A collaborative mission to convey the promise of cell remedy to sufferers with a lethal type of mind most cancers has proven dramatic outcomes among the many first sufferers to obtain the novel therapy. In a paper printed at present in The New England Journal of Drugs, researchers from the Mass Normal Most cancers Middle, a member of the Mass Normal Brigham healthcare system, shared the outcomes for the primary three affected person instances from a part 1 scientific trial evaluating a brand new strategy to CAR-T remedy for glioblastoma (GBM). The trial, referred to as INCIPIENT, is designed to guage the security of CARv3-TEAM-E T cells in sufferers with recurrent GBM. Simply days after a single therapy, sufferers skilled dramatic reductions of their tumors, with one affected person attaining near-complete tumor regression. In time, the researchers noticed tumor development in these sufferers, however given the technique’s promising preliminary outcomes, the staff will pursue methods to increase the sturdiness of response.
“This can be a story of bench-to-bedside remedy, with a novel cell remedy designed within the laboratories of Massachusetts Normal Hospital and translated for affected person use inside 5 years, to fulfill an pressing want,” mentioned Bryan Choi, MD, PhD, neurosurgeon and affiliate director of the Middle for Mind Tumor Immunology and Immunotherapy, Mobile Immunotherapy Program, Mass Normal Most cancers Middle and Division of Neurosurgery. “The CAR-T platform has revolutionized how we take into consideration treating sufferers with most cancers, however strong tumors like glioblastoma have remained difficult to deal with as a result of not all most cancers cells are precisely alike and cells inside the tumor differ. Our strategy combines two types of remedy, permitting us to deal with glioblastoma in a broader, probably more practical approach.”
The brand new strategy is a results of years of collaboration and innovation springing from the lab of Marcela Maus, MD, PhD, director of the Mobile Immunotherapy Program on the Mass Normal Most cancers Middle, Paula J. O’Keeffe chair in Oncology, and school of the Krantz Household Middle for Most cancers Analysis. Maus’ lab has arrange a staff of collaborating scientists and skilled personnel to quickly convey subsequent era genetically modified T cells from the bench to scientific trials in sufferers with most cancers.
“We’ve made an funding in growing the staff to allow translation of our improvements in immunotherapy from our lab to the clinic, to rework look after sufferers with most cancers,” mentioned Maus. “These outcomes are thrilling, however they’re additionally just the start—they inform us that we’re heading in the right direction in pursuing a remedy that has the potential to alter the outlook for this intractable illness. We haven’t cured sufferers but, however that’s our audacious objective.”
Research like this one present the promise of cell remedy for treating incurable situations. Mass Normal Brigham’s Gene and Cell Remedy Institute, the place Maus is Affiliate Head & Head of Cell Therapies, helps to translate scientific discoveries made by researchers into first-in-human scientific trials and, finally, life-changing therapies for sufferers. The Institute’s multidisciplinary strategy units it aside from others within the house, serving to researchers to quickly advance new therapies and push the technological and scientific boundaries of this new frontier.
CAR-T remedy works by utilizing a affected person’s personal cells to battle most cancers—it is named probably the most personalised technique to deal with most cancers. A affected person’s cells are extracted, modified to provide proteins on their floor known as chimeric antigen receptors, after which injected again into the physique to focus on the tumor straight. Cells used on this examine had been manufactured by the Connell and O’Reilly Households Cell Manipulation Core Facility of the Dana-Farber/Harvard Most cancers Middle.
CAR-T therapies have been accepted for the therapy of blood cancers however the remedy’s use for strong tumors is restricted. Strong tumors include blended populations of cells, permitting some most cancers cells to proceed to evade the immune system’s detection, even after therapy with CAR-T. Maus’s staff is working to beat this problem of tumor heterogeneity with an modern technique that mixes two beforehand separate methods: CAR-T and bispecific antibodies, referred to as T-cell participating antibody molecules (TEAMs). The model of CAR-TEAM for glioblastoma is designed to be straight injected right into a affected person’s mind.
Maus and colleagues beforehand developed CAR-T cells to focus on a typical most cancers mutation referred to as EGFRvIII, however when that alone had restricted results, her staff engineered these CAR-T cells to ship TEAMs towards wild-type EGFR, which isn’t detected in regular mind tissue however is expressed in additional than 80 % of instances of GBM.
The mix strategy confirmed promise in preclinical fashions of glioblastoma, encouraging the analysis staff to pursue scientific translation. In collaboration with Mass Normal neurosurgeons and neuro-oncologists, together with Elizabeth Gerstner, MD, and William Curry, MD, in addition to specialists in cell remedy supply, Matthew Frigault, MD, and immunotherapy monitoring, Kathleen Gallagher, PhD, the staff launched INCIPIENT (ClinicalTrials.gov quantity, NCT05660369), a non-randomized, open label, single-site Section 1 examine.
Three sufferers had been enrolled within the examine between March 2023 and July 2023. Sufferers’ T cells had been collected and reworked into the brand new model of CAR-TEAM cells, which had been then infused again into every affected person. Sufferers had been monitored for toxicity all through the length of the examine.
All sufferers had been handled with standard-of-care radiation and temozolomide chemotherapy and had been enrolled within the trial after illness recurrence:
- A 74-year-old man had his tumor regress quickly, however transiently after a single infusion of the brand new CAR-TEAM cells. Blood and cerebrospinal fluid from the affected person confirmed a lower in EGFRvIII and EGFR copy numbers, finally turning into undetectable.
- A 72-year-old man was handled with a single infusion of CAR-TEAM cells. Two days after receiving CAR-TEAM cells, an MRI confirmed a lower within the tumor’s measurement by 18.5 %. By day 69, the tumor had decreased by 60.7 %, and the response was sustained for over 6 months.
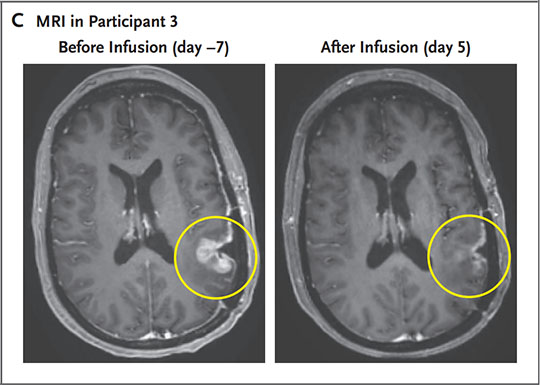
- A 57-year-old girl was handled with CAR-TEAM cells. An MRI 5 days after a single infusion of CAR-TEAM cells confirmed near-complete tumor regression.
The sufferers tolerated the infusions nicely, although almost all had fevers and altered psychological standing quickly after infusion, as was anticipated from an lively CAR-T remedy administered into the fluid across the mind. All sufferers had been noticed within the hospital earlier than discharge.
The authors notice that regardless of the outstanding responses among the many first three sufferers, they noticed eventual tumor development in all of the instances, although in a single case, there was no development for over six months. Development corresponded partly with the restricted persistence of the CAR-TEAM cells over the weeks following infusion. As a subsequent step, the staff is contemplating serial infusions or preconditioning with chemotherapy to extend the response.
“We report a dramatic and speedy response in these three sufferers. Our work thus far reveals indicators that we’re making progress, however there’s extra to do,” mentioned co-author Elizabeth Gerstner, MD, a neuro-oncologist within the Division of Neurology at Massachusetts Normal Hospital.
To study extra, go to mauslab.com.
If you’re desirous about studying extra in regards to the INCIPIENT scientific trial, please name 617-724-6226 or e mail carteamingbm@mgb.org. A member of our scientific staff will contact you inside 48 enterprise hours.
Authorship: Along with Choi, Maus and Gerstner, different authors embrace Matthew J. Frigault (MGH), Mark B. Leick (MGH), Christopher W. Mount (MGH), Leonora Balaj (MGH), Sarah Nikiforow (DFHCC), Bob S. Carter (MGH), William T. Curry (MGH), Kathleen Gallagher (MGH).
Disclosures: Disclosure kinds supplied by the authors is obtainable with the complete textual content of this text at NEJM.org.
Funding: This examine was supported by a grant to MVM from Gateway for Most cancers Analysis, the Mass Normal Most cancers Middle, Mass Normal Brigham, and philanthropic presents. Assist was additionally supplied by the Nationwide Gene Vector Biorepository at Indiana College which is funded beneath Nationwide Most cancers Institute contract HSN261201500003I Job Order No. HHSN26100077.
Paper cited: Choi BD et al. “Fast Regression of Recurrent Glioblastoma with CARv3-TEAM-E T Cells.” New England Journal of Drugs DOI: 10.1056/NEJMoa2314390
About Mass Normal Brigham
Mass Normal Brigham is an built-in educational well being care system, uniting nice minds to resolve the toughest issues in medication for our communities and the world. Mass Normal Brigham connects a full continuum of care throughout a system of educational medical facilities, group and specialty hospitals, a medical health insurance plan, doctor networks, group well being facilities, dwelling care, and long-term care providers. Mass Normal Brigham is a nonprofit group dedicated to affected person care, analysis, instructing, and repair to the group. As well as, Mass Normal Brigham is without doubt one of the nation’s main biomedical analysis organizations with a number of Harvard Medical College instructing hospitals. For extra info, please go to massgeneralbrigham.org.

