PLCγ2 is very expressed and activated in B-ALL cells
Ca2+ signaling in B cells is regulated by PLC amongst which PLCγ2 performs a key position in B lymphocyte improvement10,19. We first explored a dataset that’s a part of the Microarray Improvements In LEukemia (MILE) research program35, which accommodates 1933 sufferers with acute and power leukemia, and analyzed overrepresented pathways utilizing R2 database. Kyoto Encyclopedia of Genes and Genomes (KEGG) evaluation revealed Ca2+ signaling because the third most enriched pathway on this cohort35 (Supplementary Fig. 1a). Evaluating pathways overrepresented between leukemia (n = 1933) and non-leukemia/wholesome (n = 71) affected person samples, Ca2+ signaling appeared as one in all 13 pathways strongly enriched in leukemia (Supplementary Fig. 1b). When proscribing the evaluation to B-ALL solely (n = 80) versus non-leukemia affected person samples (n = 71), the Ca2+ signaling pathway was constantly upregulated in B-ALL (Fig. 1a). Of all six PLC relations, PLCβ2, PLCβ3, and PLCγ2 play essential roles in B cell signaling10,18. As a result of the operate of those three PLCs in B-ALL just isn’t clearly outlined, we first in contrast their mRNA ranges in a panel of 12 B-ALL cell strains36. As proven in Supplementary Fig. 1c, PLCγ2 mRNA stage was highest in all cell strains in comparison with the opposite two PLCs (PLCβ2, PLCβ3). We then in contrast the expression of PLCγ2 gene in human B-ALL to that in different most cancers sorts36. mRNA ranges of PLCγ2 have been extra extremely expressed in B-ALL than in most different human cancers (ranked 4 of 40) (Fig. 1b and Supplementary Fig. 1d). Apparently, PLCγ2 was detected primarily in B-ALL and weakly in non-leukemia affected person samples (MILE dataset35, Fig. 1c). Comparable outcomes have been obtained when evaluating PLCγ2 expression of B-ALL to regular peripheral blood mononuclear cell (PBMC) samples by in silico evaluation of public information37 utilizing the Oncomine database38 (Fig. 1d and Supplementary Fig. 1e), or DLBCL in comparison with regular samples (GEPIA information39, Supplementary Fig. 1f). These information recommend that the PLC pathway, and specifically PLCγ2, may play a task in B-ALL. As proven in Supplementary Fig. 1g, PLCγ2 hydrolyzes phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2 or PIP2) to generate two second messengers, IP3 and DAG, ensuing within the launch of Ca2+ from inner shops and PKC activation, respectively40. PKCβ is concerned in BCR signaling and B cell survival41. Constantly, evaluation of the identical public information37 confirmed elevated mRNA ranges of PLCγ2 targets comparable to ITPR3 and PRKCβ in B-ALL in comparison with regular PBMCs (Supplementary Fig. 1h–j). To additional set up a direct connection between B-ALL and PLCγ2 signaling and activation, we analyzed the entire stage of PLCγ2 and its basal stage of phosphorylation. PLCγ2 is phosphorylated on Tyr759 for enzyme exercise. A basal phosphorylation of PLCγ2 was present in B-ALL cells (Fig. 1e), which was considerably greater in B-ALL cell strains in comparison with regular B cells (Fig. 1f). We additionally discovered the next expression of complete PLCγ2 (Fig. 1g) within the two B-ALL cell strains than in regular B cells. These information recommend that in B-ALL, PLC-γ2 may very well be aberrantly activated. To validate these outcomes and assess the potential performance of PLCγ2 in B-ALL, we carried out Ca2+ signaling experiments within the absence of extracellular Ca2+, so as to mobilize solely the ER Ca2+ shops by way of the IP3 receptor (IP3R), which is a trademark of PLCγ2 activation. Nalm-6 B-ALL cells have been first handled with completely different PLC inhibitors, i.e., U73122, edelfosine, 3-Nitrocoumarin, SPK-601, D609, and manoalide, which all considerably decreased PLCγ2 constitutive exercise, even at low focus (Supplementary Fig. 2a, b), with out affecting PLCγ2 protein ranges (Supplementary Fig. 2c). IP3R-mediated ER Ca2+ launch following carbachol (Cch) (Fig. 1h, j and Supplementary Fig. 2nd) or ATP stimulation (Fig. 1i, ok and Supplementary Fig. 2e) was additionally considerably lowered in presence of PLC inhibitors, suggesting that in B-ALL, PLC-γ2 is constitutively energetic and practical.
a Venn diagram of KEGG pathway evaluation displaying enriched pathways in non-leukemia and B-ALL samples from the R2 database within the Microarray Improvements in LEukemia (MILE) dataset (n = 26 enriched pathways in non-leukemia and n = 15 in B-ALL). Examples of enriched pathways are indicated for every subset. Two-tailed t-test, p-value < 0.05 for all pathways. b PLCγ2 mRNA ranked by expression stage in B-ALL and different human tumors. Information have been extracted from the most cancers cell line encyclopedia (CCLE). Numbers seek advice from most cancers cell sorts (see Supplementary Fig. 1d for itemizing) amongst which hematologic malignancies are proven in pink. Boxplots present the imply, median, and seventy fifth to twenty fifth percentiles. c YY-graph of PLCγ2 mRNA expression ranges in non-leukemia and B-ALL samples with expression ranges ordered from left (low) to proper (excessive) from (a). One-way evaluation of variance (ANOVA) take a look at. d PLCγ2 mRNA expression graphed as imply with SD between PBMCs and B-ALL samples within the Haferlach Leukemia dataset. Information have been obtained from the Oncomine portal and proven as boxplots representing the twenty fifth to seventy fifth percentile vary, imply, and median. Two-sided t-test. e An instance of Nalm-6 cells staining with phospho-PLCγ2 antibody and isotype management. f Stream cytometry plots (left) and MFI (proper) of the phosphorylated energetic type of PLCγ2 (phospho-PLCγ2 (Tyr759)). Information are imply ± SEM (n = 3 impartial experiments). Two-tailed, unpaired Scholar’s t-test. g Stream cytometry plots (left) and imply fluorescence depth (MFI, proper) of complete PLCγ2. Information are imply ± SEM (n = 3 impartial experiments). Two-tailed, unpaired Scholar’s t-test. h, i Endoplasmic reticulum (ER) Ca2+ launch in Nalm-6 and RS4;11 cell strains stimulated with 1 mM carbachol, Cch (h) or 0.5 mM adenosine triphosphate, ATP (i). Cells have been preincubated with the indicated PLC inhibitors throughout Fura-2 loading earlier than stimulation. j, ok Quantification of maximal ER Ca2+ launch from (h, i), respectively. Information are imply ± SEM (n = 3 impartial experiments). Precise P-values from two-way ANOVA with Sidak’s a number of comparisons take a look at have been as follows: j Nalm-6: Ctr vs. U73122, p = 0.0001; Ctr vs. Edelfosine, p < 0.0001; Ctr vs. 3-Nitrocoumarin, p = 0.0002; Ctr vs. SPK-601, p = 0.0013; Ctr vs. D609, p < 0.0001; Ctr vs. Manoalide, p < 0.0001. j RS4;11: Ctr vs. U73122, p = 0.0296; Ctr vs. Edelfosine, p < 0.0186; Ctr vs. 3-Nitrocoumarin, p = 0.0013; Ctr vs. SPK-601, p = 0.0114; Ctr vs. D609, p = 0.0203; Ctr vs. Manoalide, p = 0.0484. ok Nalm-6: Ctr vs. U73122, p = 0.0109; Ctr vs. Edelfosine, p = 0.0004; Ctr vs. 3-Nitrocoumarin, p = <0.0001; Ctr vs. SPK-601, p = 0.0017; Ctr vs. D609, p = 0.0003; Ctr vs. Manoalide, p < 0.0001. ok RS4 ;11: Ctr vs. U73122, p = 0.0012; Ctr vs. Edelfosine, p < 0.0001; Ctr vs. 3-Nitrocoumarin, p = 0.0003; Ctr vs. SPK-601, p = 0.0002; Ctr vs. D609, p = 0.0002; Ctr vs. Manoalide, p = 0.0093. Supply information are offered as a Supply Information file.
PLCγ2 inhibitors lower viability in B-ALL cells and Dexamethasone-resistant B-ALL cells
To analyze whether or not the robust expression and activation of PLCγ2 noticed in B-ALL impacts cell survival and development, cell demise was evaluated in response to the inhibition of this basal exercise. As proven in Fig. 2a, b, PLCγ2 inhibitors have been capable of considerably lower cell viability in RS4;11 B-ALL cells, and Nalm-6 B-ALL cells, respectively. An inhibitory impact was additionally noticed in Reh B-ALL cells handled with U73122 (Supplementary Fig. 3a). U73122 confirmed anti-viability, even at low focus, as measured by LIVE/DEAD® staining utilizing circulation cytometry in Nalm-6 B-ALL cells (Supplementary Fig. 3b). We then investigated which PLC downstream pathway affected cell viability. PLC controls the activation of a number of downstream effectors, comparable to PKC activation and IP3R-mediated Ca2+ launch from ER, resulting in the activation of calcineurin/NFAT signaling25. As proven in Fig. 2c and Supplementary Fig. 3c, PKC inhibitors triggered cell demise in Nalm-6 and RS4;11 cells. An inhibitory impact was additionally noticed in each cell strains within the presence of 2-APB, an IP3R-mediated Ca2+ launch inhibitor42 (Fig. 2nd) or calcineurin inhibitors (Fig. 2e and Supplementary Fig. 3d). To find out whether or not this induction of cell demise was partially brought on by a rise in apoptotic cell demise, caspase-3 exercise and annexin V / iodide propidium have been measured in response to PLCγ2 or PKC inhibitor therapy. Constantly, a rise in caspase-3 exercise and important apoptotic cell demise have been detected upon PLCγ2 or PKC inhibitor therapy in Nalm-6 and RS4;11 cells (Fig. 2f, g and Supplementary Fig. 3e–g).
a, b Cell viability relative to the Ctr of RS4;11 (a) and Nalm-6 cells (b) after 48 h publicity to growing concentrations of various PLC inhibitors (n = 3 impartial experiments). c Viability normalized to the Ctr of Nalm-6 cells after 48 h publicity to growing concentrations of various PKC inhibitors (n = 3 impartial experiments). d Viability relative to the Ctr of Nalm-6 cells and RS4;11 cells after 48 h publicity to growing concentrations of 1,4,5-trisphosphate (IP3)-induced Ca2+ launch inhibitor (n = 4 impartial experiments). e Viability normalized to the Ctr of RS4;11 cells after 48 h publicity to growing concentrations of calcineurin inhibitors (n = 4 impartial experiments). f Frequency of annexin V/PI-positive Nalm-6 cells decided by circulation cytometry after 24 h of publicity to PLC inhibitors (n = 4 impartial experiments). g Caspase-3 exercise in Nalm-6 cells decided by utilizing Ac-DEVD-AFC as substrate after 24 h of publicity to PLC and PKC inhibitors. Information are imply ± SEM n = 2 impartial experiments, carried out in 7 technical replicates. h Viability relative to the Ctr of Dex-resistant Nalm-6 cells (known as Nalm-6 R) and Dex-sensitive Nalm-6 cells (known as Nalm-6 S), uncovered for 48 h with growing concentrations of Dex (n = 4 impartial experiments). i Viability relative to the Ctr of Nalm-6 R uncovered for 48 h to growing concentrations of PLC inhibitors (n = 3 impartial experiments). j, ok RNA-seq evaluation of PLCγ2 expression in glucocorticoid-sensitive (S) or -resistant (R) main B-ALL. Information are introduced as imply ± SD and have been extracted from publicly obtainable transcriptomic dataset44,45. All information have been analyzed utilizing an ordinary Scholar’s t-test (two-sided). Until in any other case indicated, information are introduced as imply ± SEM. Supply information are offered as a Supply Information file.
With the intention to examine the potential position of PLCγ2 in B-ALL resistance to GC, we generated a GC-resistant cell line by in vitro long-term publicity to dexamethasone (Dex). For this, Nalm-6 cells have been cultured with an growing focus of Dex or an equal quantity of DMSO (Dex solvent) for 44 days (Supplementary Fig. 4a). Then, cells have been cultured in regular development medium with out Dex or DMSO for 14 days earlier than any experiments. To find out Dex sensitivity, cells have been seeded in 96-well plates for twenty-four, 48, and 72 h within the presence of various concentrations of Dex (from 0 to 100 µM) and viability assay was carried out. Dex-sensitive cells cultured within the presence of Dex grew to become extremely proof against Dex (Nalm-6 R), whereas DMSO-treated cells have been nonetheless delicate (Nalm-6 S) (Fig. 2h and Supplementary Fig. 4b, c). Nalm-6 R cells have been then handled with PLCγ2 inhibitors, i.e., U73122, edelfosine, 3-Nitrocoumarin, and manoalide, which all considerably lower cell viability (Fig. 2i). An identical impact was noticed within the presence of the PKC inhibitor GF109203X (Supplementary Fig. 4d). Collectively, these outcomes point out a task of PLC signaling in sustaining GC-resistant B-ALL cell survival.
GC resistance is related to greater PLCγ2 activation in B-ALL cells
Subsequent, we investigated whether or not Dex resistance in Nalm-6 R cells was probably mediated by lowered expression of GR. Surprisingly, we discovered the next expression of GR in resistant cells than in delicate cells (Supplementary Fig. 4e). These information exhibit that GR expression just isn’t the one Dex sensitivity issue, and their relationship just isn’t all the time correlated, in accordance with earlier studies43. Since Nalm-6 R cells are delicate to PLCγ2 inhibitors (Fig. 2i), we studied the activation of PLCγ2 utilizing phospho circulation cytometry assay. Apparently, there was a robust PLCγ2 phosphorylation in resistant in comparison with delicate cells (Supplementary Fig. 4f). PLCγ2 (Fig. 2j, ok and Supplementary Fig. 4g, h) was not differentially expressed in delicate and resistant B-ALL cells in publicly obtainable transcriptomic dataset36,43,44,45. Additionally, evaluation of paired information from publicly obtainable datasets46,47 confirmed no important change in PLCγ2 expression between prognosis and relapse in B-ALL (Supplementary Fig. 3h). With the intention to verify the hyperactivation of PLC in resistant cells, we carried out a direct fluorescence-based assay to detect PLC enzymatic exercise. For this, we used Nalm-6 cells and Reh as a mannequin for prednisone good responder (PGR) and for prednisone-poor responder sufferers, respectively, as classically described48. As anticipated, PLC enzyme exercise was considerably greater in Reh cells than in Nalm-6 cells (Supplementary Fig. 4i, j). As well as, publicly obtainable relapse information from B-ALL affected person samples46 revealed a signature of differentially expressed genes from prognosis to relapse that’s completely different between early (<36 months) and late (≥36 months) relapse. Primarily based on this criterion, our unsupervised evaluation46 confirmed that PLCγ2 was considerably greater in early relapsed sufferers (≤34 months) in comparison with late relapsed (≥35 months) sufferers (Supplementary Fig. 4k). Collectively, these information recommend that Dex-resistant cells show a reliance on constitutively energetic PLC signaling.
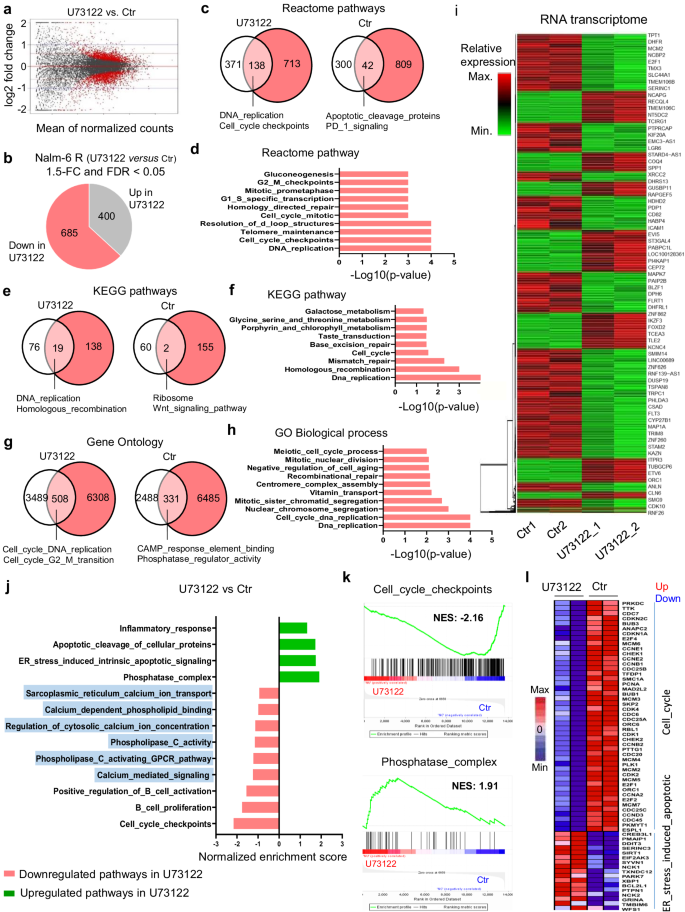
PLC inhibition dysregulates the transcriptional applications of cell cycle in Dex-resistant B-ALL cells
To outline the operate of PLC in GC-resistant B-ALL cells on the molecular stage, we subsequent carried out transcriptome evaluation in Ctr and U73122-treated-Nalm-6 R cells utilizing RNA sequencing (RNA-seq). Of the 13,654 genes (Fig. 3a), 1085 genes have been considerably differentially regulated in U73122-treated cells (±1.5-fold change in expression, adjusted p-value < 0.05), with 685 downregulated and 400 upregulated (Fig. 3b). Unbiased pathway enrichment analyses of differentially expressed genes (DEG) utilizing Reactome, KEGG and Gene ontology (GO) evaluation revealed the dysregulation of a number of signaling pathways in U73122-treated cells. Notably, 138 gene units have been considerably (p < 5%) under-enriched in Reactome evaluation (Fig. 3c), 19 gene units in KEGG evaluation (Fig. 3e) and 508 gene units in GO evaluation (Fig. 3g), suggesting that PLC controls the transcriptional program of Nalm-6 R cells in numerous methods. Of those, the cell cycle-associated gene pathway was most importantly and constantly downregulated (Fig. 3c–h). Globally, transcriptional profiles of Ctr and U73122-treated-cell subsets have been clearly distinct (Fig. 3i). Gene set enrichment evaluation (GSEA) revealed upregulation of ER stress-induced intrinsic apoptotic signaling pathway-, phosphatase complex-associated genes; downregulation of B cell proliferation- and activation-associated pathways, comparable to cell cycle, Ca2+ mediate signaling, regulation of cytosolic Ca2+ ion focus; and downregulation of PLC signaling-related pathways, comparable to PLC activating G-protein-coupled receptor pathway, PLC exercise and ER Ca2+ ion transport within the U73122-treated cells in comparison with the Ctr cells (Fig. 3j, ok). The DEG within the transcription profiles additionally had coordinately altered cell viability, particularly cell cycle-associated genes, comparable to CDC6, CDK1, E2F1 and ER stress-associated genes, comparable to XBP1, CHOP (Ddit3), PMAIP1, SIRT1, and SERINC3 (Fig. 3l). With the intention to perceive the underlying regulatory mechanisms of this elementary course of, we additionally investigated how PLC inhibition disturbs the cell cycle course of in Dex-sensitive B-ALL cells. Cells have been handled with completely different PLC inhibitors, that every one promoted cell cycle arrest on the subG1 section whereas negatively regulated the G2/M section (Supplementary Fig. 5 and Supplementary Fig. 6a–f), in accordance with our GSEA evaluation displaying robust downregulation of genes concerned in sister chromatid segregation mitotic on the G1/S transition and E2F-mediated G2/M section cell cycle gene expression (Supplementary Fig. 6g). Collectively, these findings exhibit that PLC promotes survival signaling in B-ALL cells by regulating cell cycle development and division and past that, a fancy program of gene expression.
a MA plot displaying the connection between log2-fold modifications of RNA reads in U73122 versus Ctr Nalm-6 R cells plotted towards imply expression values. Crimson dots characterize genes with an adjusted p-value < 0.05 utilizing DESeq2’s differential expression take a look at. b Differentially expressed genes (DEG) in Nalm-6 R cells after 16-h publicity to 1 µM U73122 decided by RNA-seq evaluation (false discovery price [FDR] adjusted p < 0.05, and fold change (log2 scale) ≥1.5 or ≤ −1.5) utilizing DESeq2 take a look at. c, e, g Abstract of Reactome (c), KEGG (e), and GO (g) pathway evaluation of Ctr and U73122-treated cells. Venn diagram reveals gene units which might be downregulated, non-significant (white), important with p-value < 0.05 (pink), and unregulated (pink) as decided by gene set enrichment evaluation (GSEA) Kolmogorov–Smirnov take a look at. Examples of distinctive pathways are indicated for every group. d, f, h Useful clustering of downregulated genes in U73122-exposed-Nalm-6 R cells (n = 2 organic replicates) with FDR adjusted p-value < 0.05 for KEGG (f), <0.001 for Reactome (d), <0.03 for GO Organic course of (h) utilizing GSEA (Kolmogorov–Smirnov) take a look at. Some prime pathways are indicated. i Hierarchical clustering after RNA-seq evaluation displaying DEG calculated by DESeq2 take a look at in Nalm-6 R cells after U73122 publicity (n = 2 organic replicates). j Normalized enrichment scores of considerably up- or downregulated gene units in Nalm-6 R cells after U73122 publicity (n = 2 organic replicates). ok Consultant GSEA enrichment plot displaying upregulation of phosphatase_complex-related genes and downregulation of cell cycle checkpoint-related genes in Nalm-6 R cells after U73122 publicity (n = 2 organic replicates). l Heatmap of ER_stress_induced_apoptotic and cell cycle DEG in Nalm-6 R cells after U73122 publicity (n = 2 organic replicates). Supply information are offered as a Supply Information file.
Dex publicity triggers Ca2+ signaling via the PLCγ2 pathway in B-ALL cells
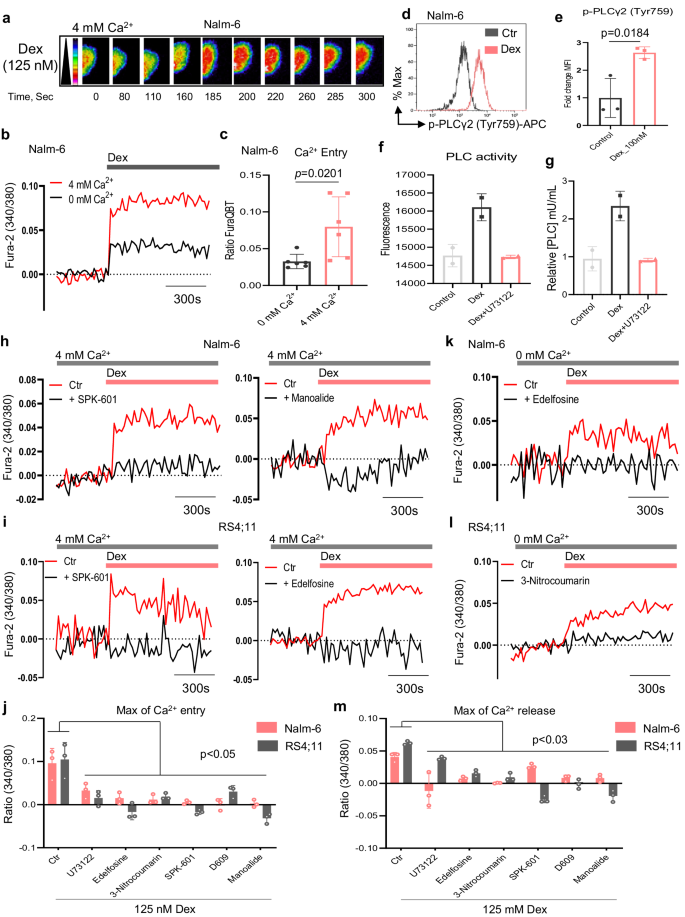
Given the significance of PLCγ2 for B cell operate10,15,16,18,19 and intracellular Ca2+ indicators for B-ALL cell resistance to Dex8, we investigated the position of PLCγ2 in Dex-mediated Ca2+ signaling in B-ALL. To optically measure cytosolic Ca2+ shifts, we used single-cell Ca2+ imaging by fluorescence microscopy following stimulation of B-ALL cells with Dex (Fig. 4a). Dex publicity resulted in elevated cytosolic Ca2+ in B-ALL cells (Fig. 4a–c and Supplementary Fig. 7a, b). Moreover, these observations have been additionally validated utilizing Fluo-4 AM probe (Supplementary Fig. 7c). In keeping with our earlier studies8, we discovered that strong and sustained cytosolic Ca2+ indicators induced by Dex have been considerably greater in 4 mM extracellular Ca2+ as in contrast with Ca2+-free buffer (0 mM extracellular Ca2+, Fig. 4b, c and Supplementary Fig. 7a, b). Since this pathway of Ca2+ entry into B-ALL cells bears the pharmacological traits of store-operated Ca2+ entry (SOCE), we measured SOCE by stimulating cells with Dex in Ca2+-free buffer, which induces activation of SOC channels following ER Ca2+ depletion, and SOCE was then evaluated by the addition of 4 mM extracellular Ca2+. Certainly, Dex considerably induced ER Ca2+ launch and SOCE (Supplementary Fig. 7d–g), suggesting that Dex in all probability regulates SOCE by way of Ca2+ launch from the ER by way of IP3 receptor channels following PLC activation. To check this speculation, we handled B-ALL cells with Dex for five min, the time required to induce a sustained Ca2+ enhance, and measured PLCγ2 activation by phospho circulation cytometry. Dex stimulation triggered a robust upregulation of phospho-PLCγ2 in B-ALL cell strains (Fig. 4d, e and Supplementary Fig. 8a, b). Furthermore, to validate the power of Dex to activate PLC in B-ALL cells, we carried out a direct fluorescence-based assay for detecting protease exercise after 24 h of Dex publicity and confirmed that Dex elevated PLC exercise in B-ALL cells, which was decreased within the presence of PLC inhibitor (Fig. 4f, g). To make sure that PLC activation by Dex was practical, we measured Dex-mediated Ca2+ within the presence of various PLC inhibitors. Dex-mediated Ca2+ signaling was prevented by PLC inhibitors, which was evident from the lower in Ca2+ launched from ER shops and the general Ca2+ entry from shops together with exterior and inner (Fig. 4h–m and Supplementary Fig. 8c–e). Most significantly, the information from the genetic silencing of PLCγ2 in addition to PLCv1 have been in line with the pharmacological inhibitor options of Dex-induced B-ALL cell demise (Supplementary Fig. 9a–d). Collectively, these outcomes exhibit that Dex-induced Ca2+ response in B-ALL cells is principally mediated by PLC.
a Coloured time-lapse photos of Nalm-6 cells present the modifications in cytosolic Ca2+ evoked by Dex in Ca2+-containing buffer. b Cytosolic Ca2+ measurements in Nalm-6 cells stimulated with 125 nM Dex in nominally Ca2+-free buffer (0 mM Ca2+) and Ca2+-containing buffer (4 mM Ca2+). c Quantification of maximal Ca2+ entry from (b). Information proven are the imply ± SEM (n = 6 impartial experiments). d Stream cytometry plots of the energetic type of PLCγ2 (phospho-PLCγ2 (Tyr759)) stimulated with Dex or Ctr for five min. e Quantification from (d) normalized to Ctr. Information are imply ± SEM (n = 3 impartial experiments). f, g Quantification of PLC exercise (f) and intracellular focus of PLC (mU/mL) (g) in Nalm-6 cells stimulated with 100 nM Dex or Ctr for twenty-four h with or with out 1 µM U73122 and normalized to Ctr in (g). Information are imply ± SEM from n = 2 impartial experiments, carried out in technical octuplicates. h, i Cytosolic Ca2+ measurements in Nalm-6 (h) and RS4;11 (i) cells stimulated with 125 nM Dex in Ca2+-containing buffer (4 mM Ca2+). Cells have been preincubated with SPK-601, and both Manoalide (h) or Edelfosine (i) throughout Fura-2 loading earlier than stimulation. All inhibitors are used at 10 µM. j Maximal cytosolic Ca2+ entry from (h, i). Information are imply ± SEM (n = 3 impartial experiments). Precise P-values from two-way ANOVA with Sidak’s a number of comparisons take a look at have been as follows: Nalm-6: Ctr vs. U73122, p = 0.0491; Ctr vs. Edelfosine, p = 0.0024; Ctr vs. 3-Nitrocoumarin, p = 0.0014; Ctr vs. SPK-601, p = 0.0005; Ctr vs. D609, p = 0.0002; Ctr vs. Manoalide, p = 0.0003. RS4;11: Ctr vs. U73122, p < 0.0001; Ctr vs. Edelfosine, p < 0.0001; Ctr vs. 3-Nitrocoumarin, p < 0.0001; Ctr vs. SPK-601, p < 0.0001; Ctr vs. D609, p = 0.0002; Ctr vs. Manoalide, p < 0.0001. ok, l Traces of ER Ca2+ launch in Nalm-6 (ok) and RS4;11 (l) cells stimulated with 125 nM Dex in Ca2+-free buffer (0 mM Ca2+). Cells have been preincubated with both 10 µM Edelfosine (ok) or 10 µM 3-Nitrocoumarin (l) throughout Fura-2 loading earlier than stimulation. m Maximal ER Ca2+ launch from (ok, l). Information are imply ± SEM (n = 3 impartial experiments). Precise P-values from two-way ANOVA with Sidak’s a number of comparisons take a look at have been as follows: Nalm-6: Ctr vs. U73122, p < 0.0001; Ctr vs. Edelfosine, p = 0.0004; Ctr vs. 3-Nitrocoumarin, p = <0.0001; Ctr vs. SPK-601, p = 0.0092; Ctr vs. D609, p = 0.0007; Ctr vs. Manoalide, p = 0.0006. RS4;11: Ctr vs. U73122, p = 0.0267; Ctr vs. Edelfosine, p < 0.0001; Ctr vs. 3-Nitrocoumarin, p < 0.0001; Ctr vs. SPK-601, p < 0.0001; Ctr vs. D609, p < 0.0001; Ctr vs. Manoalide, p < 0.0001. Information (in c, e–g) was analyzed by two-tailed, unpaired Scholar’s t-test. Supply information are offered as a Supply Information file.
PLCγ2 signaling mediates Dex resistance in B-ALL cells
Elevated cytosolic Ca2+ via PLCγ2 in each regular and pathological B cells has been proven to induce cell survival and development10,15,18. Since Dex publicity prompts PLCγ2 signaling pathway, we aimed to find out whether or not PLCγ2 inhibition may improve Dex sensitivity and reverse Dex resistance in B-ALL cells. For this, we handled B-ALL cell strains with pharmacological or genetic inactivation of PLCγ2 in addition to PLCγ1 together with Dex. Cell demise was strongly elevated by mixed therapies (Fig. 5a, b; Supplementary Figs. 9e, f 10a). The power of PLC inhibition to extend GC sensitivity was confirmed by LIVE/DEAD® (Fig. 5c, d) and annexin V/PI (Fig. 5e, f) staining. Furthermore, these observations have been validated in main blood and bone marrow (BM) samples from a cohort of 44 ALL sufferers with extremely variable in vitro Dex sensitivity (Supplementary Fig. 10b). By defining Dex sensitivity (≥20% cell demise) and Dex resistance (<20% cell demise) following Dex publicity, we discovered that 80% and 72% main diagnostic blood and BM samples have been intrinsically Dex resistant when cultured in vitro within the presence of 100 nM Dex, respectively (Supplementary Fig. 10b). Importantly, inhibition of PLC signaling with U73122 was enough to beat Dex resistance in 62% and 52% of those Dex-resistant blood and BM samples, respectively (Fig. 5g). In keeping with the lower in cell viability, the mixture of Dex and the PLC inhibitor confirmed a lower in cell proliferation, as evidenced by decreased CTV dilution (Fig. 5j, ok).
a, b Nalm-6 (a) and RS4;11 (b) cells handled with Dex, U73122, Edelfosine, 3-Nitrocoumarin, SPK-601, D609, and Manoalide alone or together. Cell mortality was decided by CCk-8 staining after 48 h of therapy. Information proven are the imply ± SEM of n = 3 (in a) and n = 3 (in b) impartial experiments. Two-tailed, unpaired Scholar’s t-test. c Nalm-6 cells handled with 100 nM Dex and 1 µM U73122 alone or together for 48 h. Alive and useless cells have been decided by LIVE/DEAD® staining Kits utilizing circulation cytometry. d Quantification of alive and useless cell populations from (c). The proportion of cells was established after normalizing cells on Ctr cells. Imply ± SEM (n = 3 impartial experiments), two-tailed, unpaired Scholar’s t-test. e Nalm-6 cells handled with 100 nM Dex and 1 µM U73122 alone or together for 48 h. Cell mortality was decided by circulation cytometry after annexin V/PI staining. f Quantification of complete apoptotic cell populations from (e). Information are imply ± SEM (n = 3 impartial experiments). Two-tailed, unpaired Scholar’s t-test. g Cell viability of Dex-resistant main diagnostic ALL blood (n = 16 sufferers) and bone marrow (n = 21 sufferers) samples uncovered to 100 nM Dex and/or 1 μM U73122 for twenty-four h, normalized to Ctr situation. Information are imply ± SD. Two-tailed, unpaired Scholar’s t-test. h, i Cell cycle evaluation of Nalm-6 cells cells after stimulation with 100 nM Dex and/or 1 μM U73122 for 72 h. Cell cycle distribution (h) and subG1 quantification (i) have been decided by PI staining adopted by FACS evaluation (n = 3 technical replicate). Two-tailed, unpaired Scholar’s t-test. j, ok Proliferation in Nalm-6 cells within the presence of 100 nM Dex and/or 1 μM U73122 for 3 days. Stream cytometry evaluation displaying CellTrace Violet (CTV) dilution (j) and proliferation quantification (ok). Information are imply ± SEM (n = 3 impartial experiments in ok). Two-tailed, unpaired Scholar’s t-test. l GSEA of RNA-Seq information displaying enrichment within the mobile response to Dex stimulus evaluating the transcriptome of Dex-resistant Nalm-6 cells handled with U73122 or Ctr (n = 2 organic replicates). See Supplementary Fig. 11c for differentially expressed gene profiles of mobile response to Dex stimulus-related genes. Supply information are offered as a Supply Information file.
To additional examine the mechanistic foundation of Dex resistance mediated by PLC signaling, we subsequent assessed the position of PLC downstream signaling pathways. PLC activation results in the manufacturing of IP3 and DAG, which subsequently and respectively set off the discharge of ER Ca2+ by way of the IP3R channel and PKC activation40. We examined the roles of those two downstream cascades, the DAG/PKC-dependent and IP3/Ca2+-dependent pathways. For this, we first used the IP3R channel inhibitor 2-APB42, which inhibits ER Ca2+ output. As anticipated, useless cells have been vastly elevated by co-treatment with 2-APB and Dex (Supplementary Fig. 10c, d). To additional set up a direct connection between Dex and PLC signaling, we analyzed PKC exercise, which is activated by PLC via DAG by utilizing an enzyme-linked immunosorbent (ELISA) assay. Dex stimulation resulted in robust upregulation of PKC exercise, which was lowered by PLC inhibitor U73122 (Supplementary Fig. 10e), suggesting that PKC is certainly activated downstream of Dex-induced PLC signaling. Furthermore, completely different PKC inhibitors considerably elevated Dex-mediated B-ALL cell demise (Supplementary Fig. 10f, g). The power of PKC inhibition to extend Dex sensitivity was confirmed by LIVE/DEAD® staining (Supplementary Fig. 10h, i). In keeping with the leads to B-ALL cell strains, the PKC inhibitor GF109203X restored Dex sensitivity in Dex-resistant main blood and BM samples (Supplementary Fig. 10j). After having established that the cell cycle was regulated by the PLC pathway, we decided whether or not this enhance in cell demise was partially brought on by cell cycle arrest. Remedy of B-ALL cells with U73122, GF106203X, and 2-APB resulted in elevated Dex-induced cell demise, as manifested by a rise in subG1 apoptotic cell inhabitants (Fig. 5h, i and Supplementary Fig. 11a, b). These outcomes strongly recommend that the PLC pathway performs a important position in inducing GC resistance, and its exercise will be focused to sensitize B-ALL cells to GC therapy.
We subsequent requested whether or not PLC inhibition may have an effect on GR transcriptional program and expression. GSEA evaluation of our RNA-seq revealed an enrichment of the GR pathway and the mobile response to Dex stimulus genes in U73122-treated cells (Fig. 5l and Supplementary Fig. 11c–e) and a rise in GR expression after therapy with manoalide (Supplementary Fig. 11f, g), additional indicating that the sensitivity of B-ALL cells to GC depends on PLC operate.
We subsequent centered our consideration on the calcineurin/NFAT signaling, one other downstream pathway managed by PLC via IP3-mediated Ca2+ signaling activation25. We investigated whether or not this pathway may have an effect on GC sensitivity in B-ALL cells. We discovered that calcineurin inhibitors (cyclosporin A and tacrolimus, FK506) additionally considerably elevated Dex sensitivity in B-ALL cell strains (Supplementary Fig. 12). To evaluate whether or not PLCγ2 inhibition may reverse Dex resistance, we used our strongly Dex-resistant Nalm-6 R cells (Fig. 2h and Supplementary Fig. 4b, c). PLC, PKC, and calcineurin inhibitors have been enough to beat Dex resistance (Supplementary Fig. 13a–g). In keeping with earlier studies45, inhibiting mTOR by sirolimus/rapamycin or Akt by MK-2206 (Supplementary Fig. 13h), two pathways interconnected with PLCγ2/Ca2+/PKC signaling and implicated in B cell improvement and most cancers10,49, additionally reversed Dex resistance. Collectively, these information present proof that GC paradoxically induce their very own resistance by activating the pro-survival signaling of the PLC/Ca2+/PKC pathway.
CXCR4 is very expressed in B-ALL cells and positively correlated with PLCy2
We hypothesized that the activation of PLCγ2 signaling by Dex was mediated by cell floor receptor signaling. A complete-genome correlation community evaluation of PLCγ2 utilizing the R2 database recognized 281 genes whose transcript expression strongly correlated with these of PLCγ2 in MILE dataset35 (Fig. 6a, b, Supplementary Fig. 14a, Pearson’s r > 0.5, p < 1.8E-125). In an try to determine genes correlated with PLC but additionally concerned in Ca2+ signaling, we in contrast these genes co-expressed with PLC to the general public Ca2+ signature genes from the Gene Ontology Organic Course of (GOBP) Gene Set (Supplementary Fig. 14b). We discovered that six of those genes have been concerned in Ca2+-mediated intracellular sign transduction (Fig. 6a, b). Excluding the PPP3CC (Protein Phosphatase 3 Catalytic Subunit Gamma) gene, the opposite 5 are all genes encoding cell floor proteins in people. Of the six recognized targets, CXCR4 mRNA was essentially the most expressed within the 12 B-ALL cell strains obtainable within the CCLE dataset36 (Fig. 6c), whereas CD22 was reasonably expressed, and the expression of SLC9A1 (Solute Service household 9A1), CCR7 (C-C Motif Chemokine Receptor 7), PPP3CC and CXCR5 (C-X-C Motif Chemokine Receptor 5) was very low or undetectable in these cell strains.
a Venn diagram depicting the overlap between genes strongly correlated (r > 0.5) with PLCγ2 within the MILE dataset versus Ca2+ mediated signaling genes in GO organic processes. b Distribution of all MILE dataset genes primarily based on their co-expression coefficient (Pearson’s r) relative to PLCγ2. Genes linked to Ca2+ mediated signaling highlighted in pink are considerably enriched among the many co-expressed genes. c Heatmap displaying expression profiles of overlapping genes from (a) in numerous B-ALL cell strains. Information extracted from the most cancers cell line encyclopedia (CCLE). d CXCR4 mRNA ranked by expression stage in B-ALL and different human tumors. Information extracted from the CCLE. Numbers seek advice from most cancers cell sorts with hematological malignancies depicted in pink (see Supplementary Fig. 15a). Boxplots present the imply, median, and seventy fifth to twenty fifth percentiles. e Averaged cytosolic Ca2+ measurements in Nalm-6 cells stimulated with 125 nM Dex with or with out AMD3465. Information are imply ± SEM from (n = 6 for Ctr and n = 3 for antagonist; n-values correspond to impartial experiments). f Quantification of maximal Ca2+ entry of Nalm-6 cells stimulated with 125 nM Dex within the absence or presence of various CXCR4 antagonists. Information are the imply ± SEM from (n = 6 for Ctr, n = 4 for WZ811, and n = 3 for AMD3465 and AMD3100; n-values correspond to impartial experiments). ****p < 0.0001. g Consultant circulation cytometry plots of CXCR4 floor expression in Nalm-6 cells transfected with small interfering RNA Ctr (siCtr) or siCXCR4. h Consultant cytosolic Ca2+ measurements in Nalm-6 cells transfected with both siCtr or siCXCR4 then stimulated with 125 nM Dex in nominally Ca2+-containing buffer (2 mM Ca2+). i Quantification of maximal Ca2+ entry from (h). Information are imply ± SEM (n = 3 impartial experiments, p = 0.0209). j Cytosolic Ca2+ measurements in Nalm-6 cells transduced with both CRISPR-Ctr or CRISPR-Cxcr4 then stimulated with 100 nM SDF-1α in nominally Ca2+-containing buffer (4 mM Ca2+). ok Quantification of maximal Ca2+ entry from (j). Information are imply ± SEM (n = 5 impartial experiments). ****p < 0.0001. l Cytosolic Ca2+ measurements in Nalm-6 cells transduced with both CRISPR-Ctr or CRISPR-Cxcr4 then stimulated with 125 nM Dex in nominally Ca2+-containing buffer (4 mM Ca2+). m Quantification of maximal Ca2+ entry from (l). Information are imply ± SEM (n = 5 impartial experiments). ****p < 0.0001. n, o Consultant circulation cytometry plots of the energetic type of PLCγ2 (phospho-PLCγ2 (Tyr759)) induced by 125 nM Dex in Nalm-6 cells transfected with both CRISPR-Ctr or CRISPR-Cxcr4 (n) and in Nalm-6 cells within the absence or presence of AMD3465 (o). p, r Consultant circulation cytometry plots of CXCR4 floor expression of Nalm-6 cells stimulated with 125 nM Dex for five min at 37 °C. q, s Quantification of CXCR4 MFI from (p, r), respectively. Information normalized to Ctr are imply ± SEM (n = 3 impartial experiments). t Nalm-6 cells have been stimulated in a tradition medium with both 100 nM Dex, 100 nM SDF-1α or medium alone. Cells have been both transferred instantly on ice (T = 0) or after incubation for 1 or 2 h at 37 °C (T = 1 or 2 h) earlier than staining with anti-human CXCR4 mAb. Values characterize the proportion of staining, 100% similar to unstimulated cells processed in parallel. Information proven are the imply ± SEM (n = 2 for Dex, n = 3 for SDF-1α impartial experiments). u Consultant circulation cytometry plots of CXCR4 floor expression of Nalm-6 cells stimulated with 125 nM Dex for twenty-four h. v Cytosolic Ca2+ measurements in Nalm-6 cells stimulated with 125 nM Dex and 100 nM SDF-1α in buffer containing 4 mM Ca2+. Information are imply ± SEM (n = 3 impartial experiments). w Quantification of maximal Ca2+ entry from (v). Information characterize the imply ± SEM (n = 7 impartial experiments, carried out in duplicates). All statistical significance was analyzed by two-tailed, unpaired Scholar’s t-test. *p < 0.05, and ****p < 0.0001. Supply information are offered as a Supply Information file.
Along with the optimistic correlation between PLCγ2 and CXCR4 noticed in MILE leukemia cohort35 (Supplementary Fig. 14c, Pearson’s r = 0.529, p = 1.11E-139), these two genes have been additionally positively correlated within the DLBCL dataset39 (Supplementary Fig. 14d, Pearson’s r = 0.51, p = 0.00027). We then prolonged our comparability of the CXCR4 gene in human B-ALL to different most cancers sorts36. mRNA ranges of CXCR4 have been extra extremely expressed in B-ALL than in most different human cancers (ranked 3 of 40) (Fig. 6d and Supplementary Fig. 15a). This expression was related to hypomethylation in human B-ALL in comparison with most different cancers (ranked 37 of 38) (Supplementary Fig. 15b, c), an epigenetic modification noticed in B-ALL50 properly described as a tumor stimulator that promotes mitotic cell division51. Evaluation of publicly obtainable information revealed that CXCR4 mRNA was extremely expressed in main samples of B-ALL and T-ALL in comparison with regular samples52 (Supplementary Fig. 15d). This enhance was additionally noticed within the GC-resistant B-ALL main samples in comparison with the delicate ones45 (Supplementary Fig. 15e). We subsequent carried out a meta-analysis utilizing R2 database survival evaluation to additional assess the position of CXCR4 in medical outcomes. The evaluation revealed that cancers with greater CXCR4 expression had considerably worse total survival (Supplementary Fig. 15f, i). Collectively, these information set up the significance of the CXCR4 pathway in B-ALL.
CXCR4 mediates Dex-induced PLCγ2/Ca2+ axis activation in B-ALL cells
We subsequent assessed whether or not the correlation between PLCγ2 and CXCR4 had practical penalties in B-ALL cell signaling and whether or not it was important for Dex sensitivity. We first checked the expression of CXCR4 on our cell fashions. CXCR4 is very expressed on the floor of B-ALL cell strains and downregulated upon CXCR4 blockade utilizing small molecule antagonists (Supplementary Fig. 16a–d). PLCγ2 has beforehand been proven to play a task in CXCR4-mediated B cell migration16. CXCR4 antagonist alone had no impact on Ca2+ signaling (Supplementary Fig. 16e). The pure ligand of CXCR4 (SDF-1α) publicity resulted in elevated cytosolic Ca2+ in B-ALL cells, which was abrogated within the presence of CXCR4 antagonists (Supplementary Fig. 16f–m). These outcomes point out that CXCR4 is functionally energetic in B-ALL cells. Subsequent, we investigated the position of CXCR4 within the regulation of Dex-induced intracellular Ca2+ indicators. Apparently, we discovered that the strong and sustained cytosolic Ca2+ indicators induced by Dex have been considerably curtailed within the presence of CXCR4 antagonists (Fig. 6e, f and Supplementary Fig. 16n–s). To validate the position of CXCR4 in Ca2+ signaling of Dex, in a primary method, we negatively regulated the expression of CXCR4 utilizing small interfering RNA (siRNA) in Nalm-6 cells (Fig. 6g–i). Correlatively, Dex-induced Ca2+ signaling upon Dex publicity was decreased in Nalm-6 transfected with siRNA CXCR4 (siCXCR4) as in comparison with siCtr (Fig. 6h, i). As a second method, we mutated endogenous CXCR4 in two B-ALL cell strains cells utilizing CRISPR/Cas9 with Inexperienced fluorescent protein (GFP), a marker of cell transfection with the CRISPR-Cas9 plasmid concentrating on CXCR4 (CRISPR-Cxcr4) (Supplementary Fig. 16t). B-ALL cells have been then sorted for GFP positivity. Stream cytometry assays indicated a considerable lower in CXCR4 floor expression in CRISPR-Cxcr4 cells in contrast with CRISPR-Ctr cells (Supplementary Fig. 16u, v). Importantly, Ca2+ signaling induced by SDF-1 was utterly suppressed within the CRISPR-Cxcr4 cells, indicating the non-functionality of the CXCR4 receptor within the transfected cells (Fig. 6j, ok and Supplementary Fig. 16w, x). Subsequently, we measured Dex-induced Ca2+ signaling in these transfected cells and located that Dex didn’t induce will increase within the cytosolic Ca2+ in CRISPR-Cxcr4 cells, demonstrating that CXCR4 controls Dex-induced Ca2+ signaling in B-ALL cells (Fig. 6l, m and Supplementary Fig. 16y, z). Since CXCR4 is coupled to G-protein-mediated signaling pathway, which regulates Ca2+ signaling primarily by way of IP3 manufacturing and Ca2+ launch from the ER by way of IP3R channels, we evaluated this by stimulating cells with Dex in Ca2+-free buffer within the presence of CXCR4 antagonists. We noticed decreased ER Ca2+ launch in CXCR4 antagonist-treated cells in comparison with Ctr in response to Dex stimulation (Supplementary Fig. 17a–d). We additionally measured SOCE as described above. As anticipated, Dex-mediated SOCE was considerably decreased within the presence of CXCR4 antagonists (Supplementary Fig. 17e–h). Since SOCE and IP3-mediated ER Ca2+ launch are triggered by PLC activation, we handled B-ALL cells with Dex for five min, the time required to induce a sustained Ca2+ enhance, and measured PLCγ2 activation by phospho circulation cytometry within the presence of CXCR4 antagonists, CRISPR-Cxcr4 or siCXCR4. Dex triggered a robust upregulation of phospho-PLCγ2 in B-ALL cell strains, which was impaired in CRISPR-Cxcr4 and siCXCR4 cells in addition to after publicity to CXCR4 antagonists (Fig. 6n, o and Supplementary Fig. 17i–l). Complete PLCγ2 expression just isn’t affected by CXCR4 antagonists or CRISPR-Cxcr4 (Supplementary Fig. 17m–p). Collectively, these outcomes exhibit that Dex-induced Ca2+ response in B-ALL cells is principally mediated by PLCγ2 via CXCR4.
As a result of earlier research established that the CXCR4 receptor is quickly internalized upon activation53,54, we decided whether or not Dex-induced CXCR4 internalization in B-ALL cells. As a primary method, we handled B-ALL cells with Dex for five min and measured CXCR4 floor expression by circulation cytometry. As proven in Fig. 6p–s and Supplementary Fig. 17q, r, Dex stimulation triggered a robust downregulation of CXCR4 floor expression in 4 B-ALL cell strains, i.e., (Nalm-6, Reh, RS4;11 and HAL-01), constantly with earlier studies demonstrating that CXCR4 internalization happens 2–5 min after activation53,55. The downregulation of CXCR4 induced by Dex could also be due partially to its internalization moderately than its inhibition as a result of Dex induces Ca2+ signaling, which is blocked by the inhibition of CXCR4. As well as, primarily based on the concept chilly impedes the internalization of membrane receptors54, we stimulated B-ALL cells with SDF-1α or Dex and instantly transferred them on ice (at 4 °C, T = 0) or incubated at 37 °C for 1 or 2 h (T = 1, 2 h). As proven in Fig. 6t, CXCR4 cell floor expression on B-ALL cells was downregulated after 1 h or 2 h of publicity to Dex or SDF-1α at 37 °C. As proven in Supplementary Fig. 17s–u, and Fig. 6u, Dex-mediated downregulation of CXCR4 expression was additionally noticed over time (as much as 20–24 h after stimulation), although the impact was lowered, suggesting that the receptor recycles poorly after stimulation, as demonstrated for SDF-156. We subsequent investigated whether or not Dex-mediated CXCR4 internalization was a driver or an end result of its activation, by figuring out if Dex may induce Ca2+ signaling when CXCR4 internalization was prevented (i.e., at +4 °C moderately than +37 °C). We discovered that within the absence of CXCR4 internalization, Ca2+ signaling was certainly triggered by Dex, confirming that internalization of CXCR4 was only a secondary impact of its activation (Supplementary Fig. 18a–e). Moreover, the GC receptor antagonist (RU486) had no impact on Dex-mediated Ca2+ signaling (Supplementary Fig. 18f). In a second method, we investigated whether or not Dex may intrude with the power of SDF-1 to stimulate Ca2+ signaling in B-ALL cells. Apparently, when SDF-1 was added after Dex, i.e., on the time of most Ca2+ peak (Fig. 6v, w and Supplementary Fig. 19a–d), no additive impact on Ca2+ entry was noticed, suggesting that Dex is an efficient competitor of SDF-1 for CXCR4 activation of Ca2+ signaling. In any other case, when Dex was added after SDF-1 (Supplementary Fig. 19e, f), a slight however non-significant additive impact on Ca2+ entry was noticed. These outcomes recommend that the unresponsiveness of SDF-1 will be primarily attributed to CXCR4 receptor internalization by Dex. To substantiate this, we used one other molecule thapsigargin (TG), able to stimulating Ca2+ (Supplementary Fig. 19g, h) by blocking sarcoplasmic-endoplasmic Ca2+-ATPase, due to this fact a distinct pathway to CXCR4, as proven in Supplementary Fig. 19i, j, the place the CXCR4 antagonist AMD3100 had no impact on ER Ca2+ launch and SOCE induced by TG. As suspected, when TG was added after Dex (Supplementary Fig. 19k–r), or when Dex was added after TG (Supplementary Fig. 19s–v), the impact on Ca2+ entry was additive, suggesting that Dex acted to mobilize Ca2+ by way of a CXCR4-specific and dependent mechanism. Since CXCR4 is localized in lipid rafts57 and localization in lipid rafts can impression Ca2+ signaling by influencing G-protein and PLC activation, we disrupted lipid rafts by methyl-β-cyclodextrin (M-βCD) and confirmed a lower in Dex-induced Ca2+ signaling in B-ALL cell strains and affected person samples (Supplementary Fig. 19w–z). Collectively, these outcomes exhibit that Dex stimulates CXCR4 signaling in B-ALL cells and thereby regulates PLCγ2 activation and signaling.
CXCR4 inhibition enhances Dex sensitivity in B-ALL cell strains in vitro and in vivo
To evaluate whether or not Dex-mediated CXCR4 stimulation may have practical penalties on B-ALL cell survival, we first assessed the oncogenic exercise of CXCR4 in human cancers utilizing the Undertaking Achilles database58, which is a genome-wide shRNA library to determine genes that have an effect on most cancers cell survival and proliferation. On this evaluation, we discovered that B-ALL confirmed a adverse shRNA rating in response to CXCR4 shRNA knockdown however which was additionally a lot decrease than in most different human cancers (ranked 29 of 31) (Fig. 7a and Supplementary Fig. 20a), indicating that CXCR4 pathway is important for B-ALL cell survival in accordance with earlier report33. Moreover, in keeping with earlier findings34, CXCR4 activation by SDF-1 elevated the viability of B-ALL cells (Supplementary Fig. 20b). We subsequent investigated whether or not SDF-1-mediated B-ALL cell viability modulated Dex sensitivity. This evaluation revealed a discount in Dex sensitivity within the presence of SDF-1 (Supplementary Fig. 20c, d). The dose-response graph of SDF-1 indicated {that a} focus as little as 5 ng/mL rendered HAL-01 B-ALL cells utterly proof against Dex (Supplementary Fig. 20e). Apparently, CXCR4 antagonists have been enough to revive Dex sensitivity within the presence of SDF-1 (Supplementary Fig. 20f), indicating the performance of its receptor, CXCR4. Aiming to validate the potential position of CXCR4 signaling on the sensitivity of B-ALL cells to Dex, we subsequent handled B-ALL cell strains with completely different CXCR4 antagonists, i.e., AMD3100, AMD3465, WZ811 or MSK122, together with Dex (Supplementary Fig. 20g, h). B-ALL cells have been handled with CXCR4 antagonists at doses that didn’t have an effect on cell development. On this evaluation, B-ALL cell demise was strongly elevated by mixed therapies (Fig. 7b and Supplementary Fig. 20i, j). The power of CXCR4 signaling inhibition to extend GC sensitivity was confirmed by annexin V/PI staining (Fig. 7c–f) in CRISPR-Cxcr4 cells in addition to in siCxcr4 Nalm-6 cells (Supplementary Fig. 20k). Furthermore, inhibition of CXCR4 signaling with AMD3100 was enough to beat Dex resistance in 37% and 48% of Dex-resistant blood and BM samples, respectively (Fig. 7g). It ought to be famous that these CXCR4 antagonists had no impact on GR (Supplementary Fig. 21a, b), and that GR expression was not altered in CRISPR-Cxcr4 cells (Supplementary Fig. 21c, d). To apprehend the mechanisms resulting in the advance of Dex sensitivity mediated by CXCR4 signaling inhibition, we decided whether or not this enhance in cell demise was related to cell cycle arrest. Remedy of B-ALL cells with AMD3100 resulted in elevated Dex-induced cell demise, as manifested by a major quantity of subG1 apoptotic cell inhabitants (Fig. 7h and Supplementary Fig. 21e, f). We subsequent investigated the impact of inhibiting CXCR4 and PLC in vivo. For this, Nalm-6-luciferase-GFP cells have been injected in immunodeficient NSG mice. By utilizing circulation cytometry Ca2+ evaluation in Nalm-6 GFP+ cells derived from the blood of mice at day 27 (Fig. 8a), we noticed that the rise in Ca2+ induced by Dex therapy was lowered within the presence of AMD (Fig. 8b). Constantly, Dex therapy extended mouse total survival (Fig. 8c, d and Supplementary Fig. 21g). Importantly, AMD3465 additional ameliorated survival indicating that blocking CXCR4 signaling in vivo augments sensitivity to Dex by regulating Ca2+ signaling. To additional verify the relevance of the proposed signaling axis and to enhance the translatability of our work in GC-resistant preclinical fashions, we used two in vivo approaches. First, we made our Nalm-6-LUC/GFP cells proof against GC (technique described in Supplementary Fig. 4a). Supplementary Fig. 21h reveals the validation of the mannequin within the NSG mouse mannequin. We then examined this resistant line and confirmed related outcomes to the delicate line (Fig. 8e–h). We additionally confirmed on this experiment that PLC inhibition with U73122 improved mouse survival within the presence of Dex (Fig. 8e–h). Second, we used the intrinsically Dex-resistant B-ALL cell line RCH-ACV11 and confirmed related outcomes utilizing CXCR4 and PLC inhibitors (Fig. 8i). Taken collectively, these information assist a mannequin (Supplementary Fig. 21i) wherein Dex, via activation of CXCR4 signaling, paradoxically induces its personal resistance by activation of the PLC signalosome resulting in apoptotic pathway braking, which in flip is enough to antagonize the proapoptotic results of Dex.
a CXCR4 shRNA ranked by Achilles rating stage in B-ALL and different human tumors. Information have been extracted from the CCLE. Numbers seek advice from most cancers cell sorts (see Supplementary Fig. 20a) amongst which hematological malignancies are depicted in pink. Boxplots present the imply, median, and seventy fifth–twenty fifth percentiles. b Nalm-6 cells have been handled with Dex together with completely different CXCR4 inhibitors. Cell mortality was decided by CCk-8 staining after 48 h. The proportion of useless cells was established after normalizing cells on Ctr cells. Information proven are the imply ± SEM (n = 4 impartial experiments). Two-tailed, unpaired Scholar’s t-test. c, e Cell mortality of CRISPR/Cas9 CXCR4-invalidated RS4,11 and Nalm-6 cells handled with Dex (nM) for 48 h decided by circulation cytometry after annexin V/PI staining. The proportion of early and late apoptotic cells was calculated by the proportion of annexin V-FITC optimistic and annexin V-FITC optimistic plus annexin V-FITC/PI-positive inhabitants, respectively. Information are the imply ± SEM (n = 4 impartial experiments). Two-tailed, unpaired Scholar’s t-test. c ****p < 0.0001, **p = 0.0027 (early); ***p = 0.0004, **p = 0.0069 (late). e ****p < 0.0001, ***p = 0.0005 (late). d, f Consultant circulation cytometry plots from (c, e). g Viability of blood (n = 16 sufferers) and bone marrow (n = 21 sufferers) cells from 37 Dex-resistant main diagnostic ALL affected person samples (see Supplementary Fig. 10b) uncovered to 100 nM Dex and/or 25 μM AMD3100 for twenty-four h, normalized to Ctr situation, Statistical significance was decided by two-tailed, unpaired Scholar’s t-test. Information are the imply ± SD. *p = 0.0136 (blood), **p = 0.0095 (bone marrow). The experiment was carried out underneath the identical circumstances as in Supplementary Fig. 10b. h Cell cycle evaluation of Nalm-6 cells after stimulation with 100 nM Dex and/or 25 μM AMD3100 for 48 h. Cell cycle distribution decided by circulation cytometry after PI staining. Statistical significance was decided by two-tailed, unpaired Scholar’s t-test. *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001. Supply information are offered as a Supply Information file.
a Experimental design of NSG mice therapy experiments. BLI bioluminescence imaging. b Stream cytometry Ca2+ evaluation in Nalm-6 GFP+ cells within the blood at day 27. c Kaplan–Meier survival evaluation of mice transplanted with Nalm-6 GFP-luc cells and handled with Ctr (n = 4), Dex (n = 6), AMD3465 (n = 4), or a mix of Dex and AMD3465 (n = 6), n-values correspond to particular person mice. Statistical significance was calculated with the Log-rank (Mantel-Cox) take a look at. d An instance of bioluminescence imaging of Nalm-6 GFP/luc at day 25 of tumor problem from (c). e Experimental design of NSG mice therapy experiments utilizing Dex-resistant B-ALL fashions. BLI bioluminescence imaging. f Kaplan–Meier survival evaluation of mice transplanted with resistant (R) Nalm-6 GFP-luc cells and handled with Ctr (n = 4), Dex (n = 5), AMD3465 (n = 4), U73122 (n = 4) or a mix of Dex and AMD3465 (n = 5) or and U73122 (n = 6), n-values correspond to particular person mice. Statistical significance was calculated with the Log-rank (Mantel-Cox) take a look at. g An instance of bioluminescence imaging of Nalm-6 R GFP/luc at day 19 of tumor problem from (f). h Averaged of the median survival over time is proven for the completely different therapy teams from (f). Information are the imply ± SEM from (n = 4 for Ctr, AMD3465, U73122; n = 5 for Dex and Dex+AMD and n = 6 for Dex+U73122; n-values correspond to particular person mice). Boxplots present the imply, median, and seventy fifth–twenty fifth percentiles. i Kaplan–Meier survival evaluation of mice transplanted with resistant RCH-ACV cells and handled with Ctr (n = 4), Dex (n = 4), AMD3465 (n = 4), U73122 (n = 4) or a mix of Dex and AMD3465 (n = 5) or and U73122 (n = 5). Statistical significance was calculated with the Log-rank (Mantel-Cox) take a look at. Supply information are offered as a Supply Information file.