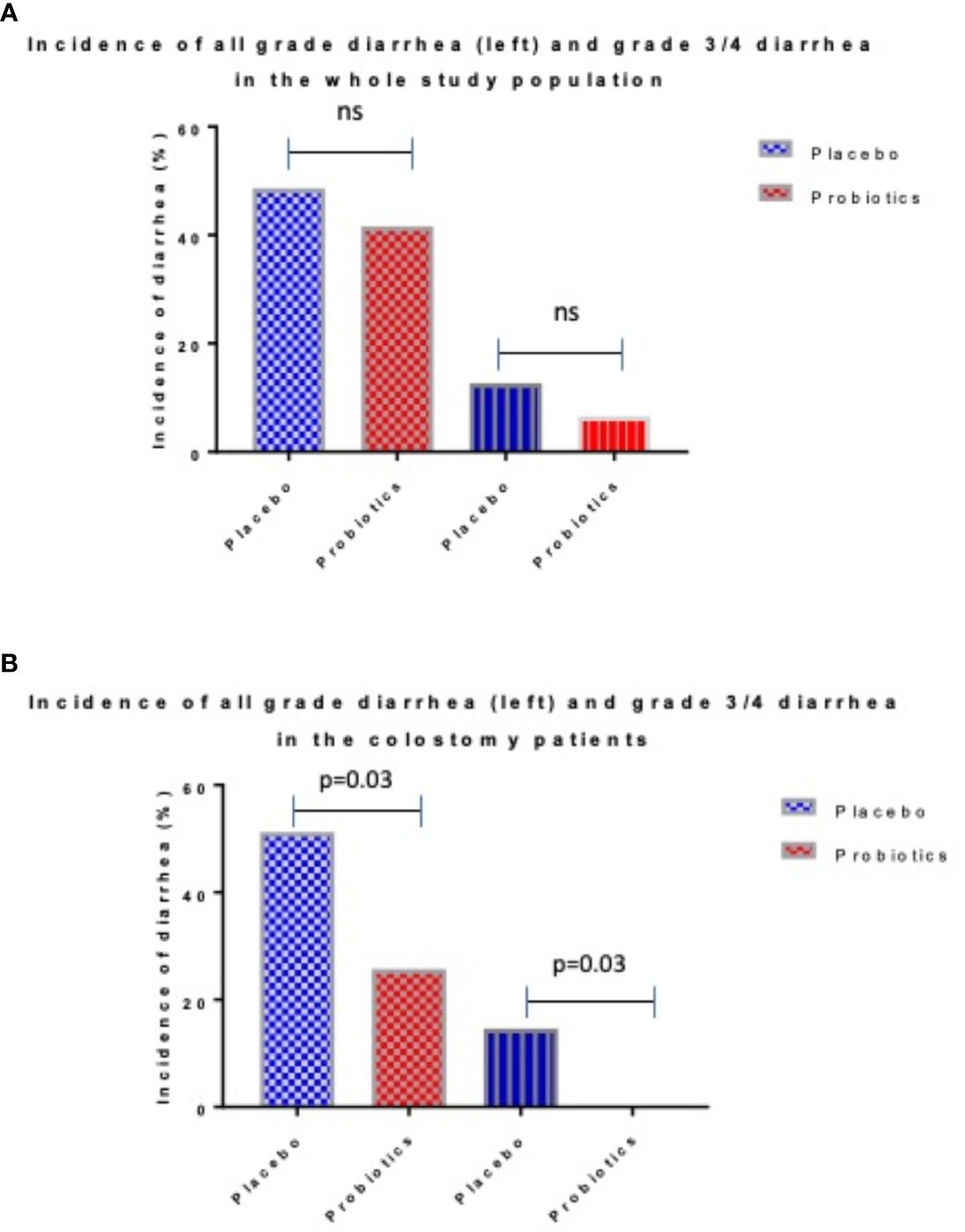
Determine 2 Incidence of diarrhea in entire research populations (A) and sufferers with colostomy (B).
Dialogue
On this pooled evaluation, the administration of probiotics didn’t yield statistically important reductions in grade 3/4 diarrhea, general diarrhea incidence, or enterocolitis in comparison with the placebo group. Nevertheless, a subgroup evaluation recognized a profit for sufferers with colostomy receiving probiotics, displaying considerably decrease incidences of any diarrhea and grade 3/4 diarrhea in comparison with the placebo group. Sufferers with colostomy within the probiotic arm additionally had no circumstances of enterocolitis, in distinction to 7.3% within the placebo arm. Moreover, sufferers within the probiotic arm required numerically much less salvage remedy (loperamide) than these within the placebo arm. Importantly, no infections have been noticed associated to the probiotic strains used within the research.
Animal fashions specializing in irinotecan administration have revealed shifts in microbiota composition, marked by elevated presence of intestinal Enterobacteriaceae spp. and Clostridium cluster XL, accompanied by heightened pro-inflammatory cytokines and alterations in mucosa composition resulting in decreased adhesion websites (19, 20). These modifications contribute to a lower in symbiotic micro organism and a rise in opportunistic pathogens. Whereas quite a few preclinical information recommend the potential advantages of probiotics in mitigating irinotecan-induced gastrointestinal toxicity, scientific proof stays restricted (21–23). A potential observational trial hints on the ameliorative results of Lentilactobacillus kefiri LKF01 (Fefibios®) on extreme irinotecan-induced diarrhea in most cancers sufferers (24). Conversely, a part II/III, randomized, double-blind, placebo-controlled research failed to fulfill its main endpoint of decreasing grade 3/4 irinotecan-induced diarrhea utilizing a high-concentration multi-strain probiotic complement (25). This remark aligns with our trials (17, 18). The disparity underscores the complexity of translating preclinical findings into scientific efficacy and emphasizes the necessity for additional investigation into the optimum probiotic methods for managing irinotecan-induced diarrhea.
Each these trials had the identical eligibility standards, which enabled information pooling. As a result of totally different durations of research therapy, solely the primary 6 weeks of remedy have been used for this evaluation. Variations in end result in every trial might be associated to the totally different probiotic formulation used in addition to totally different incidences of diarrhea in management arms, which might be associated to raised administration of irinotecan toxicity within the final years. Regardless of these variations, each trials constantly confirmed probably the most pronounced impact of probiotics within the prevention of diarrhea in sufferers with a colostomy (17, 18). There was no overlap in any probiotic pressure utilized in these scientific trials. Nevertheless, each formulation contained Lactobacillus and Bifidobacterium, that are extensively utilized in quite a few probiotic merchandise, thus rising the generalizability of research outcomes. Considering the outcomes of the same trial revealed within the summary kind (25), we recommend that the efficacy of probiotics in decreasing irinotecan-induced diarrhea within the unselected affected person inhabitants is unlikely. These outcomes can’t exclude the potential helpful impact of intestine microbiome modification by different probiotic formulation and/or fecal microbiota transplantation within the research affected person inhabitants handled with irinotecan-based chemotherapy. Sadly, any of the utilized probiotic formulation underwent preclinical testing in animal fashions of irinotecan-induced diarrhea, which might additionally have an effect on research outcomes. Future research assessing another intervention to switch the intestine microbiome composition ought to incorporate preclinical testing earlier than continuing to a scientific setting.
In our evaluation, the administration of probiotics was related to a considerably decreased incidence of diarrhea in colostomy sufferers. We are able to’t assess if this might be associated to a lower in bowel beta-glucuronidase exercise as a result of probiotics and/or if that is achieved by one other mechanism. Whereas the incidence of diarrhea in colostomy sufferers on the placebo arm and/or grade ¾ diarrhea was not totally different in comparison with the entire research inhabitants, this was dramatically decreased on the probiotic arm. Whereas shorter bowel size could also be a contributing issue, it is usually doable that variations in microbiome composition might be influencing this remark. To higher perceive this phenomenon, future research ought to examine the pre- and post-treatment composition of the intestine microbiota, in addition to measure beta-glucuronidase exercise. Animal fashions confirmed that the microbiome composition in colostomy is totally different in comparison with regular bowel (26). In a rat mannequin with left colostomy, a major affect on the expansion curve of rats was noticed. Evaluation of the intestinal microbiota indicated that colostomy primarily influenced the cecal microbiota slightly than the colonic microbiota. Notably, there was a rise within the variety of enterococci in each the ileum and cecum and elevated ranges of cecal lactobacilli, contributing to the promotion of lactic acid micro organism in colostomized rats. Apparently, there have been no substantial variations within the translocation of intestinal micro organism to inside organs (spleen, kidneys, lungs, or liver) amongst colostomized, laparotomized, and management rats, no matter their food regimen. The administration of heat-killed Lactobacillus acidophilus pressure LB (inactive probiotic micro organism) exhibited an inclination to stimulate bifidobacteria, doubtlessly influenced by culture-medium fermentation substances within the pharmaceutical product. Nevertheless, this stimulatory impact was abolished by laparotomy and colostomy. Moreover, a pattern in the direction of a probiotic-like impact, unaffected by colostomy, was noticed, as counts of lactobacilli tended to extend in each the cecum and colon of all animals fed with Lactobacillus acidophilus LB (26).
In CRC sufferers with colostomy, variations in microbial composition have been noticed as nicely, displaying a discount in anaerobic micro organism, notably affecting Alistipes, Akkermansia, Intestinimonas, and methane-producing archaea. Gene perform evaluation indicated an underrepresentation of methane and short-chain fatty acid manufacturing in sufferers with a stoma. Furthermore, the presence of a stoma correlated with general decreased taxonomic range however elevated range within the KEGG ((Kyoto Encyclopedia of Genes and Genomes) pathway. Primarily based on the outcomes, sufferers with a stoma exhibit diminished ranges of helpful microbes for most cancers immunotherapy. This research underscores {that a} stoma can considerably alter each taxonomic and purposeful profiles in fecal microbiota, emphasizing its potential as a confounding consider fecal microbiota analyses (27). Accordingly, sufferers with low vs. high-output ileostomy displayed variations in microbiota composition, notably within the share of Bacteroidota between the high-output and low-output teams (14.8% vs 0.5%; p=0.01) (28). One other research investigated the consequences of a probiotic system (Ecologic®825) on the grownup human small intestinal ileostoma microbiota. The findings indicated that supplementation with the probiotic system decreased the expansion of pathobionts, equivalent to Enterococcaceae and Enterobacteriaceae, and decreased ethanol manufacturing. These modifications have been related to important alterations in nutrient utilization and resistance to perturbations. The probiotic-mediated alterations, which coincided with an preliminary improve in lactate manufacturing and a lower in pH, have been adopted by a pointy improve within the ranges of butyrate and propionate (29).
This pooled evaluation, past a number of benefits, has some limitations as nicely. Firstly, scientific trials make the most of totally different probiotic formulation, contributing to the heterogeneity of trials. Furthermore, any of the utilized probiotic formulation underwent preclinical testing in animal fashions of irinotecan-induced diarrhea. Each trials lack compliance measurement in addition to evaluation of intestine colonization by probiotic system and/or the measurement of stool beta-glucuronidase exercise or one other potential biomarker of probiotic efficacy. Regardless of the pooled evaluation of the 2 trials, the statistical energy of a number of subgroups stays low because of the small pattern dimension of the primary trial. Nevertheless, this evaluation permits us to verify the outcomes of probiotic profit in sufferers with colostomy as there was solely a pattern of profit within the Probio-SK-005 research (18).
Conclusions
In conclusion, this mixed evaluation means that probiotics might be helpful in irinotecan-induced diarrhea prevention in colorectal most cancers sufferers with colostomy. We suggest that the preservation of wholesome microbiota composition might be the straightforward, efficient, and unhazardous strategy to cut back gastrointestinal toxicity of irinotecan-based chemotherapy. Future analysis ought to prioritize mechanistic research to analyze the hyperlink between stool beta-glucuronidase exercise and the chance of irinotecan-induced diarrhea. It’s also important to judge varied probiotic formulation and fecal microbiome switch methods to cut back the incidence of chemotherapy-associated diarrhea. Nevertheless, one main problem is that the majority present approaches have been one-size-fits-all, neglecting the distinctive composition of a person’s authentic microbiome, its colonization resistance, dietary influences, concomitant medicines, and host components that may all affect the microbiome. To deal with this complexity, it’s essential to combine broad translational analysis into intervention research, amassing and characterizing organic samples from varied time factors to know the intricate interplay between microbiome modification approaches, biomarkers of change, and scientific endpoints. This may assist optimize therapy methods and enhance affected person outcomes. Till the supply of recent pre- and scientific information on this setting, we recommend that the administration of probiotics formulation containing Lactobacillus and Bifidobacterium strains in colostomy sufferers handled with irinotecan-based chemotherapy appears prudent. Nevertheless, there isn’t any proof to assist the function of probiotic administration in unselected populations aimed toward decreasing irinotecan-induced diarrhea.
Information availability assertion
The uncooked information supporting the conclusions of this text will probably be made obtainable by the authors, with out undue reservation.
Ethics assertion
The research involving people have been permitted by Moral Committee of Nationwide Most cancers Institute, Bratislava, Slovakia. The research have been performed in accordance with the native laws and institutional necessities. The individuals supplied their written knowledgeable consent to take part on this research.
Creator contributions
MM: Conceptualization, Information curation, Formal evaluation, Funding acquisition, Investigation, Methodology, Challenge administration, Sources, Supervision, Validation, Visualization, Writing – authentic draft, Writing – evaluate & modifying. BK: Conceptualization, Methodology, Writing – authentic draft, Writing – evaluate & modifying. JC: Investigation, Writing – authentic draft, Writing – evaluate & modifying. RD: Conceptualization, Funding acquisition, Investigation, Methodology, Writing – authentic draft, Writing – evaluate & modifying. MR: Investigation, Supervision, Writing – authentic draft, Writing – evaluate & modifying. BBy: Investigation, Writing – authentic draft, Writing – evaluate & modifying. PK: Investigation, Writing – authentic draft, Writing – evaluate & modifying. SJ: Investigation, Writing – authentic draft, Writing – evaluate & modifying. SP: Investigation, Writing – authentic draft, Writing – evaluate & modifying. VV: Investigation, Writing – authentic draft, Writing – evaluate & modifying. MW: Investigation, Writing – authentic draft, Writing – evaluate & modifying. MS: Investigation, Writing – authentic draft, Writing – evaluate & modifying. BBr: Investigation, Writing – authentic draft, Writing – evaluate & modifying. DSu: Investigation, Writing – authentic draft, Writing – evaluate & modifying. SC: Investigation, Supervision, Validation, Writing – authentic draft, Writing – evaluate & modifying. DSv: Information curation, Investigation, Methodology, Sources, Supervision, Writing – authentic draft, Writing – evaluate & modifying. LD: Conceptualization, Formal evaluation, Investigation, Supervision, Validation, Writing – authentic draft, Writing – evaluate & modifying.
Funding
The creator(s) declare that no monetary assist was acquired for the analysis, authorship, and/or publication of this text.
Acknowledgments
We prefer to acknowledge Assoc. Prof. Milos Mikus for administrative assist and significant enter. We thank sufferers and their households for collaborating within the research.
Battle of curiosity
RD is employed by S&D Pharma.
The remaining authors declare that the analysis was performed within the absence of any business or monetary relationships that might be construed as a possible battle of curiosity.
Writer’s word
All claims expressed on this article are solely these of the authors and don’t essentially characterize these of their affiliated organizations, or these of the writer, the editors and the reviewers. Any product that could be evaluated on this article, or declare that could be made by its producer, just isn’t assured or endorsed by the writer.
References
1. Bossi P, Antonuzzo A, Cherny NI, Rosengarten O, Pernot S, Trippa F, et al. Diarrhoea in grownup most cancers sufferers: ESMO Scientific Apply Tips. Ann Oncol. (2018) 29:iv126–iv42. doi: 10.1093/annonc/mdy145
PubMed Summary | CrossRef Full Textual content | Google Scholar
3. Collinson S, Deans A, Padua-Zamora A, Gregorio GV, Li C, Dans LF, et al. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst Rev. (2020) 12:CD003048. doi: 10.1002/14651858.CD003048.pub4
PubMed Summary | CrossRef Full Textual content | Google Scholar
4. Danis R, Mego M, Antonova M, Stepanova R, Svobodnik A, Hejnova R, et al. Orally administered probiotics within the prevention of chemotherapy (+/- radiotherapy)-induced gastrointestinal toxicity: A scientific evaluate with meta-analysis of randomized trials. Integr Most cancers Ther. (2022) 21:15347354221144309. doi: 10.1177/15347354221144309
PubMed Summary | CrossRef Full Textual content | Google Scholar
5. Goodman C, Keating G, Georgousopoulou E, Hespe C, Levett Okay. Probiotics for the prevention of antibiotic-associated diarrhoea: a scientific evaluate and meta-analysis. BMJ Open. (2021) 11:e043054. doi: 10.1136/bmjopen-2020-043054
PubMed Summary | CrossRef Full Textual content | Google Scholar
6. Guo Q, Goldenberg JZ, Humphrey C, El Dib R, Johnston BC. Probiotics for the prevention of pediatric antibiotic-associated diarrhea. Cochrane Database Syst Rev. (2019) 4:CD004827. doi: 10.1002/14651858.CD004827.pub5
PubMed Summary | CrossRef Full Textual content | Google Scholar
7. Ferencik M, Ebringer L, Mikes Z, Jahnova E, Ciznar I. [Successful modification of human intestinal microflora with oral administration of lactic acid bacteria]. Bratisl Lek Listy. (1999) 100:238–45.
8. Bowen JM, Stringer AM, Gibson RJ, Yeoh AS, Hannam S, Keefe DM. VSL3 probiotic therapy reduces chemotherapy-induced diarrhea and weight reduction. Most cancers Biol Ther. (2007) 6:1449–54. doi: 10.4161/cbt.6.9.4622
PubMed Summary | CrossRef Full Textual content | Google Scholar
10. Sanders ME, Merenstein DJ, Reid G, Gibson GR, Rastall RA. Creator Correction: Probiotics and prebiotics in intestinal well being and illness: from biology to the clinic. Nat Rev Gastroenterol Hepatol. (2019) 16:642. doi: 10.1038/s41575-019-0199-6
PubMed Summary | CrossRef Full Textual content | Google Scholar
12. Michael M, Brittain M, Nagai J, Feld R, Hedley D, Oza A, et al. Section II research of activated charcoal to stop irinotecan-induced diarrhea. J Clin Oncol. (2004) 22:4410–7. doi: 10.1200/JCO.2004.11.125
PubMed Summary | CrossRef Full Textual content | Google Scholar
13. de Jong FA, Kehrer DF, Mathijssen RH, Creemers GJ, de Bruijn P, van Schaik RH, et al. Prophylaxis of irinotecan-induced diarrhea with neomycin and potential function for UGT1A1*28 genotype screening: a double-blind, randomized, placebo-controlled research. Oncologist. (2006) 11:944–54. doi: 10.1634/theoncologist.11-8-944
PubMed Summary | CrossRef Full Textual content | Google Scholar
14. Karthaus M, Ballo H, Abenhardt W, Steinmetz T, Geer T, Schimke J, et al. Potential, double-blind, placebo-controlled, multicenter, randomized part III research with orally administered budesonide for prevention of irinotecan (CPT-11)-induced diarrhea in sufferers with superior colorectal most cancers. Oncology. (2005) 68:326–32. doi: 10.1159/000086971
PubMed Summary | CrossRef Full Textual content | Google Scholar
15. Hu S, Ding Q, Zhang W, Kang M, Ma J, Zhao L. Intestine microbial beta-glucuronidase: a significant regulator in feminine estrogen metabolism. Intestine Microbes. (2023) 15:2236749. doi: 10.1080/19490976.2023.2236749
PubMed Summary | CrossRef Full Textual content | Google Scholar
16. Valenti Moreno V, Brunet Vidal J, Manzano Alemany H, Salud Salvia A, Llobera Serentill M, Cabezas Montero I, et al. Prevention of irinotecan related diarrhea by intestinal alkalization. A pilot research in gastrointestinal most cancers sufferers. Clin Transl Oncol. (2006) 8:208–12. doi: 10.1007/s12094-006-0012-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
17. Mego M, Chovanec J, Vochyanova-Andrezalova I, Konkolovsky P, Mikulova M, Reckova M, et al. Prevention of irinotecan induced diarrhea by probiotics: A randomized double blind, placebo managed pilot research. Complement Ther Med. (2015) 23:356–62. doi: 10.1016/j.ctim.2015.03.008
PubMed Summary | CrossRef Full Textual content | Google Scholar
18. Mego M, Danis R, Chovanec J, Jurisova S, Bystricky B, Porsok S, et al. Randomized double-blind, placebo-controlled multicenter part III research of prevention of irinotecan-induced diarrhea by a probiotic combination containing Bifidobacterium BB-12((R))Lactobacillus rhamnosus LGG((R)) in colorectal most cancers sufferers. Entrance Oncol. (2023) 13:1168654. doi: 10.3389/fonc.2023.1168654
PubMed Summary | CrossRef Full Textual content | Google Scholar
19. Lin XB, Dieleman LA, Ketabi A, Bibova I, Sawyer MB, Xue H, et al. Irinotecan (CPT-11) chemotherapy alters intestinal microbiota in tumour bearing rats. PloS One. (2012) 7:e39764. doi: 10.1371/journal.pone.0039764
PubMed Summary | CrossRef Full Textual content | Google Scholar
20. Yue B, Gao R, Wang Z, Dou W. Microbiota-host-irinotecan axis: A brand new perception towards irinotecan chemotherapy. Entrance Cell Infect Microbiol. (2021) 11:710945. doi: 10.3389/fcimb.2021.710945
PubMed Summary | CrossRef Full Textual content | Google Scholar
21. Qiu Y, Zhang J, Ji R, Zhou Y, Shao L, Chen D, et al. Preventative results of selenium-enriched Bifidobacterium longum on irinotecan-induced small intestinal mucositis in mice. Benef Microbes. (2019) 10:569–77. doi: 10.3920/BM2018.0096
PubMed Summary | CrossRef Full Textual content | Google Scholar
22. Sezer A, Usta U, Cicin I. The impact of Saccharomyces boulardii on decreasing irinotecan-induced intestinal mucositis and diarrhea. Med Oncol. (2009) 26:350–7. doi: 10.1007/s12032-008-9128-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
23. Wang Y, Solar L, Chen S, Guo S, Yue T, Hou Q, et al. The administration of Escherichia coli Nissle 1917 ameliorates irinotecan-induced intestinal barrier dysfunction and intestine microbial dysbiosis in mice. Life Sci. (2019) 231:116529. doi: 10.1016/j.lfs.2019.06.004
PubMed Summary | CrossRef Full Textual content | Google Scholar
24. Ghidini M, Nicoletti M, Ratti M, Tomasello G, Lonati V, Ghilardi M, et al. Lactobacillus kefiri LKF01 (Kefibios((R))) for prevention of diarrhoea in most cancers sufferers handled with chemotherapy: A potential research. Vitamins. (2021) 13. doi: 10.3390/nu13020385
PubMed Summary | CrossRef Full Textual content | Google Scholar
25. Sharma A, Chaudhary SP, Raina V, Shukla NK, Sreenivas V, Prakash S, et al. Remaining outcomes of a part II/III, randomized, double blind, placebo-controlled research to analyze the efficacy of a excessive efficiency multistrain probiotic, on chemotherapy induced diarrhea in most cancers sufferers receiving fluropyrimidines and/or irinotecan primarily based remedy. Ann Oncol. (2018) 29. doi: 10.1093/annonc/mdy424.085
26. Rigon-Zimmer Okay, Mullie C, Tir-Touil-Meddah A, Buisson P, Leke L, Canarelli JP. Impression of colostomy on intestinal microflora and bacterial translocation in younger rats fed with heat-killed Lactobacillus acidophilus pressure LB. Folia Microbiol (Praha). (2008) 53:89–93. doi: 10.1007/s12223-008-0013-2
PubMed Summary | CrossRef Full Textual content | Google Scholar
27. Sakai SA, Aoshima M, Sawada Okay, Horasawa S, Yoshikawa A, Fujisawa T, et al. Fecal microbiota in sufferers with a stoma decreases anaerobic micro organism and alters taxonomic and purposeful diversities. Entrance Cell Infect Microbiol. (2022) 12:925444. doi: 10.3389/fcimb.2022.925444
PubMed Summary | CrossRef Full Textual content | Google Scholar
28. Matsuzawa H, Munakata S, Kawai M, Sugimoto Okay, Kamiyama H, Takahashi M, et al. Evaluation of ileostomy stool samples reveals dysbiosis in sufferers with high-output stomas. Biosci Microbiota Meals Well being. (2021) 40:135–43. doi: 10.12938/bmfh.2020-062
PubMed Summary | CrossRef Full Textual content | Google Scholar
29. Jansma J, Thome NU, Schwalbe M, Chatziioannou AC, Elsayed SS, van Wezel GP, et al. Dynamic results of probiotic system ecologic(R)825 on human small intestinal ileostoma microbiota: a community idea strategy. Intestine Microbes. (2023) 15:2232506. doi: 10.1080/19490976.2023.2232506
PubMed Summary | CrossRef Full Textual content | Google Scholar