Introduction
Colorectal most cancers (CRC) happens when the cells within the colon or rectum’s lining develop uncontrollably, forming a tumor. By way of most cancers prevalence, colorectal most cancers is the third commonest most cancers and it’s the second main reason for cancer-related deaths worldwide (1). Roughly 20% of CRC sufferers have already got metastases on the time of analysis, and half of the sufferers with localized sickness ultimately develop metastases (2). This situation requires a number of therapy traces and is usually difficult to deal with because of the metastatic nature of the most cancers. Sufferers with metastatic CRC are usually believed to have a poor prognosis, with a relative 5-year survival price of 14%, in comparison with 90% and 71% in these with localized and regional CRC respectively (3). The evolution of simpler therapeutic strategies, together with liver and lung metastases surgical procedure, and new anticancer medicine, has resulted in a distinguished enchancment within the prognosis for metastatic CRC (mCRC) sufferers over the past 20 years (4). Regardless of that, it stays incurable generally. New analysis suggests the mixture of trifluridine and tipiracil with bevacizumab as a promising various for sufferers who’ve beforehand been handled for mCRC. The Meals and Drug Administration (FDA) has permitted this mixture remedy due to its promising outcomes (5). Quite a few medical trials have evaluated each the effectivity and security of this mixture remedy. On this evaluate, we talk about the pathogenesis and medical options of metastatic colorectal most cancers. Moreover, this evaluate goals to supply a complete overview of the efficacy and security profile of this mixture remedy within the administration of metastatic colorectal most cancers. Our objective is to contribute to the present understanding of this therapy choice, in the end enhancing affected person care and outcomes.
Metastatic colorectal most cancers
Colorectal most cancers (CRC) ranks because the third most recognized most cancers globally. Moreover, it’s the main reason for cancer-related deaths in each women and men, following lung most cancers. Regardless of in depth analysis, the precise causes of colorectal carcinoma stay unclear. Early detection of CRC performs a vital function in enhancing affected person outcomes; nevertheless, people with colorectal most cancers regularly current with atypical medical signs or solely obscure indicators through the preliminary levels of the illness, leading to a low early analysis price (6, 7).
Epidemiology
Colorectal most cancers (CRC) reveals geographical variation with extra circumstances reported in developed nations in comparison with much less developed ones. In 2023, the estimated variety of adults in the USA who will probably be recognized with colorectal most cancers is 153,020 (8). Worldwide, colorectal most cancers is the second most deadly most cancers for each women and men, following lung most cancers. Knowledge from Globocan 2020 reveals that CRC brought about 935,173 deaths, accounting for 9.4% of all cancer-related fatalities (9).
Etiology
The precise reason for colorectal most cancers (CRC) stays elusive, however a number of contributing elements have been recognized. Roughly 20% of CRC circumstances are related to genetic elements with first-generation family members of CRC sufferers dealing with a three-fold larger threat of growing most cancers. Genetic syndromes like Familial Adenomatous Polyposis (FAP) and Lynch syndrome have been linked to hereditary CRC. Non-cancerous situations like colorectal polyps, adenomas, ulcerative colitis, and Crohn’s illness can contribute to CRC improvement. Danger elements for CRC have been proven to incorporate age, carcinogenic exposures, smoking, race, gender, IBD, sedentary way of life, weight problems, and even pelvic radiation remedy (7, 10).
Signs
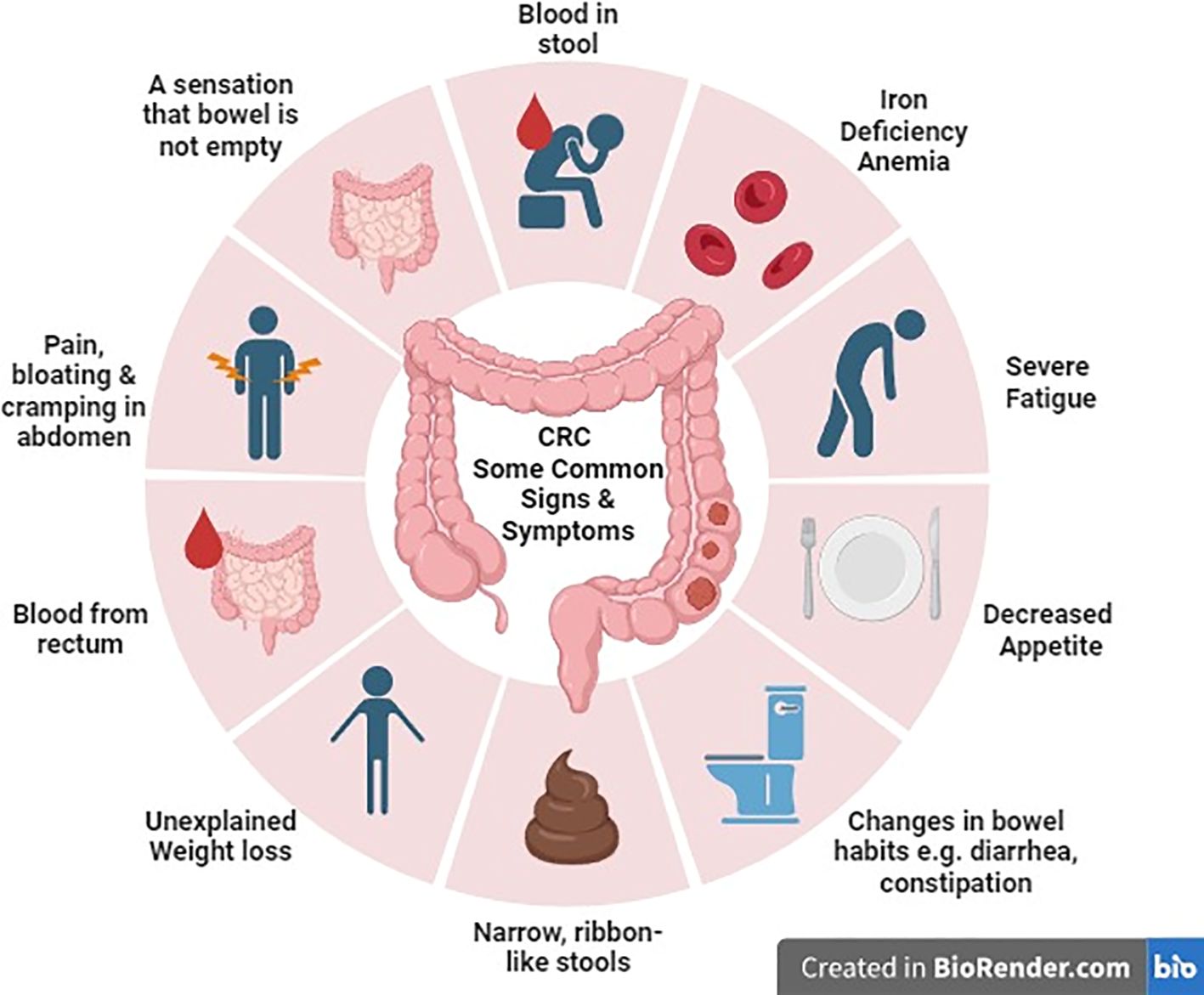
Widespread signs that people ought to concentrate on embrace alterations in bowel habits comparable to diarrhea, constipation, or narrowing of stool. Moreover, signs could embody rectal bleeding which might manifest as both shiny pink or darkish, tar-like stools. Different warning signs contain persistent belly discomfort, ache, or bloating, unexplained and sudden weight reduction, fixed fatigue and low power ranges even after sufficient relaxation, and iron deficiency anemia ensuing from ongoing bleeding, resulting in fatigue, weak spot, and pallor (11) (Determine 1).
In metastatic colorectal most cancers, signs can fluctuate based mostly on tumor dimension and the websites of metastasis. For instance, bone involvement could lead to ache, fractures, constipation, and elevated calcium ranges. Lung metastasis could result in signs like breathlessness, coughing, ache, and fatigue. Liver metastasis may cause nausea, fatigue, swelling, elevated belly dimension, and jaundice. Stomach lymph node involvement could lead to bloating, belly swelling, and lack of urge for food. Moreover, if metastasis happens in mind or spinal twine, it might manifest as ache, confusion, reminiscence points, complications, imaginative and prescient issues, speech difficulties, and seizures (12).
Pathogenesis
CRC is a multifactorial illness that begins as progressive modifications within the floor epithelial cells of colorectal mucosa. Hyperplasia and adenoma formation in these cells can ultimately advance into carcinoma. This improvement is often initiated by carcinogenic elements which trigger alterations in DNA. Morphology consists of epithelial hyperplasia, atypical hyperplasia, adenoma formation, carcinoma in situ, and invasive carcinoma (13).
With current improvements in analysis, notably, three confirmed molecular mechanisms underpin CRC’s incidence and improvement (Determine 2).
1. Chromosomal instability (CIN), primarily occurring in familial adenomatous polyposis (FAP), includes structural modifications in DNA, selling most cancers development by growing clonal variety.
2. Genetic mutations, comparable to these present in Lynch syndrome and different sporadic mismatch restore (MMR) mutations, play a job in CRC.
3. CpG islands hypermethylation, in particular gene promoter areas contributes to CRC improvement, generally involving the aberration of genes like APC, DCC, P53, Ok-RAS, c-MYC, MCC, and MMR-related genes.
Importantly, these molecular pathways could coexist in CRC sufferers (14). Moreover, genomic and epigenomic instabilities, together with chromosomal instability (CIN), microsatellite instability (MSI), non-MSI hypermutability, aberrant DNA methylation, and international DNA hypomethylation, considerably contribute to CRC formation. Understanding these instabilities in CRC, in addition to their implications for polyp-to-cancer development and prevention methods, is essential for advancing our data of this illness and enhancing affected person care (15).
Therapy for CRC could fluctuate relying on the stage of the most cancers and the extent of its unfold. These therapy modalities can embrace surgical procedures, radiation remedy, chemotherapy, and focused remedy. The survival charges additionally fluctuate considerably by stage. Due to this fact, early intervention is essential, particularly in circumstances of metastatic CRC (Stage 4) the place the five-year relative survival price is roughly 15.6% (16).
Previous medical therapy
The anti-EGFR remedy
Cetuximab and panitumumab are monoclonal antibodies developed to particularly deal with the EGFR (Epidermal Development Issue Receptor) pathway in people with metastatic colorectal most cancers, whether or not they have acquired prior therapy or are newly recognized, even encompassing these with confirmed refractory situations. Cetuximab is a hybrid antibody combining mouse and human parts, whereas panitumumab is totally humanized. The EGFR protein’s expression ranges, assessed by means of immunohistochemical evaluation, are a predictive issue for therapy response. It was assumed that sufferers with excessive EGFR expression would profit extra from these antibodies. Nonetheless, medical trials in different strong tumors revealed restricted predictive worth for EGFR expression (17, 18).
A number of medical trials have evaluated the effectiveness of cetuximab and panitumumab in treating metastatic colorectal most cancers. RAS and BRAF are essential genes within the EGFR signaling pathway, serving as biomarkers for mCRC. Mutations in these genes result in EGFR pathway activation, affecting the efficacy of anti-EGFR focused remedy (2). The part III randomized medical trials demonstrated that people with metastatic colorectal most cancers and mutations in codon 12 or 13 of the KRAS gene didn’t expertise any benefits from present process therapy with these antibodies. The presence of KRAS mutations emerged as a damaging predictor, signifying their resistance to anti-EGFR remedy. Sufferers with KRAS mutations displayed notably shorter intervals of progression-free intervals and no discernible survival profit (19–21).
Equally, combining these anti-EGFR therapies with XELOX-based chemotherapy regimens didn’t persistently present advantages (22). On this context, solely a slight benefit in response price was famous in people with wild-type KRAS tumors with none noteworthy enhancements within the total survival price. Moreover, research combining anti-EGFR and anti-VEGF therapies alongside chemotherapy didn’t yield optimistic outcomes (23).
Aflibercept
Aflibercept, or vascular endothelial development issue TRAP (VEGF TRAP), is a glycoprotein used as a secondary therapy choice for metastatic colorectal most cancers when the illness progresses after a previous oxaliplatin-based therapy routine. Its mechanism of motion includes binding VEGF-A extra strongly than the pure receptors, thus blocking the binding of endogenous ligands to their respective receptors. This motion inhibits VEGF receptor activation and endothelial cell proliferation, in the end stopping the expansion of latest vessels that offer tumors with oxygen and important vitamins. Aflibercept binds to PlGF (placental development issue), performing as a decoy receptor and sequestering PlGF. This prevents PlGF from exerting its angiogenic results and additional contributes to inhibiting new blood vessel formation. In a Velour medical trial, Aflibercept’s efficacy and security have been assessed in metastatic colorectal most cancers sufferers handled with Oxaliplatin or Avastin. The outcomes confirmed a major rise in survival price, from 12 months to 13.5 months. Moreover, there was a discount within the relative threat of dying and illness development by 24%. Nonetheless, regardless of these promising outcomes, resistance to Aflibercept therapy develops, thus lowering its advantages. Due to this fact, damaging facets needs to be rigorously evaluated within the total therapy plan (24, 25).
Ramucirumab
Ramucirumab is classed as an IgG1 humanized monoclonal antibody and serves as a secondary therapeutic choice for people recognized with metastatic colorectal carcinoma who skilled illness development after receiving customary therapy. Its goal web site is the Vascular Endothelial Development Issue Receptor-2, making it a novel inhibitor of angiogenesis when in comparison with different well-known anti-vascular brokers like bevacizumab and Aflibercept. Medical knowledge signifies that when mixed with FOLFIRI, Ramucirumab offers a major benefit in regards to the period of total survival price, with an extension of six weeks in comparison with the usual FOLFIRI therapy alone. Nonetheless, it’s value noting that the therapy with FOLFIRI/Ramucirumab did include some opposed occasions. The commonest ones embrace fatigue, neutropenia, hypertension, and thrombocytopenia. (26, 27).
Regorafenib
Regorafenib is a potent inhibitor of multi-kinase that regulates the expansion of most cancers cells by an intricate system of development elements and receptors. Regorafenib is a medicine used within the therapy of progressive metastatic colorectal most cancers (mCRC). To reinforce anticancer remedy’s effectiveness, blocking a number of pathways concerned on this course of is essential. These pathways embrace FGFR (Fibroblast Development Issue Receptor), which is important for cell differentiation and proliferation; PDGFR (Platelet Derived Development Issue Receptor), which performs a job in recruiting and maturing pericytes and the TIE-2 receptor, a tyrosine kinase enzyme that performs a job within the formation of latest blood vessels (angiogenesis), is current on endothelial cells and is essential for the event of blood vessels. In human tumors, substances that bind to TIE-2 receptors are regularly elevated. Therefore, multi-target inhibitors have emerged as a promising analysis space in most cancers therapy. In a Section III CORRECT trial, regorafenib was evaluated, evaluating metastatic colorectal most cancers sufferers with progressive illness after the preliminary therapy routine. Sufferers have been randomized to obtain regorafenib and placebo in a 2:1 ratio. Regardless of the preliminary promise, it’s vital to notice that Regorafenib didn’t meet its main aims in part III medical trials. Whereas concentrating on a number of pathways could seem logical, it doesn’t at all times translate into improved total survival (OS) charges for mCRC sufferers. Regorafenib has acquired authorization to be used as a third-line therapy choice for sufferers with restricted alternate options, notably these pre-treated with particular chemotherapy regimens. Nonetheless, its efficacy varies relying on particular person affected person elements, together with particular genetic mutations (28, 29).
Latest remedy
The Meals and Drug Administration has permitted the usage of LONSURF (trifluridine and tipiracil) together with bevacizumab for adults with metastatic colorectal most cancers (mCRC) who’ve beforehand acquired chemotherapy with fluoropyrimidine, oxaliplatin, and irinotecan, in addition to anti-VEGF organic remedy, and, if their RAS standing is wild-type, anti-EGFR remedy. It’s value noting that the FDA had beforehand permitted LONSURF as a standalone therapy for this indication again in September 2015 (30).
In keeping with the outcomes of the Section 3 SUNLIGHT trial, utilizing LONSURF together with bevacizumab has proven vital enhancements in total survival (OS) and progression-free survival (PFS) for sufferers with metastatic colorectal most cancers (mCRC) who’ve skilled illness development or can’t tolerate two earlier chemotherapy remedies, compared to utilizing LONSURF as a standalone therapy. These optimistic outcomes have resulted within the US Meals and Drug Administration designating this mixed therapy as a Breakthrough Remedy in August 2023 (31).
LONSURF® (trifluridine/tipiracil)
LONSURF consists of trifluridine (FTD), an analog of thymidine nucleoside, and tipiracil (TPI), an inhibitor of thymidine phosphorylase, in a exact molar ratio of 1:0.5 (or a weight ratio of 1:0.471) (32). Trifluridine undergoes a collection of phosphorylation steps inside cells and subsequently integrates into DNA inflicting structural harm and dysfunction within the DNA. That is thought-about the primary approach during which trifluridine exerts its anticancer results though it might additionally play a job in inhibiting the enzyme thymidylate synthase by way of trifluridine monophosphate (33).
After being taken orally, trifluridine will get quickly deactivated by thymidine phosphorylase within the intestines and liver, leading to low oral bioavailability. Nonetheless, when tipiracil hydrochloride, an orally lively inhibitor of thymidine phosphorylase, is run alongside trifluridine, it reduces the preliminary degradation of trifluridine throughout its preliminary journey by means of the physique, growing its systemic publicity (33).
Based mostly on the findings of a randomized part 2 research (J003–10040030), TAS-102 obtained approval in Japan within the yr 2014 (34). Subsequently, it gained approval from the US Meals and Drug Administration in 2015 and the European Medicines Company in 2016, supported by knowledge derived from the worldwide part 3 RECOURSE research (35).
Throughout the placebo-controlled RECOURSE trial in Section III, sufferers have been prescribed FTD/TPI at a beginning dosage of 35 mg/m². They took this orally twice a day for 5 days (Day 1–5) and once more for 5 extra days (Day 8–12) inside a 28-day therapy cycle. The research signifies that FTD/TPI therapy considerably improved median total survival (OS) in comparison with the placebo group (7.1 months versus 5.3 months; hazard ratio [HR]: 0.68). This optimistic end result was constant throughout numerous subgroups. Many of the sufferers receiving FTD/TPI skilled opposed occasions (AEs) of grade ≥3 severity. Nonetheless, lower than 10% of them reported no non-hematologic toxicities of grade ≥3. Sufferers reported grade ≥3 neutropenia, anemia, and thrombocytopenia in 38%, 18%, and 5% of circumstances, respectively, and solely 4% of them skilled febrile neutropenia (35). Though it demonstrates effectiveness in numerous affected person subcategories and displays a well-tolerated security profile, trifluridine-tipiracil monotherapy affords solely modest enhancements in total survival (36, 37).
LONSURF is allowed to be used in sufferers who’re adults and have been recognized with metastatic gastric or gastroesophageal junction adenocarcinoma. These sufferers will need to have been beforehand handled with at the very least two traces of chemotherapy, which included fluoropyrimidine, a platinum agent, and both a taxane or irinotecan. Moreover, if relevant, their therapy will need to have additionally included HER2/neu-targeted remedy (32).
Bevacizumab
Bevacizumab is a well-established cancer-fighting remedy that focuses on VEGF, which leads to the inhibition of angiogenesis. When used for strong tumors, it’s thought to “normalize” the event of irregular blood vessels inside tumors and improve the distribution of chemotherapy medicine to cancerous tissues (38). This concept is supported by animal research involving colorectal most cancers xenografts, which have demonstrated that when trifluridine is given alongside bevacizumab, it results in elevated ranges of phosphorylated trifluridine inside cells (39). Moreover, the inclusion of nintedanib, an angiokinase inhibitor improves the mixing of trifluridine into DNA (40).
Bevacizumab (Avastin), marketed as an anticancer drug, acquired its preliminary FDA approval in 2004. This approval licensed its utilization as a viable therapy various for sufferers with metastatic colon or rectal carcinoma within the first or second line of remedy, alongside intravenous 5-fluorouracil-based chemotherapy (41). Tebutt’s research discovered that when instantly in comparison with capecitabine, therapy with Bevacizumab confirmed an extended Development-Free Survival (PFS) period of 8.5 months in comparison with 5.7 months. Nonetheless, there have been no vital variations noticed in Total Survival (OS), which remained at 18.9 months for each teams (42). The Hurwitz trial aimed to substantiate the earlier findings by evaluating IFL to IFL with Bevacizumab (BEVA). The trial concluded that mixture remedy was superior in numerous facets, together with the next total response price of 45% (in comparison with 35%), an prolonged Development-Free Survival (PFS) period of 10.6 months (in comparison with 6.2 months), and an extended Total Survival (OS) period of 20.3 months (in comparison with 15.6 months) (43). The approval from FDA for the usage of BEVA on this therapy context was granted because of the compelling outcomes noticed within the performed research.
Groundbreaking FDA approval: LONSURF® and bevacizumab combo for metastatic colorectal most cancers in adults
“Trifluridine and tipiracil (marketed by Taiho Oncology, Inc. as LONSURF) together with bevacizumab acquired FDA approval on August 2, 2023, for treating metastatic colorectal most cancers (mCRC) that was beforehand managed with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy, in addition to anti-VEGF organic remedy. It’s also permitted for mCRC sufferers with RAS wild-type who acquired anti-EGFR remedy” (30).
LONSURF, a mixture remedy administered orally to deal with metastatic colorectal most cancers, operates by means of the collaborative affect of trifluridine (FTD) and tipiracil (TPI). FTD, an analog with cytotoxic properties, exerts its anti-cancer results by integrating into DNA and inducing structural harm. Nonetheless, its effectiveness is hindered by fast enzymatic degradation facilitated by thymidine phosphorylase within the intestines and liver. Incorporating TPI, a thymidine phosphorylase inhibitor, mitigates this degradation, leading to an augmented systemic publicity to FTD. Moreover, the continual inhibition of angiogenesis emerges as a pivotal technique in managing metastatic colorectal most cancers. Bevacizumab, famend for its skill to focus on vascular endothelial development issue (VEGF), assumes a crucial function on this context by impeding angiogenesis. Demonstrating medical exercise even past illness development, the persistent inhibition of VEGF with bevacizumab proves efficient in sufferers with metastatic colorectal most cancers. By means of the mixing of FTD–TPI with bevacizumab, this therapeutic strategy not solely harnesses the cytotoxic potential of FTD but additionally prolongs the suppression of angiogenesis, presenting a synergistic technique for treating metastatic colorectal most cancers. This twin intervention comprehensively addresses the direct influence on most cancers cells and the supportive microenvironment, culminating in a complete and productive therapeutic end result (31).
The pairing of FTD/TPI and bevacizumab has demonstrated effectiveness in treating colorectal most cancers (CRC) in mouse xenograft fashions. Furthermore, it has exhibited promising response within the administration of superior mCRC by means of two preliminary medical trials initiated by investigators (39, 44, 45). In each medical trials (44, 46), the dose assigned and therapy schedule of FTD/TPI mirrored that of the RECOURSE trial (35), Inside every 28-day therapy cycle, bevacizumab was given intravenously at a dose of 5 mg/kg on days 1 and 15.
The preliminary trial, generally known as C-TASK FORCE (44), was performed in Japan in 2014. This single-arm research concerned 25 sufferers with metastatic CRC who had turn into proof against all customary therapies. After 16 weeks, the progression-free survival (PFS) price stood at 42.9% (with an 80% confidence interval between 27.8% and 59.0%). The median PFS was 3.7 months (with a 95% confidence interval starting from 2.0 to five.4 months), whereas the median total survival (OS) was 11.4 months (with a 95% confidence interval between 7.6 and 13.9 months). Notably, grade ≥3 neutropenia was noticed in 18 sufferers (72%), though the incidence of febrile neutropenia was comparatively low, affecting solely 4 sufferers (16%). Importantly, no opposed occasions related to the remedy have been noticed resulting in therapy discontinuation (44).
One other trial, performed in Denmark from 2017 to 2018, 93 sufferers with refractory metastatic CRC have been randomly assigned in a 1:1 ratio to obtain both FTD/TPI mixed with bevacizumab or FTD/TPI alone (46). In a survival evaluation performed with knowledge as much as February 2019, it was noticed that FTD/TPI together with bevacizumab led to considerably extended median progression-free survival (PFS) and total survival (OS) when in comparison with FTD/TPI monotherapy. Particularly, the outcomes confirmed a distinction in PFS (4.6 vs. 2.6 months; p = 0.0015) and OS (9.4 vs. 6.7 months; p = 0.028) in favor of the FTD/TPI plus bevacizumab group. Importantly, this disparity in OS continued even after an extra yr of follow-up (9.9 vs. 6.0 months; p = 0.03) (47). Though grade ≥3 neutropenia occurred extra regularly amongst sufferers receiving FTD/TPI plus bevacizumab, it’s noteworthy that treatment-related severe opposed occasions have been rare in each therapy teams (47).
The protection and effectiveness of LONSURF with bevacizumab have been assessed within the SUNLIGHT trial (NCT04737187), a worldwide research involving 492 people with metastatic colorectal most cancers (mCRC). These sufferers had acquired a most of two prior chemotherapy regimens and both skilled illness development or had intolerance to their final therapy. The primary objectives of the research revolved round assessing total survival (OS) and progression-free survival (PFS) as key end result measures. Statistically vital enchancment was noticed in OS amongst sufferers who have been randomized to obtain LONSURF plus bevacizumab in comparison with those that acquired LONSURF alone (Hazard ratio 0.61; 95% CI: 0.49, 0.77; one-sided p<0.001). Median OS was 10.8 months within the LONSURF plus bevacizumab group (95% CI: 9.4, 11.8) and seven.5 months within the LONSURF group (95% CI: 6.3, 8.6). Median PFS was 5.6 months within the LONSURF plus bevacizumab group (95% CI: 4.5, 5.9) and a couple of.4 months within the LONSURF group (95% CI: 2.1, 3.2) (Hazard ratio 0.44; 95% CI: 0.36, 0.54; one-sided p<0.001) (31). The enhancements in OS and PFS noticed with the mixture of LONSURF and bevacizumab have been linked to the preservation of the sufferers’ quality-of-life from the start of the therapy till cycle 6. Notably, there have been no noteworthy alterations in imply scores inside any of the subdomains assessed within the EORTC QLQ-C30 and EuroQol EQ-5D-5L health-related quality-of-life questionnaires (45). The regularly noticed opposed occasions and laboratory abnormalities in LONSURF plus bevacizumab-treated sufferers (occurring in ≥20% of circumstances) included neutropenia, anemia, thrombocytopenia, fatigue, nausea, elevated AST, elevated ALT, elevated alkaline phosphatase, low sodium, diarrhea, belly ache, and decreased urge for food (31).
This coupling of LONSURF with bevacizumab didn’t elevate the chance of significant opposed occasions or occasions resulting in therapy discontinuation. When FTD–TPI was mixed with bevacizumab, superior survival outcomes have been noticed in comparison with FTD–TPI alone, notably in most prespecified subgroups of sufferers. The protection profile of FTD–TPI plus bevacizumab aligned with expectations, and people who acquired the mixture exhibited a chronic preservation of efficiency standing in comparison with these handled solely with FTD–TPI. This compelling physique of proof strongly helps the rationale for adopting the trifluridine/tipiracil and bevacizumab mixture as a promising and well-tolerated therapeutic strategy within the administration of metastatic colorectal most cancers.
Medical trials exhibiting results of bevacizumab plus trifluridine/tipiracil on pre-treated colorectal most cancers
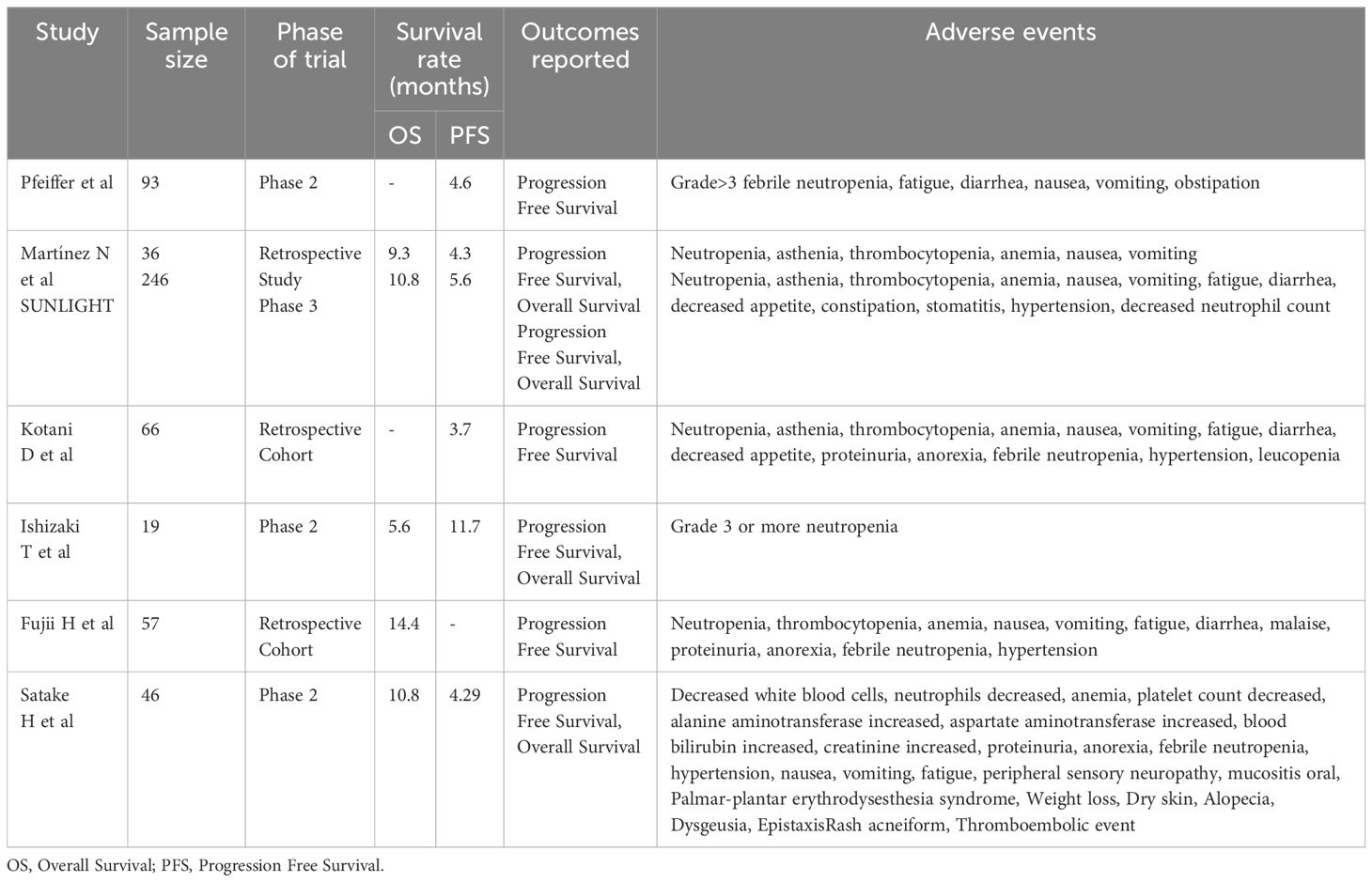
Proof from a number of medical trials and preclinical research has demonstrated the mixture of trifluridine and tipiracil together with bevacizumab as a exceptional therapy routine for closely pretreated metastatic colorectal most cancers (mCRC). In keeping with a part 2 trial carried out by Pfeiffer et al. that recruited round 93 sufferers, TAS-102 with bevacizumab demonstrated clinically notable enhancement in median progression-free survival from 2.6 months as a result of TAS-102 monotherapy to 4.6 months within the administration of metastatic colorectal most cancers that was unmanageable by means of chemotherapy together with exceptionally well-tolerated security profile. By way of security, the mixture group was comparatively safer than the monotherapy apart from the extreme neutropenia which was larger within the mixture group. Because the efficacy of trifluridine and tipiracil has been proven by quite a few trials, the intention of this research was to display its impact together with bevacizumab, nevertheless, small variety of randomised sufferers along with evaluation of PFS by investigator and lack of unbiased committee’s validation might restrict the reliability of outcomes (47).
Contemplating the evaluation of various research, a retrospective observational evaluation, 36 sufferers insensitive to mainstay remedy got TAS-102 2 occasions a day for days one to 5 and eight to 12 for 4 weeks with Bevacizumab on the primary and fifteenth day. Many sufferers acquired this therapy as third-line remedy. After a median follow-up of 11.6 months, the median progression-free survival was 4.3 months, and the median OS was 9.3 months. The remedy confirmed opposed results of Neutropenia (74.3%), asthenia (65.7%), anemia (54.8%) and thrombocytopenia (34.3%) with no experiences of febrile neutropenia and deaths associated to the therapy. This research reveals the efficacy of TAS-102 with bevacizumab in topics with refractory CRC in common medical use (48).
Imposing the outcomes of the aforementioned trial, FTD/TPI and bevacizumab mixture in a worldwide part 3 SUNLIGHT trial that included a complete of 246 sufferers, notably prolonged the PFS from 2.4 months to five.6 months. Furthermore, it confirmed vital prolongation of total survival from 7.5 months with a 30% price at 12 months with out bevacizumab to 10.8 months with a 43% price with bevacizumab. It’s notably noteworthy as a result of that is the one trial that has exhibited the clinically vital efficacy of bevacizumab after second line remedy as a 3rd line remedy. Moreover, it exceptionally made the results of this mixture evident in sufferers during which RAS mutation was carried by round 69% of topics, the next than generally recorded proportion of any such mutation in distinction to different trials which established solely marginal total survival advantages in RAS mutated CRC sufferers. Security outcomes in each teams have been comparable, nevertheless, as noticed beforehand, extreme neutropenia was reported to be larger within the mixture group than within the monotherapy group (49). These supreme modifications in survival price provoke the accelerated endorsement of this mixture by the Meals and Drug Administration (FDA) (50).
With a purpose to examine the efficacy and security of Trifluridine/Tipiracil plus bevacizumab in sufferers with closely pretreated mCRC, a retrospective cohort research was performed during which 66 sufferers enrolled with CRC acquired Trifluridine/Tipiracil with Bevacizumab remedy. The research confirmed vital enhance within the Median progression-free survival as 3.7 months with the mixture remedy whereas it was reported to be 2.2 months in sufferers receiving the monotherapy and the PFS price at 16 weeks was reported to be 46.6% and 33.9% respectively. The research reveals that sufferers within the Trifluridine/Tipiracil plus bevacizumab had considerably longer PFS and median OS. For security, the remedy reported a comparatively larger prevalence of grade >3 Neutropenia in individuals with pretreated CRC receiving the mixture remedy than within the Trifluridine/Tipiracil monotherapy group as noticed beforehand. Due to this fact, this research reveals that trifluridine/tipiracil plus bevacizumab helps lengthen PFS in sufferers with beforehand handled mCRC exhibiting tolerable opposed impact (51).
To evaluate the security of Trifluridine/Tipiracil with bevacizumab as a mixture remedy for sufferers with metastatic colorectal most cancers, a the part II research with 19 sufferers receiving the mixture therapy of Trifluridine/Tipiracil with bevacizumab was performed. All of the sufferers acquired the mixture remedy biweekly as third-line chemotherapy. The research indicated the median progression-free survival was 5.6 months and OS was 11.5 months. 5 sufferers acquired response and the management price of illness got here out to be 9 out of 12. Solely a single affected person encountered treatment-induced unwanted side effects of neutropenia (grade 3 or above) which consequently highlights the security of the remedy. Therefore, this research reveals that the mixture of TAS-102 with bevacizumab accelerates tumor depreciation by lowering the prevalence of neutropenia, enhancing survival, and represents an efficient therapeutic alternative for advanced-stage mCRC sufferers present process third-line therapy or past (52).
On prime of its extraordinary impact on survival, mixture of TAS-102 with bevacizumab possesses distinctive antitumor exercise than both agent alone as proved by a research revealed in Oncology experiences the place this composition exhibited higher outcomes on relative most cancers quantity following fourteen days after drug consumption and interval consumed for the relative most cancers quantity to develop 5 occasions (53).
To guage whether or not the efficacy of TAS-102 may very well be improved by combining it with Bevacizumab, in a retrospective research, a complete of 57 sufferers have been noticed. 21 sufferers have been administered TAS-102 with bevacizumab and 36 sufferers administered TAS-102 with out bevacizumab have been enrolled. The median OS was recorded at 14.4 months within the T-B class (sufferers receiving TAS-120 plus bevacizumab) and 4.5 months within the T group (sufferers receiving TAS-102 monotherapy). The median TTF was additionally considerably longer in T-B group (5.6 months) than in T group (2.1 months). Evaluating the efficacy and security, the research recorded a larger incidence of hypertension within the T-B class than within the T class (23.8% vs. 0.0%, p = 0.005). The polytherapy of TAS-102 with bevacizumab considerably enhanced the general survival, excessive antitumor exercise and different indicators, related to improved medical outcomes in topics with mCRC (54).
Consequently, within the part Ib/II trial evaluating biweekly mixture remedy with TAS-102 and BEVA, 46 sufferers illiberal to chemotherapy got TAS-102 with bevacizumab. The median progression-free survival was reported to be 4.29 months and total survival was 10.86 months. The illness management price is 59.1% with opposed results comparable to leukopenia (15.9%), neutropenia (15.9%), and hypertension (40.9%). This research concludes that TAS-102 plus Bevacizumab encourages anti-cancer efficacy with a positive security profile (55).
In addition to, a pooled evaluation of 29 research performed by Yoshino et al. displayed its medical efficacy as DCR related to the mixture was 64% than FTD/TPI alone, equally, increment in median PFS was seen from 2.6 to 4.2 months, 3% to 9% at 12 months, 8.1 months to 9.8 months of median OS and 38% from 32% at 12 months. Nonetheless, large hole between sufferers within the intervention group (n=289) and within the management group (n=9383), excessive heterogeneity and publication bias could function confounding bias and problem the authenticity of those outcomes, notably vital impact of heterogeneity on pooled Kaplan- Meier curves could affect the correct interpretation of the reported outcomes. Security profiles of each teams have been comparable apart from grade 3 neutropenia which was larger in FTD/TPI + BEVA group than within the monotherapy group, nevertheless, these outcomes usually are not fir for medical implication owing to inconsistency within the reporting of opposed occasions throughout the research (56). These medical trials exhibiting the important thing findings of medical trials on Trifluridine/Tipiracil plus Bevacizumab are talked about in Desk 1.
Desk 1 Abstract of key findings of the included medical trials on Trifluridine/Tipiracil plus Bevacizumab.
Advances in therapeutic methods for inflammatory bowel disease-associated colorectal most cancers: focused interventions and prognostic insights
Inflammatory bowel illness (IBD) consists of ulcerative colitis (UC) and Crohn’s illness (CD). IBD is related to elevated threat of CRC as a result of power irritation, genetic elements, and disrupted intestine microbiota interactions. This elevated CRC threat is pushed by inflammation-induced dysplasia, resulting in colitis-associated CRC (IBD-CRC) (57). Because of aggressive histological traits and decreased resectability of the tumors, the prognosis of IBD-CRC tends to be extra unfavorable than sporadic CRC. Persistent irritation results in particular molecular mechanisms, which trigger oxidative stress-related harm and DNA double-strand breaks. It will definitely results in growing ranges of molecular markers linked to oxidative damage (58). Nonetheless, the exact pathogenesis of IBD-related CRC stays a topic of ongoing analysis.
Each sporadic CRC and IBD-CRC share pathogenic options comparable to chromosomal and microsatellite instabilities, together with DNA hypermethylation. Nonetheless, not like sporadic CRC, IBD-CRC usually doesn’t comply with the traditional adenoma–carcinoma sequence. As an alternative, it progresses from low-grade dysplasia (LGD) to high-grade dysplasia (HGD) and ultimately to CRC (58). Latest analysis suggests another “Massive Bang” mannequin of CRC improvement within the context of irritation. On this mannequin, a number of tumor-initiating mutations come up concurrently in a single clone reasonably than progressively accumulating over time. This accelerated mutagenesis is believed to end result from colon irritation, resulting in abrupt tumor initiation (59).
Owing to the clear correlation between IBD and CRC, utilization of medication primarily immunosuppressant and non-steroidal anti-inflammatory medicine might considerably pave the best way in lowering the chance of IBD- associated CRC. Whereas figuring out numerous prognostic elements for colorectal most cancers in sufferers with IBD, a meta-analysis of 164 research, performed by Wijnands AM et al. demonstrated decreased risk of CRC in sufferers who acquired 5- Amino salicylic acid (5- ASA) (60). Imposing these outcomes, one other meta-analysis that included round 15,000 topics confirmed 5-ASA to have chemo preventive impact within the improvement of CRC in IBD sufferers with optimum upkeep dosage of 1.2g/day or extra (61). Not solely 5-ASA, thiopurines additionally play a vital function in decreasing the chance of CRC in IBD sufferers, as indicated by the decreased beta catenin exercise after topical administration of thiopurine thioguanine in a murine mannequin of colitis related most cancers (62). Equally, one other pooled evaluation of 11 cohorts and 16 case management research displayed protecting results of thiopurines in opposition to CRC in IBD sufferers, notably in those that had extended period of sickness (greater than 8 years) (63). Anti-tumor necrosis issue alpha brokers have additionally been exceptional in prevention of CRC in IBD sufferers. A big multicenter research evaluating 62,007,510 sufferers confirmed that IBD sufferers handled with anti- TNF brokers had much less probability to develop CRC. (64) Ongoing analysis is essential for understanding IBD-CRC mechanisms, growing new biomarkers, and implementing early detection methods to mitigate the influence of those cancers on public well being.
Conclusion
The mixture remedy of trifluridine and tipiracil with bevacizumab has exhibited promising outcomes within the therapy of beforehand handled metastatic colorectal most cancers. This complete overview has proven the efficacy of mixture remedy in enhancing the well being of mCRC sufferers. Along with that, remedy with trifluridine and tipiracil together with bevacizumab has a manageable security profile, with probably the most traditional opposed problems being neutropenia, fatigue, and diarrhea. The FDA has permitted this mixture remedy because it expands the therapy choices for sufferers with mCRC and helps the general survival of those people. On the similar time, it’s nonetheless vital to notice that this remedy might not be appropriate for each affected person, and an individualized therapy plan needs to be thought-about for such sufferers. Continued approval of this remedy will depend on the medical advantages and beneficial prognosis, so additional analysis and medical trials needs to be performed to achieve most efficacy.
Creator contributions
TR: Conceptualization, Supervision, Writing – unique draft. RR: Writing – unique draft. TS: Writing – unique draft, Writing – evaluate & enhancing. AS: Writing – unique draft. AK: Writing – unique draft. HZ: Writing – unique draft.
Funding
The authors declare that no monetary help was acquired for the analysis, authorship, and/or publication of this text.
Battle of curiosity
The authors declare that the analysis was performed within the absence of any industrial or monetary relationships that may very well be construed as a possible battle of curiosity.
Writer’s be aware
All claims expressed on this article are solely these of the authors and don’t essentially characterize these of their affiliated organizations, or these of the writer, the editors and the reviewers. Any product which may be evaluated on this article, or declare which may be made by its producer, just isn’t assured or endorsed by the writer.
References
3. Wang J, Li S, Liu Y, Zhang C, Li H, Lai B. Metastatic patterns and survival outcomes in sufferers with stage IV colon most cancers: A population-based evaluation. Most cancers Med. (2020) 9:361–73. doi: 10.1002/CAM4.2673
PubMed Summary | CrossRef Full Textual content | Google Scholar
4. Ciardiello F, Ciardiello D, Martini G, Napolitano S, Tabernero J, Cervantes A. Medical administration of metastatic colorectal most cancers within the period of precision drugs. CA Most cancers J Clin. (2022) 72:372–401. doi: 10.3322/CAAC.21728
PubMed Summary | CrossRef Full Textual content | Google Scholar
7. Duan B, Zhao Y, Bai J. Colorectal most cancers: an outline. Gastrointestinal Cancers. (2022), 301–12. doi: 10.36255/EXON-PUBLICATIONS-GASTROINTESTINAL-CANCERS-COLORECTAL-CANCER
14. Duan B, Zhao Y, Bai J, Duan X, Luo X, Zhang R, et al. Colorectal most cancers: an outline. Gastrointestinal Cancers. (2022), 1–12. doi: 10.36255/EXON-PUBLICATIONS-GASTROINTESTINAL-CANCERS-COLORECTAL-CANCER
15. Grady WM, Markowitz SD. The molecular pathogenesis of colorectal most cancers and its potential software to colorectal most cancers screening. Dig Dis Sci. (2015) 60:762–72. doi: 10.1007/S10620-014-3444-4/METRICS
PubMed Summary | CrossRef Full Textual content | Google Scholar
18. Martinetti D, Costanzo R, Kadare S, Alimehmeti M, Colarossi C, Canzonier V, et al. KRAS and BRAF mutational standing in colon most cancers from Albanian sufferers. Diagn Pathol. (2014) 9:1–7. doi: 10.1186/S13000-014-0187-7/TABLES/3
PubMed Summary | CrossRef Full Textual content | Google Scholar
20. Sartore-Bianchi A, Moroni M, Veronese S, Carnaghi C, Bajetta E, Luppi G, et al. Epidermal development issue receptor gene copy quantity and medical end result of metastatic colorectal most cancers handled with panitumumab. J Clin Oncol. (2007) 25:3238–45. doi: 10.1200/JCO.2007.11.5956
PubMed Summary | CrossRef Full Textual content | Google Scholar
21. Lièvre A, Bachet JB, Le Corre D, Boige V, Landi B, Emile JF, et al. KRAS mutation standing is predictive of response to cetuximab remedy in colorectal most cancers. Most cancers Res. (2006) 66:3992–5. doi: 10.1158/0008-5472.CAN-06-0191
PubMed Summary | CrossRef Full Textual content | Google Scholar
22. Msc H, Mrcp M, Frcpath J, Meade AM, Seymour MT, Wilson RH, et al. Addition of cetuximab to oxaliplatin-based fi rst-line mixture chemotherapy for therapy of superior colorectal most cancers: outcomes of the randomised part 3 MRC COIN trial. Lancet. (2011) 377:2103–14. doi: 10.1016/S0140
PubMed Summary | CrossRef Full Textual content | Google Scholar
23. CALGB/SWOG 80405: Section III trial of irinotecan/5-FU/leucovorin (FOLFIRI) or oxaliplatin/5-FU/leucovorin (mFOLFOX6) with bevacizumab (RV) or cetuximab (CET) for sufferers (pts) with KRAS wild-type (wt) untreated metastatic adenocarcinoma of the colon or rectum (MCRC). Students@Duke publication. Obtainable on-line at: https://students.duke.edu/publication/1085517 (Accessed September 10, 2023).
24. Chiron M, Bagley RG, Pollard J, Mankoo PK, Henry C, Vincent L, et al. Differential antitumor exercise of aflibercept and bevacizumab in patient-derived xenograft fashions of colorectal most cancers. Mol Most cancers Ther. (2014) 13:1636–44. doi: 10.1158/1535-7163.MCT-13-0753
PubMed Summary | CrossRef Full Textual content | Google Scholar
25. Van Cutsem E, Tabernero J, Lakomy R, Prenen H, Prausová J, Macarulla T, et al. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a part III randomized trial in sufferers with metastatic colorectal most cancers beforehand handled with an oxaliplatin-based routine. J Clin Oncol. (2012) 30:3499–506. doi: 10.1200/JCO.2012.42.8201
PubMed Summary | CrossRef Full Textual content | Google Scholar
26. Sims TN, Gao B, Phillips R, Lowy I. Potential predictive and prognostic biomarkers recognized in baseline plasma samples from the VELOUR trial. J Clin Oncol. (2015) 33:638–8. doi: 10.1200/JCO.2015.33.3_SUPPL.638
27. Tabernero J, Cohn AL, Obermannova R, Bodoky G, Garcia-Carbonero R, Ciuleanu T-E, et al. RAISE: A randomized, double-blind, multicenter part III research of irinotecan, folinic acid, and 5-fluorouracil (FOLFIRI) plus ramucirumab (RAM) or placebo (PBO) in sufferers (pts) with metastatic colorectal carcinoma (CRC) progressive throughout or following first-line mixture remedy with bevacizumab (bev), oxaliplatin (ox), and a fluoropyrimidine (fp). J Clin Oncol. (2015) 33(3_suppl):512–2. doi: 10.1200/JCO.2015.33.3_SUPPL.512
28. Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, et al. Regorafenib monotherapy for beforehand handled metastatic colorectal most cancers (CORRECT): A global, multicentre, randomised, placebo-controlled, part 3 trial. Lancet. (2013) 381:303–12. doi: 10.1016/S0140-6736(12)61900-X
PubMed Summary | CrossRef Full Textual content | Google Scholar
31. Prager GW, Taieb J, Fakih M, Ciardiello F, Cutsem EV, Elez E, et al. Trifluridine-tipiracil and bevacizumab in refractory metastatic colorectal most cancers. N Engl J Med. (2023) 388:1657–67. doi: 10.1056/NEJMOA2214963
PubMed Summary | CrossRef Full Textual content | Google Scholar
34. Yoshino T, Mizunuma N, Yamazaki Ok, Nishina T, Komatsu Y, Baba H, et al. TAS-102 monotherapy for pretreated metastatic colorectal most cancers: a double-blind, randomised, placebo-controlled part 2 trial. Lancet Oncol. (2012) 13:993–1001. doi: 10.1016/S1470-2045(12)70345-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
35. Mayer RJ, Van Cutsem E, Falcone A, Yoshino T, Garcia-Carbonero R, Mizunuma N, et al. Randomized trial of TAS-102 for refractory metastatic colorectal most cancers. N Engl J Med. (2015) 372:1909–19. doi: 10.1056/NEJMOA1414325
PubMed Summary | CrossRef Full Textual content | Google Scholar
36. Van Cutsem E, Mayer RJ, Laurent S, et al. The subgroups of the part III RECOURSE trial of trifluridine/tipiracil (TAS-102) versus placebo with finest supportive care in sufferers with metastatic colorectal most cancers. Eur J Most cancers. (2018) 90:63–72. doi: 10.1016/J.EJCA.2017.10.009
PubMed Summary | CrossRef Full Textual content | Google Scholar
37. Van Cutsem E, Falcone A, Garcia-Carbonero R, Komatsu Y, Pastorino A, Peeters M, et al. Proxies of high quality of life in metastatic colorectal most cancers: analyses within the RECOURSE trial. ESMO Open. (2017) 2(5):E000261. doi: 10.1136/ESMOOPEN-2017-000261
PubMed Summary | CrossRef Full Textual content | Google Scholar
39. Tsukihara H, Nakagawa F, Sakamoto Ok, Ishida Ok, Tanaka N, Okabe H, et al. Efficacy of mixture chemotherapy utilizing a novel oral chemotherapeutic agent, TAS-102, along with bevacizumab, cetuximab, or panitumumab on human colorectal most cancers xenografts. Oncol Rep. (2015) 33:2135–42. doi: 10.3892/OR.2015.3876
PubMed Summary | CrossRef Full Textual content | Google Scholar
40. Suzuki N, Nakagawa F, Matsuoka Ok, Takechi T. Impact of a novel oral chemotherapeutic agent containing a mixture of trifluridine, tipiracil and the novel triple angiokinase inhibitor nintedanib, on human colorectal most cancers xenografts. Oncol Rep. (2016) 36:3123–30. doi: 10.3892/OR.2016.5208
PubMed Summary | CrossRef Full Textual content | Google Scholar
42. Tebbutt NC, Wilson Ok, Gebski VJ, Zannino D, van Hazel GA, Robinson B, et al. Capecitabine, bevacizumab, and mitomycin in first-line therapy of metastatic colorectal most cancers: outcomes of the Australasian Gastrointestinal Trials Group Randomized Section III MAX Examine. J Clin Oncol. (2010) 28:3191–8. doi: 10.1200/JCO.2009.27.7723
PubMed Summary | CrossRef Full Textual content | Google Scholar
43. Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal most cancers. N Engl J Med. (2004) 350:2335–42. doi: 10.1056/NEJMOA032691
PubMed Summary | CrossRef Full Textual content | Google Scholar
44. Kuboki Y, Nishina T, Shinozaki E, et al. TAS-102 plus bevacizumab for sufferers with metastatic colorectal most cancers refractory to plain therapies (C-TASK FORCE): an investigator-initiated, open-label, single-arm, multicentre, part 1/2 research. Lancet Oncol. (2017) 18:1172–81. doi: 10.1016/S1470-2045(17)30425-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
45. Pfeiffer P, Yilmaz M, Möller S, et al. TAS-102 with or with out bevacizumab in sufferers with chemorefractory metastatic colorectal most cancers: an investigator-initiated, open-label, randomised, part 2 trial. Lancet Oncol. (2020) 21:412–20. doi: 10.1016/S1470-2045(19)30827-7
PubMed Summary | CrossRef Full Textual content | Google Scholar
46. Pfeiffer P, Yilmaz M, Möller S, et al. TAS-102 with or with out bevacizumab in sufferers with chemorefractory metastatic colorectal most cancers: an investigator-initiated, open-label, randomised, part 2 trial. Lancet Oncol. (2020) 21:412–20. doi: 10.1016/S1470-2045(19)30827-7
PubMed Summary | CrossRef Full Textual content | Google Scholar
47. Pfeiffer P, Winther S, Yilmaz M, Möller S, Zitnjak D, Poulsen L, et al. PD-7 Up to date survival evaluation of the Danish randomized research evaluating trifluridine/tipiracil with or with out bevacizumab in sufferers with chemo-refractory metastatic colorectal most cancers. Ann Oncol. (2020) 31:S214. doi: 10.1016/j.annonc.2020.04.463
48. Martínez-Lago N, Chucla TC, De Castro BA, Ponte RV, Rendo CR, Rodriguez MIG-R, et al. Efficacy, security and prognostic elements in sufferers with refractory metastatic colorectal most cancers handled with trifluridine/tipiracil plus bevacizumab in a real-world setting. Sci Rep. (2022) 12:14612. doi: 10.1038/S41598-022-18871-9
PubMed Summary | CrossRef Full Textual content | Google Scholar
49. Tabernero J, Taieb J, Prager GW, Ciardiello F, Fakih M, Leger C, et al. Trifluridine/tipiracil plus bevacizumab for third-line administration of metastatic colorectal most cancers: SUNLIGHT research design. Future Oncol. (2021) 17:1977–85. doi: 10.2217/FON-2020-1238
PubMed Summary | CrossRef Full Textual content | Google Scholar
51. Kotani D, Kuboki Y, Horasawa S, Kaneko A, Nakamura Y, Kawazoe A, et al. Retrospective cohort research of trifluridine/tipiracil (TAS-102) plus bevacizumab versus trifluridine/tipiracil monotherapy for metastatic colorectal most cancers. BMC Most cancers. (2019) 19:1253. doi: 10.1186/S12885-019-6475-6
PubMed Summary | CrossRef Full Textual content | Google Scholar
52. Ishizaki T, Mazaki J, Enomoto M, Shigoka M, Kasahara Ok, Matsudo T, et al. Potential multicenter part II research of biweekly TAS-102 and bevacizumab for metastatic colorectal most cancers. Anticancer Res. (2021) 41:2157–63. doi: 10.21873/ANTICANRES.14988
PubMed Summary | CrossRef Full Textual content | Google Scholar
53. Tsukihara H, Nakagawa F, Sakamoto Ok, Ishida Ok, Tanaka N, Okabe H, et al Efficacy of mixture chemotherapy utilizing a novel oral chemotherapeutic agent, TAS-102, along with bevacizumab, cetuximab, or panitumumab on human colorectal most cancers xenografts. Oncol Rep. (2015) 33:2135–42. doi: 10.3892/OR.2015.3876
PubMed Summary | CrossRef Full Textual content | Google Scholar
54. Fujii H, Matsuhashi N, Kitahora M, Takahashi T, Hirose C, Iihara H, et al. Bevacizumab together with TAS-102 improves medical outcomes in sufferers with refractory metastatic colorectal most cancers: A retrospective research. Oncologist. (2020) 25:e469–76. doi: 10.1634/THEONCOLOGIST.2019-0541
PubMed Summary | CrossRef Full Textual content | Google Scholar
55. Satake H, Kato T, Oba Ok, Kotaka M, Kagawa Y, Yasui H, et al. Section Ib/II research of biweekly TAS-102 together with bevacizumab for sufferers with metastatic colorectal most cancers refractory to plain therapies (BiTS research). Oncologist. (2020) 25:e1855–63. doi: 10.1634/THEONCOLOGIST.2020-0643
PubMed Summary | CrossRef Full Textual content | Google Scholar
56. Yoshino T, Taieb J, Kuboki Y, Pfeiffer P, Kumar A, Hochster HS. Trifluridine/tipiracil with or with out bevacizumab in metastatic colorectal most cancers: outcomes of a scientific evaluate and meta-analysis. Ther Adv Med Oncol. (2023) 15. doi: 10.1177/17588359221146137
PubMed Summary | CrossRef Full Textual content | Google Scholar
57. Nardone OM, Zammarchi I, Santacroce G, Ghosh S, Iacucci M. Irritation-driven colorectal most cancers related to colitis: from pathogenesis to altering remedy. Cancers. (2023) 15:2389. doi: 10.3390/CANCERS15082389
PubMed Summary | CrossRef Full Textual content | Google Scholar
58. Sato Y, Tsujinaka S, Miura T, Kitamura Y, Suzuki H, Shibata C. Inflammatory bowel illness and colorectal most cancers: epidemiology, etiology, surveillance, and administration. Cancers (Basel). (2023) 15(16):4154. doi: 10.3390/CANCERS15164154
PubMed Summary | CrossRef Full Textual content | Google Scholar
60. Wijnands AM, de Jong ME, Lutgens MWMD, Hoentjen F, Elias SG, Oldenburg B. Prognostic elements for superior colorectal neoplasia in inflammatory bowel illness: systematic evaluate and meta-analysis. Gastroenterology. (2021) 160:1584–98. doi: 10.1053/J.GASTRO.2020.12.036
PubMed Summary | CrossRef Full Textual content | Google Scholar
61. Qiu X, Ma J, Wang Ok, Zhang H. Chemopreventive results of 5-aminosalicylic acid on inflammatory bowel disease-associated colorectal most cancers and dysplasia: a scientific evaluate with meta-analysis. Oncotarget. (2017) 8:1031–45. doi: 10.18632/ONCOTARGET.13715
PubMed Summary | CrossRef Full Textual content | Google Scholar
62. Sheng YH, Giri R, Davies J, Schreiber V, Alabbas S, Movva R, et al. A nucleotide analog prevents colitis-associated most cancers by way of beta-catenin independently of irritation and autophagy. Cell Mol Gastroenterol Hepatol. (2021) 11:33–53. doi: 10.1016/J.JCMGH.2020.05.012
PubMed Summary | CrossRef Full Textual content | Google Scholar
63. Zhu Z, Mei Z, Guo Y, et al. Decreased threat of inflammatory bowel disease-associated colorectal neoplasia with use of thiopurines: a scientific evaluate and meta-analysis. J Crohns Colitis. (2018) 12:546–58. doi: 10.1093/ECCO-JCC/JJY006
PubMed Summary | CrossRef Full Textual content | Google Scholar