FAM83A mRNA is upregulated in LUSC tissues
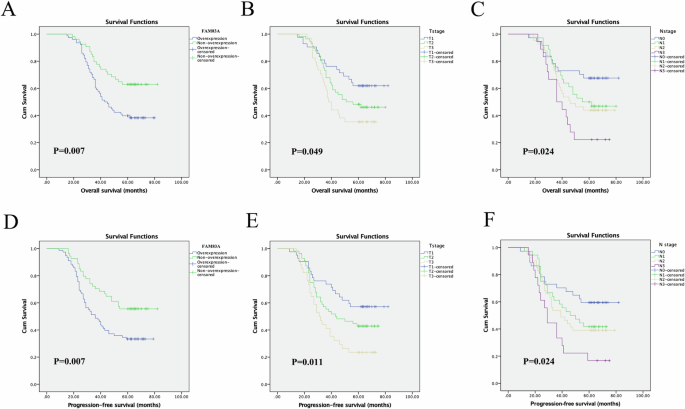
A complete of 555 samples (503 cancerous tissues and 52 regular tissues) from TCGA database have been included. We discovered that FAM83A mRNA was considerably upregulated in LUSC tissues in contrast with regular tissues (median worth: 13.464 transcripts per million vs. 0.411 transcripts per million, P < 0.001; Fig. 1A). In subgroup evaluation based mostly on histologic subtype, FAM83A mRNA was upregulated in LUSC–not in any other case specified (NOS) kind however not in basaloid, papillary, and small cell sorts. Apart from, FAM83A was upregulated in LUSC tissue of phases 1, 2, and three in contrast with regular tissues, in addition to in LUSC tissues of N0, N1, and N2. In contrast with regular controls, FAM83A mRNA was overexpressed in all affected person subgroups based mostly on age, race, gender, and smoking behavior (all P < 0.001, Fig. 1E–H), aside from the subgroup of sufferers aged 21–40 years. By looking the Oncomine database, a complete of two GEO-sourced datasets have been discovered. Meta-analysis revealed that FAM83A mRNA ranges have been considerably greater in LUSC tissues than in regular lung tissues (P = 0.032, Fig. 2A). Furthermore, as proven in Fig. 2B, C, the outcomes from the datasets have been per the findings of the abovementioned meta-analysis. To confirm the expression degree of FAM83A protein in LUSC tissues, we carried out IHC in 132 pairs of LUSC and paracancerous tissues. No vital relationship was discovered between FAM83A expression and clinicopathological options similar to age, gender, smoking, consuming, T stage, N stage, and differentiation (Desk 1). FAM83A was overexpressed in 78 (59.1%) cancerous tissues and 19 (14.4%) paracancerous tissues (P < 0.001).
A Meta-analysis of the 2 datasets on FAM83A mRNA ranges in LUSC tissue vs. regular lung tissue within the Oncomine database. B, C FAM83A mRNA expression degree within the Garber Lung (GSE quantity: GSE3398) and Hou Lung (GSE quantity: GSE19188) datasets. D General survival (OS) curves of LUSC sufferers based mostly on the differential expression ranges of FAM83A mRNA (low vs. excessive) within the TCGA database. E 5-year and F 10-year OS curves of FAM83A mRNA prognostic curve based mostly on the Kaplan–Meier database.
The prognostic worth of FAM83A in LUSC
Primarily based on the TCGA database, excessive degree of FAM83A mRNA was considerably related to poorer OS of LUSC sufferers (P = 0.00048, Fig. 2D). We additional browsed the Kaplan–Meier database, displaying that greater FAM83A mRNA expression degree additionally predicted poorer 5- and 10-year OS (each P < 0.001) (Fig. 2E, F). Subsequent, we analyzed the prognostic worth of FAM83A protein degree utilizing our personal knowledge. Of the 132 sufferers who offered FFPE most cancers tissues, 64 (48.5%) survived greater than 5 years after pneumonectomy, and 68 (51.5%) died through the follow-up interval. The imply OS time for all the sufferers was 50.5 ± 17.7 months (vary, 15–82 months), and the imply PFS time was 45.5 ± 20.6 months (vary, 9–82 months). Kaplan–Meier analyses adopted by the log-rank take a look at have been carried out to calculate the impact of the clinicopathological components on the OS and PFS charges. Excessive FAM83A protein expression was considerably related to decreased 5-year OS (P = 0.007) and PFS (P = 0.007) (Fig. 3, Desk 2). Moreover, through multivariate evaluation, FAM83A expression degree was proven to be an unbiased prognostic issue for OS (hazard ratio [HR], 2.160; 95% confidence interval [CI], 1.374–3.396, P = 0.006), in addition to PFS (HR, 2.266; 95% CI, 1.367–3.756, P = 0.002; Desk 2). Apart from, T and N phases have been vital prognostic indicators for PFS (P = 0.008 and P = 0.033, respectively).
FAM83A promotes LUSC proliferation
We additional studied the impact of FAM83A on the proliferation of LUSC cells. Lentivirus transfection technique was used for FAM83A upregulation, and plasmid was used for FAM83A downregulation. CCK8 take a look at confirmed that after upregulation of FAM83A, the cloning formation capability of H1703 and H520 cell traces considerably elevated, whereas it was considerably decreased within the siFAM83A group (Fig. 4A, B, each P < 0.001). The identical development was noticed in CCK-8 proliferation experiments (Fig. 4C).
A–C Colony formation and CCK-8 experiments have been performed in FAM83A-overexpression and -knockout teams. FAM83A overexpression elevated the colony formation and cell proliferation capability, whereas FAM83A knockdown inhibited its colony formation and cell proliferation capability. D The ferroptosis options proven by the electron microscopy after FAM83A knockdown. The mitochondrial quantity decreased, the double-membrane density elevated, and the mitochondrial ridge decreased or disappeared within the siFAM83A group. E Western blot revealed that FAM83A downregulation inhibited glutathione peroxidase 4 (GPX4) expression, whereas GPX4 upregulated in FAM83A overexpression group. F FAM83A overexpression inhibited ferroptosis, together with reducing Fe2+, ROS, and MDA content material and rising GSH degree, whereas the siFAM83A group induced the other outcomes. G, H Colony formation and I CCK8 experiments revealed that FAM83A overexpression elevated LUSC proliferation, whereas FIN56 counteracted its operate.
We additional explored the regulatory mechanism. Apparently, electron microscopy revealed that the mitochondrial quantity decreased, the double-membrane density elevated, and the mitochondrial ridge decreased or disappeared within the siFAM83A group (Fig. 4D), that are typical options of ferroptosis [15]. To additional decide the presence of ferroptosis in LUSC cells, we examined the expression of glutathione peroxidase 4 (GPX4), which is the important protein for ferroptosis. We discovered that FAM83A downregulation inhibited GPX4 expression (Fig. 4E). Furthermore, we analyzed the intracellular Fe2+ content material, ROS, malondialdehyde (MDA), a lipid peroxide product, and glutathione (GSH), an necessary substrate of GPX4. We discovered that FAM83A overexpression may inhibit ferroptosis, together with reducing Fe2+, ROS, and MDA content material and rising GSH degree, whereas the siFAM83A group confirmed the other outcomes (Fig. 4F). FIN56, a ferroptosis inducer, was used to validate the function of FAM83A in ferroptosis. Colony formation and CCK8 experiments revealed that FAM83A overexpression elevated the proliferation capability of LUSC, whereas FIN56 counteracted the operate of FAM83A (Fig. 4G–I).
FAM83A inhibits ferroptosis through the Wnt/β-catenin pathway
It has been reported that the Wnt/β-catenin signaling pathway performs an necessary function in ferroptosis. The β-catenin/TCF4 transcription advanced instantly binds to the promoter area of GPX4 and induces its expression, ensuing within the suppression of ferroptosis [16]. After regulation of FAM83A, we detected the protein ranges of the Wnt/β-catenin pathway. We discovered that FAM83A knockdown may enhance the expression degree of GSK-3β, and decreased the expression degree of p-GSK-3β (Fig. 5A), indicating that FAM83A may induce the activation of the Wnt/β-catenin pathway through enhance the phosphorylation of GSK-3β. GSK3β phosphorylates β-catenin and leads to the degradation of β-catenin, which inhibits the exercise of the Wnt/β-catenin signaling pathway [17]. We arose the speculation that FAM83A may inhibit GSK3β operate, which decreases the expression degree of p-β-catenin and elevated β-catenin expression. In consequence, upregulated β-catenin may transport into the nucleus and promote the transcription of GPX4. To additional confirm the function of the Wnt/β-catenin pathway in FAM83A-mediated ferroptosis in LUSC, we used XAV939 to inhibit the transcription operate of Wnt/β-catenin pathway. FAM83A overexpression elevated the protein expression ranges of β-catenin and GPX4, in addition to the power of cell development and clonogenicity, whereas the rescue experiment demonstrated that inhibition of the Wnt/β-catenin pathway counteracted the operate of FAM83A (Fig. 5B–E). As for ferroptosis-associated GSH, MDA, ROS, and iron content material, we additionally discovered the identical tendency (Fig. 5F).
A FAM83A knockdown elevated the expression ranges of GSK-3β and p-β-catenin, and decreased the expression ranges of p-GSK-3β and β-catenin. B FAM83A overexpression elevated the protein expression ranges of β-catenin and GPX4, in addition to the power of cell development (C) and clonogenicity (D, E), whereas inhibition of the Wnt/β-catenin (XAV-939) counteracted its operate. F FAM83A inhibited ferroptosis (the decreased degree of iron content material, MDA, ROS, and elevated GSH), whereas XAV-939 counteracted its impact.
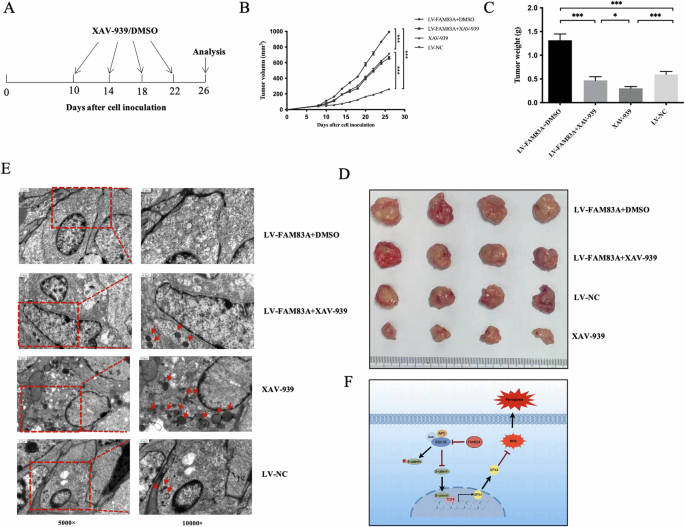
Apart from, in vivo experiments validated that FAM83A overexpression may inhibit ferroptosis and promote the tumor development. On the similar time, XAV-939 elevated ferroptosis and inhibited tumor development. As well as, the inhibition of ferroptosis by FAM83A overexpression may very well be rescued by wnt/β-catenin pathway inhibitor, XAV-939 (Fig. 6A–E). Taken collectively, FAM83A may promote LUSC cell development by inhibiting ferroptosis through activating the Wnt/β-catenin signaling pathway.
A Schedule of in vivo administration method. B Tumor development curves of tumor-bearing mice following totally different therapies. C Tumor weight in several teams on the research endpoint. D Tumor pictures of the sacrificed mice on the research endpoint. E The variation development of ferroptosis noticed through electron microscope evaluation in 4 teams. F Schematic diagram of the organic function of FAM83A in ferroptosis regulation through the wnt/β-catenin pathway in lung squamous cell most cancers.