1 Introduction
Prostate most cancers (PCa) stands as one of the vital prevalent malignancies affecting males globally, holding a distinguished place amongst male malignant tumors on a worldwide scale. In gentle of the anticipated rise within the aged inhabitants, PCa is poised to grow to be a pivotal concern in illness prevention and administration (Miller et al., 2022). The defining options of PCa primarily contain resistance to androgen deprivation and the propensity for metastasis. Analysis signifies that an estimated one-third of sufferers will finally transition to castration-resistant prostate most cancers (CRPC) (Coleman et al., 2020; Fares et al., 2020). A big majority of CRPC circumstances culminate in bone metastasis, a pivotal issue that influences each the standard of life and the survival charges of PCa sufferers (Zhang et al., 2021). The metastatic cascade of tumor cells encompasses dynamic transformations within the mobile cytoskeleton, the place reshaping is instrumental in fueling invasion and metastasis, reflecting alterations in cell morphology, migration capabilities, cytokinesis, and phagocytic exercise (Xu et al., 2023). Comprised of actin filaments, microtubules, and intermediate filaments, the cytoskeleton has emerged as a focus in current investigations demonstrating how anticancer drugs can transform cytoskeletal elements to confer therapeutic advantages (Ahmad et al., 2023).
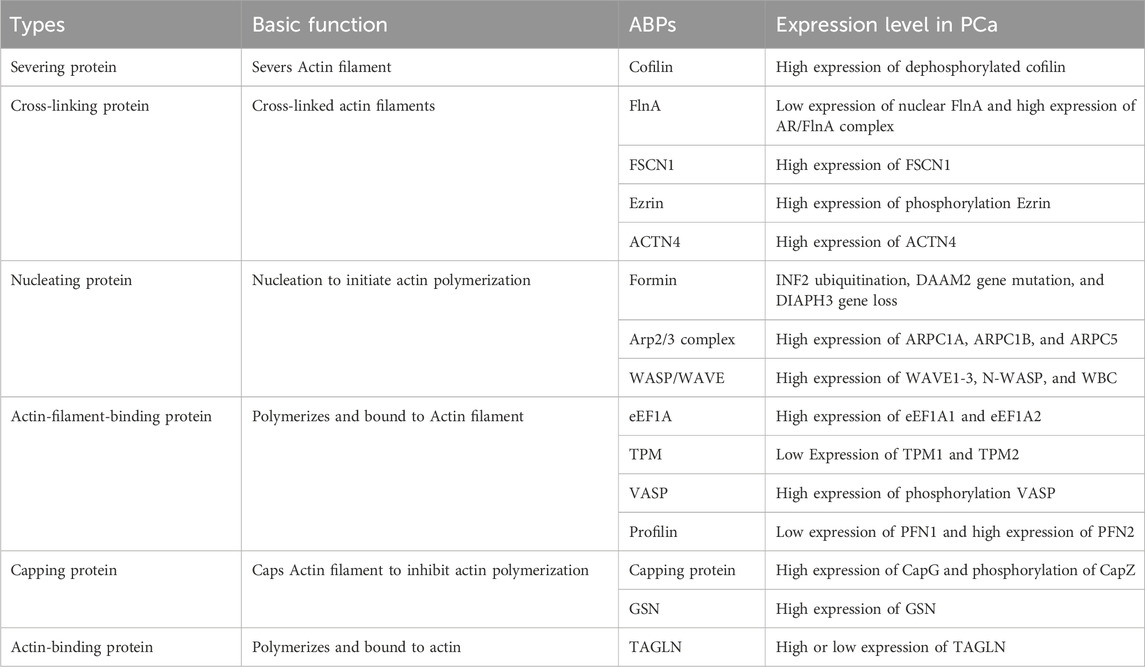
Actin, a predominant constituent inside the mobile cytoskeleton, performs indispensable roles in processes like mobile form modulation, division, and migration (Merino et al., 2020). Nonetheless, the dynamics of actin are finely tuned by a repertoire of actin-binding proteins (ABPs) (Gao and Nakamura, 2022). Working as key regulators, ABPs oversee actin’s involvement in nucleation, elongation, and disassembly processes, successfully bridging the mobile membrane and the nucleus (Kadzik et al., 2020). Since their preliminary characterization within the Nineteen Seventies, over 160 distinct ABPs have been cataloged (Zhang et al., 2021). Illustratively depicted in Determine 1, ABPs might be categorized into a number of varieties based mostly on their roles in modulating completely different sides of actin meeting, encompassing ABPs, actin filament-binding proteins, severing proteins, nucleating proteins, capping proteins, and cross-linking proteins. With various functionalities, numerous ABPs facilitate the aggregation of quite a few actin molecules, orchestrate the formation of latest filaments, facilitate filament elongation, cap filament ends to curb elongation, sever filaments, and interlink filaments (Vakhrusheva et al., 2022). Current literature highlights the involvement of ABPs within the initiation and migration of PCa by way of the regulation of oncogene expression, exemplified by cofilin, the Arp2/3 advanced, TPM1, FlnA, and TAGLN (Izdebska et al., 2020; Kmeťová Sivoňová et al., 2021). Constructing upon these insights, this evaluate succinctly surveys the current developments in our understanding of varied ABPs related to PCa, providing novel avenues for combatting and mitigating the development of this illness.
Determine 1. The connection between ABPs and the cytoskeleton. The cytoskeleton consists of actin filaments, microtubules, and intermediate filaments. The dynamics of actin filaments are regulated by numerous ABPs. Primarily based on the variations in regulating numerous points of actin meeting, ABPs might be divided into various kinds of proteins, together with ABPs, actin filament-binding proteins, severing proteins, nucleating proteins, capping proteins, and cross-linking proteins.
2 Actin filament severing in PCa
Cofilin, a 19 kDa actin filament-severing protein extensively current in eukaryotic cells (Shishkin et al., 2017), is a key member of the ADF/cofilin household, which encompasses ADF (actin-depolymerizing issue), cofilin-1, and cofilin-2, pivotal in regulating the dynamics of mobile actin filaments (Maciver and Hussey, 2002). Whereas ADF and cofilin-1 exhibit widespread expression throughout numerous non-muscle tissues, cofilin-2 is predominantly present in muscle tissues like the guts and prostate, with cofilin-1 being the topic of extra complete research (Bamburg and Wiggan, 2002). Notably, cofilin-1 emerges as a promising pathological biomarker in PCa tissues, providing insights into tumor development, metastasis, and therapy outcomes (Lu et al., 2015; Stark et al., 2017; Kiełb et al., 2023; Sousa-Squiavinato and Morgado-Díaz, 2024). Shishkin discovered an upregulation of mRNA and protein ranges of cofilin-1 in PCa cells in comparison with non-malignant cells (Shishkin et al., 2017). Moreover, differential protein expression analyses in PCa cell strains with various metastatic skills spotlight cofilin-1 as a considerably distinct protein, suggesting its very important position in most cancers invasion and metastasis (Chen et al., 2020). Enhanced filopodium formation and cell migration capabilities in cofilin-overexpressing PCa cells additional underscore the pivotal involvement of cofilin-1 in tumor development (Collazo et al., 2014). Current investigation elucidates the position of cofilin-1 in driving most cancers migration by way of uneven actin polymerization, thereby influencing the cell motility cycle and extracellular matrix interactions (Warner et al., 2024), hinting on the intricate mechanisms by which cofilin regulates actin filaments to affect PCa development.
Varied regulatory mechanisms modulate the exercise of cofilin-1, together with phosphorylation/dephosphorylation, phosphatidylinositol 4, 5-bisphosphate binding, and intracellular pH regulation (Bravo-Cordero et al., 2013; Sousa-Squiavinato and Morgado-Díaz, 2024). Notably in PCa cells, the phosphorylation/dephosphorylation of cofilin-1 at serine 3 (Ser3) is a predominant occasion. Phosphorylation of cofilin Ser3, primarily catalyzed by LIM area kinases (LIMK1 and LIMK2) below regular circumstances (Mizuno, 2013), results in the inactivation of cofilin, thereby stabilizing the actin cytoskeleton (J. Park et al., 2021). LIMK1/2-mediated phosphorylation of cofilin emerges as a essential therapeutic goal in PCa administration, inhibiting cofilin severing exercise, cytoskeletal reorganization, filopodia formation, and chromosomal abnormalities to impede PCa development at early levels (Davila et al., 2003; 2007; Shah and Prepare dinner, 2023). Given the pivotal position of the androgen receptor (AR) in PCa incidence and development, LIMK inhibitors present promise in lowering PCa cell motility, diminishing AR protein stability and transcriptional exercise, thereby holding therapeutic potential in PCa therapy (Mardilovich et al., 2015).
In PCa cells, as depicted in Determine 2, cofilin-1 features as a downstream effector of Rho household GTPases signaling (B. Chen et al., 2019). Rho household GTPases, together with RhoA, RhoB, Rac1, and Rac2, elicit tumor-suppressive results by activating PAK4, ROCK, or immediately stimulating the LIMK/cofilin signaling pathway (Ahmed et al., 2008; Alfano et al., 2012; Lee et al., 2019; X; Zhang et al., 2019). Notably, inside the context of most cancers epithelial-mesenchymal transition (EMT), Remodeling Development Issue-β (TGF-β) assumes a pivotal position in modulating cell cytoskeleton alterations by way of the activation of Rho GTPases signaling pathways. Investigations have indicated cofilin, as a downstream goal of TGF-β, drives the TGF-β-induced invasion and metastasis of PCa cells (Zhu et al., 2006; Collazo et al., 2014). Conversely, alpha-2-macroglobulin (α2-MG), nuclear clusterin (nCLU), and anchoring protein AKAP2 exert reverse management over cofilin dephosphorylation and activation, thereby influencing the dynamics of actin filaments to advertise PCa cell invasion and metastasis (Misra et al., 2005; Moretti et al., 2011; Reggi et al., 2024). Given these findings, the modulation of cofilin phosphorylation to control actin cytoskeleton dynamics in most cancers cells emerges as a promising avenue for potential therapeutic intervention in PCa.
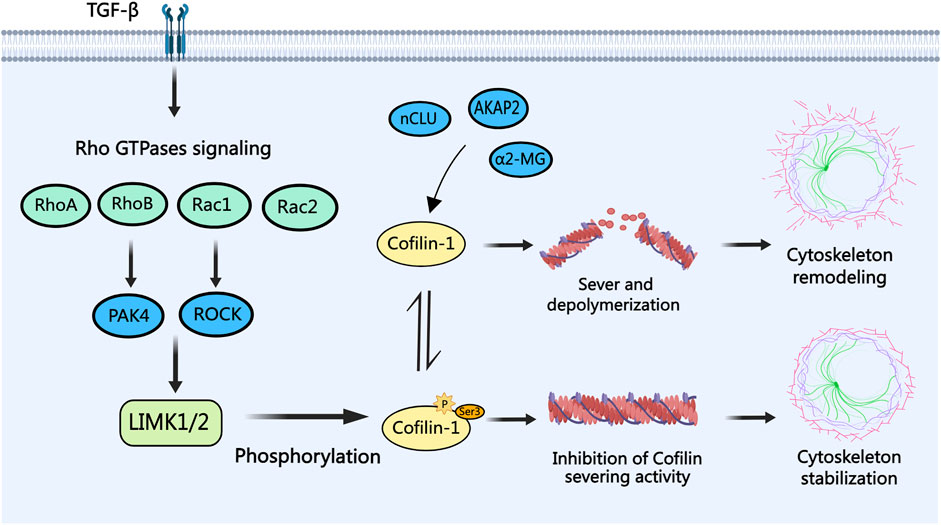
Determine 2. The position of cofilin-1 in PCa. Remodeling progress factor-beta (TGF-β) modulates Rho household GTPases (RhoA, RhoB, Rac1, and Rac2) signaling, resulting in cofilin-1 phosphorylation by way of LIMK1/2. Phosphorylated cofilin-1 loses its severing exercise, guaranteeing actin cytoskeleton stability. Conversely, activation of cofilin-1 by proteins like α2-MG, nCLU, and AKAP2 by way of dephosphorylation facilitates actin filament severing and depolymerization, culminating in cytoskeletal reorganization, filopodia formation, and consequent migration and invasion of PCa cells.
Cofilin-1 performs a vital position within the pharmacological mechanisms of varied therapeutic medicine for PCa. As an illustration, docetaxel prompts the cofilin-1/Paxillin signaling pathway, prompting apoptosis in PCa by suppressing cofilin-1 expression (Xiao et al., 2016). Pérez-Martínez utilized nanodelivery know-how to manage a particular siRNA concentrating on cofilin-1 to human PCa cells, downregulating cofilin-1 expression and enhancing docetaxel-induced cytotoxicity (Pérez-Martínez et al., 2012). In one other examine, Lampe employed nanodelivery know-how to manage the drug cabazitaxel, leading to elevated expression of phosphorylated cofilin at Ser3, impacting most cancers cell migration and invasion pathways (Lampe et al., 2023). Thus, chemotherapy medicine can exhibit anticancer results by inhibiting cofilin-1 expression or deactivating phosphorylated cofilin-1. Resistance to PCa therapy may additionally be linked with cofilin-1, as revealed in a proteomic examine evaluating docetaxel-sensitive and -resistant PCa cells by Zu et al., figuring out cofilin as a key differential protein in PCa cell migration (Zu et al., 2015). Enzalutamide, a extensively used AR inhibitor, is related to drug resistance in superior therapy levels. Paller’s examine on the mix remedy of the TGF-β receptor I inhibitor galunisertib and the antiandrogen enzalutamide demonstrated important discount in cofilin expression ranges, thereby optimizing the therapeutic response to enzalutamide (Paller et al., 2019). Investigations on azithromycin resistance mechanisms unveiled that cofilin-1 triggers the P38 MAPK signaling pathway, facilitating actin filament cytoskeletal reorganization and elevating the expression and drug efflux exercise of multidrug resistance protein 1, consequently compromising the efficacy of PCa therapy (Chen et al., 2020). Lively elements derived from pure vegetation inhibit cell viability and induce apoptosis in PCa cells mediated by way of cofilin-1. For instance, ursolic acid, a conventional Chinese language drugs part, induces apoptosis in PCa cells by concentrating on the ROCK/PTEN-mediated cofilin-1 mitochondrial translocation (Gai et al., 2016; Mu et al., 2018). Huperzine E, a key part in conventional Chinese language drugs, triggers apoptosis in human PCa cells by participating cofilin-1 and mTORC1 pathways (X. He et al., 2017). In distinction, Huperzine F enhances cofilin-1 phosphorylation, impeding PCa cell proliferation by way of the promotion of actin aggregation and cofilin-actin rod formation (Ren et al., 2012). The compound curcumin suppresses cell motility by diminishing protein kinase D1 downstream goal exercise and elevating cofilin phosphorylation ranges (Sundram et al., 2012). Subsequent to therapy with thapsigargin, there’s a notable rise in cofilin-1 protein expression in PCa cells, hindering tumor progress by modulation of the F-actin/cofilin-1/paxillin pathway and the Akt-mTOR pathway (Huang et al., 2018).
Tumor suppressor genes and oncogenes play pivotal roles within the initiation and development of PCa. Alterations or inactivation of those genes can precipitate tumor progress and metastasis. HSPB6, a big tumor suppressor gene, carries prognostic implications; decreased expression in PCa is usually related to a bleak outlook. Feng et al. investigated therapeutic activators and recognized 8-Br-cGMP as able to activating HSPB6 and inducing dephosphorylation of phosphorylated Cofilin, eliciting apoptosis in PCa cells. Notably, the synergistic impact of 8-Br-cGMP and quinidine notably bolstered HSPB6 transcription ranges, successfully restraining PCa progress and providing promising prospects for PCa therapy (Feng et al., 2024). Overexpression of miR-608 targets RAC2/PAK4/LIMK1/cofilin, thereby impeding PCa development (X. Zhang et al., 2019). Equally, upregulation of the tumor suppressor gene miR-143 thwarts the LIMK1/cofilin signaling pathway, reinstating arsenic-induced PCa apoptosis (Ngalame et al., 2016). Oncogenic Circ-FOXO3 boosts the viability, motility, and proliferation of PCa cells by way of the miR-1299/cofilin-2 axis, whereas suppressing apoptosis (P. Li et al., 2021). Moreover, the oncogene lncRNA-SOX2 enhances cell proliferation and migration in PCa by way of the miR-369–3p/cofilin-2 axis (Wo et al., 2019). Overexpression of the oncogene ERG in prostate epithelium spurs cofilin exercise, fostering EMT that fuels prostate tumor genesis (Griner et al., 2015). Moreover, the oncogene SOX11 reveals heightened transcriptional activation in metastatic PCa tumors, elevating cofilin exercise and cell migratory capabilities (Hirokawa et al., 2020). Thus, cofilin emerges as a vital downstream effector protein influenced by an array of tumor suppressor genes and oncogenes, regulating the mobile cytoskeleton by modulating actin filaments, governing cell proliferation, apoptosis, and migration, and enjoying a pivotal position within the evolution and development of PCa. An array of therapeutic methods concentrating on cofilin is detailed in Desk 1.
3 Actin filament cross-linking in PCa
3.1 Filamin (FlnA)
FlnA, a 280 kDa actin-crosslinking protein, acts as a pivotal nuclear transcription regulator immediately influencing AR performance within the nucleus, contributing to the pathogenesis and development of PCa (Loy et al., 2003). Current investigations counsel that serum FlnA ranges can function an revolutionary biomarker for stratified PCa screening, surpassing PSA in screening efficacy (Panigrahi et al., 2019; Leitão et al., 2022; Mahaveer Chand et al., 2024). Performance of FlnA in PCa cells varies based mostly on its mobile localization, as illustrated in Determine 3: whereas cytoplasmic localization of FlnA fosters tumor progress and migration, nuclear localization hampers tumor development and migration (Shao et al., 2016). The position of calpain is essential for the nuclear translocation of FlnA and subsequent AR activation. Usually, calpain cleaves FlnA to generate a 90 kDa carboxy-terminal fragment (FlnACT), which navigates to the nucleus by way of cell signaling pathways and regulates transcription elements. In AR-negative cells, calpain inhibitors safeguard FlnA and the AR ligand-binding area from cleavage (T. Liu et al., 2014). In a examine carried out by Salimi, inhibition of calpain-mediated FlnA cleavage resulted in lowered manufacturing of FlnACT, subsequently diminishing the proliferation, migration, and colony-forming capabilities of PCa cells (Salimi et al., 2018). Moreover, McGrath confirmed that calpain-mediated cleavage of FlnA drives the nuclear localization of the AR transcriptional coactivator FHL2. FHL2 activation sustains AR exercise in CRPC, thereby impeding the effectiveness of Enzalutamide therapy in CRPC circumstances (McGrath et al., 2013). Consequently, combining calpain inhibitors with ADT could emerge as a viable therapy technique to impede or postpone the development of PCa.
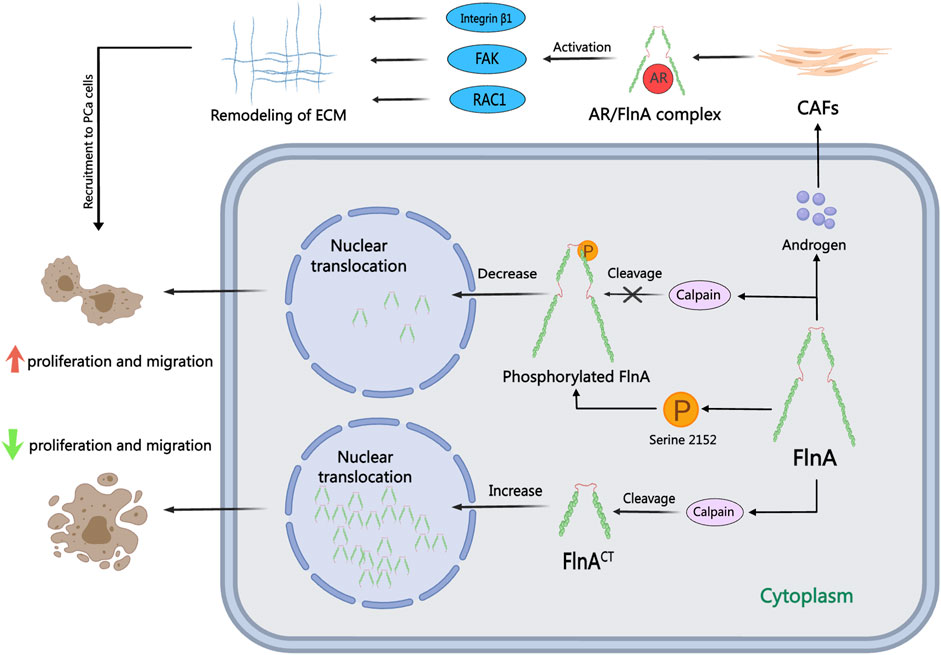
Determine 3. The position of FlnA in PCa. FlnA can bear calpain-induced cleavage to provide a 90 kDa carboxy-terminal fragment (FlnACT), which translocates to the nucleus by way of mobile signaling cascades. Within the cytoplasm, FlnA promotes tumor proliferation and migration, whereas nuclear localization inhibits these processes. Within the presence of androgens, FlnA varieties an AR/FlnA/integrin β1 advanced activating Rac1, FAK, and MMP-2 pathways, driving extracellular matrix reworking and enhancing the invasion functionality of CAFs.
FlnA, a pivotal AR protein accomplice, performs a big position within the metastasis and dissemination of PCa in response to androgens (Castoria et al., 2017). The notable lack of FlnA’s nuclear localization is a trademark of CRPC, primarily attributed to FlnA phosphorylation, which hampers cleavage and nuclear translocation. This alteration disrupts the transcriptional profile orchestrated by AR, but augmentation of nuclear FlnA expression can reestablish androgen sensitivity in CRPC cells (Savoy et al., 2015). In investigations on resistance to antiandrogen drugs like bicalutamide, Wang induced the re-entry of FlnA into the nucleus, resulting in diminished Akt phosphorylation. Nuclear FlnA expression can impede the proliferation of androgen-independent cells, thus reinstating sensitivity to bicalutamide therapy. Protein Kinase A (PKA) inhibitors have the capability to hinder FlnA phosphorylation at Ser 2,152, thereby impeding FlnA cleavage and its nuclear translocation (Wang et al., 2007). Furthermore, PKA inhibitors can suppress AR transcriptional exercise and impair cell progress in low-androgen environments (Bedolla et al., 2009). The mix of the pure product genistein with polysaccharides induces FlnA cleavage and nuclear localization, inhibiting FlnA phosphorylation and consequently sensitizing PCa cells to ADT (Mooso et al., 2012). AR/FlnA complexes might be utilized to gauge susceptibility and monitor illness development in PCa (Zhang et al., 2023). Stimulated by androgens, the AR/FlnA advanced in cancer-associated fibroblasts (CAFs) recruits integrin β1 and prompts Rac1 and FAK, thereby facilitating the migration of PCa cells (Castoria et al., 2011). Di Donato has pioneered the event of AR-derived peptides in laboratory settings that may selectively neutralize the impact of androgens on PCa cells and CAFs. These peptides inhibit PCa cell invasion by disrupting the meeting of the AR/FlnA/integrin β1 advanced and activating downstream Rac1, FAK, and MMP-2 signaling pathways (Di Donato et al., 2015; Di Donato et al., 2021a; Di Donato et al., 2021b). This focused intracellular method towards the AR/FlnA advanced in PCa-associated fibroblasts demonstrates enhanced efficacy in overcoming resistance to second-generation inhibitors in PCa.
Furthermore, FlnA contributes to the pathogenesis and development of PCa by way of a myriad of mechanisms past its reference to the AR. Of their examine, Cahuzac et al. recognized that the PARP inhibitor olaparib targets pre-activated autophagy, leading to diminished SQSTM1/p62 nuclear localization and an elevation in FlnA expression, enhancing therapy flexibility (Cahuzac et al., 2022). Enhanced ranges of Remodeling Acidic Coiled-Coil Protein 3 (TACC3) disrupt the interplay between FlnA and meckelin, impeding main cilia formation in PCa cells (Qie et al., 2020). The tumor-suppressive position of the TGF-β-mediated FlnA/R-Smad signaling pathway is noticed in regular prostate epithelial cells (S. Assinder and Cole, 2011). Moreover, the lengthy non-coding RNA LINC01002 demonstrates potential anti-cancer efficacy in PCa by concentrating on the miR-650/FlnA pathway (Qian et al., 2023). Whereas the involvement of FlnA in modulating the aforementioned molecular pathways is acknowledged, its exact mechanisms of motion stay incompletely understood. Future investigations ought to completely discover these pathways to advance the event of therapeutic methods specializing in FlnA regulation.
3.2 Fascin-1 (FSCN1)
FSCN1, a part of the Fascin household of ABPs, possesses a molecular weight of roughly 55 kDa (H. Liu et al., 2021). This protein is extensively studied in most cancers cell metastasis because of its position in cross-linking actin filaments into densely packed parallel constructions (Z. Li et al., 2022). Neuroendocrine prostate most cancers (NEPC), a high-grade subtype of PCa, solely reveals FSCN1 expression (Turpin et al., 2023), underlining its affiliation with elevated invasiveness, metastasis, and decreased affected person survival charges (Darnel et al., 2009; Zhang et al., 2023). Overexpression of FSCN1 in PCa cells triggers the era of invasive pseudopodia, fostering cell invasion, migration, and EMT (Sarantelli et al., 2023), making FSCN1 a compelling therapeutic goal for PCa.
Research have outlined FSCN1’s involvement in PCa development by way of various mobile signaling pathways. The actin-binding protein Cortactin collaborates with FSCN1, modulating extracellular vesicle (EV) launch, pseudopodia formation, MMP-9 secretion, and associated processes (Van Audenhove et al., 2014; Beghein et al., 2018). Synaptotagmin-like 2 (SYTL2) is intertwined with excessive metastasis charges, superior tumor staging, and poor prognosis in PCa. SYTL2 upregulation impedes the proteasomal degradation of FSCN1 and enhances PCa cell mobility by regulating pseudopodia formation. The SYTL2-FSCN1-pseudopodia axis holds promise as a therapeutic goal for metastatic PCa (Z. Li et al., 2023). N-Myc proto-oncogene protein (N-Myc) escalation has been linked to the malignant development of PCa. An intricate interaction exists between N-Myc and FSCN1, the place N-Myc potentiates FSCN1 expression and, conversely, FSCN1 can suppress N-Myc expression (G. He et al., 2020). Moreover, miR-145, miR-24–3p, and miR-145–5p, downregulated in PCa, act as tumor suppressors by immediately modulating FSCN1 (Fuse et al., 2011). The oncogenic lengthy non-coding RNA colon cancer-associated transcript 1 (CCAT1), upregulated in PCa, regulates miR-24–3p expression. Knockdown of CCAT1 enhances paclitaxel sensitivity in PCa by modulating the miR-24–3p/FSCN1 pathway (X. Li et al., 2020). Equally, the lengthy non-coding RNA PCa-associated transcript 1 (PCAT-1) contributes to PCa by lowering miR-145–5p expression, selling cell proliferation, migration, invasion, and hindering apoptosis by way of FSCN1 activation (Weibo et al., 2017). Histone H3 serves a vital position within the temporal and spatial regulation of nuclear FSCN1. Nuclear FSCN1 exerts anti-cancer results by way of actin bundling for chromatin group and environment friendly DNA restore (Lawson et al., 2022). Inhibiting histone H3 phosphorylation, inflicting nuclear FSCN1 accumulation and actin filament meeting, could clarify the anti-cancer properties of nuclear FSCN1. As proven in Determine 4, these collective findings unravel assorted signaling mechanisms by way of which FSCN1 influences PCa metastasis and development. Nonetheless, focused therapeutic medicine particularly for FSCN1 are missing, paving the way in which for future improvement of multi-targeted medicine to concurrently inhibit various FSCN1-associated signaling pathways. Notably, the small molecule inhibitor NP-G2-044 concentrating on FSCN1 has proven efficacy in ocular neovascularization (Bai et al., 2023), hinting at its potential in PCa because it inhibits cell migration and filopodia formation.
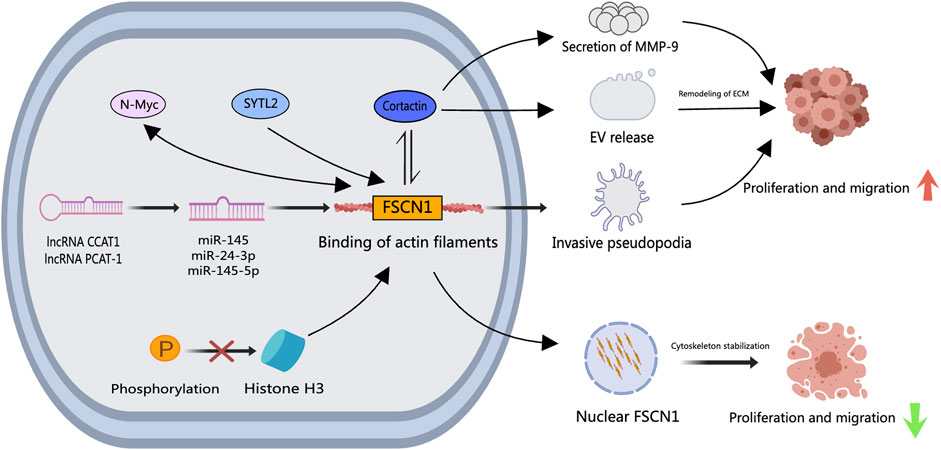
Determine 4. The position of FSCN1 in PCa. Upstream signaling elements similar to N-Myc, SYTL2, CCAT1, and PCAT-1 improve FSCN1 expression in PCa cells, the place a bidirectional regulatory mechanism between N-Myc and FSCN1 exists. FSCN1 facilitates the formation of invasive podosomes, selling PCa cell invasion and metastasis. Along with Cortactin, FSCN1 regulates the discharge of Extracellular Vesicles and MMP-9 secretion, inducing extracellular matrix reworking that facilitates PCa cell invasion and metastasis. Inhibition of histone H3 ends in nuclear FSCN1 accumulation and actin filament meeting, exerting repressive results on PCa cell invasion and metastasis.
3.3 Ezrin
Ezrin, a member of the Ezrin-radixin-moesin (ERM) protein household, features as a vital hyperlink between the plasma membrane and actin cytoskeleton, profoundly influencing most cancers development and metastasis (Clucas and Valderrama, 2014; Kawaguchi and Asano, 2022). Extremely prevalent in PCa cells and circulating tumor cells (CTCs), Ezrin serves as a helpful biomarker for tumor metastasis and prognostication (Z. Chen et al., 2022; Valdman et al., 2005). Analysis signifies that Ezrin governs the migratory and invasive capabilities of PCa cells by way of phosphorylation (Chuan et al., 2006). Within the presence of androgens, Ezrin phosphorylation triggers c-Myc oncogene overexpression. This elevated c-Myc protein synthesis and degradation inhibition confer an invasive phenotype upon PCa cells, with downstream signaling pathways involving Akt and GSK-3β (Chuan et al., 2010). Notably, the anti-adhesive transmembrane sialomucin Podocalyxin varieties a posh with Ezrin, the place Podocalyxin expression correlates with elevated Ezrin phosphorylation. The Podocalyxin/Ezrin advanced induces heightened MMP expression, enhanced MAPK and PI3K exercise, culminating in a extra aggressive PCa phenotype (Sizemore et al., 2007). Furthermore, the cell floor adhesion molecule CD44 complexes with Ezrin to reinforce tumor cell adhesion and invasion when co-localized in PCa-endothelial cells, facilitating tumor development (Herrlich et al., 2000; Harrison et al., 2002). Conversely, Protein 4.1B—a structural area protein incorporating Ezrin—acts as a unfavourable modulator in PCa, impeding its development and metastasis (Wong et al., 2007). Within the realm of conventional Chinese language drugs, the energetic part baicalin downregulates Ezrin in PCa cells and mice, restraining cell proliferation, inducing apoptosis, arresting the cell cycle, and lowering tumor dimensions. Depletion of Ezrin heightens baicalin’s inhibitory affect on PCa cell proliferation, underscoring the pivotal regulatory position of baicalin intervention and Ezrin ranges in PCa development (Ma et al., 2020). Moreover, Ezrin small molecule inhibitors like NSC 305787 and NSC 668394, initially employed in osteosarcoma therapy (Bulut et al., 2012), maintain potential for PCa remedy by hindering Ezrin phosphorylation and its interplay with actin.
3.4 α-Actinin-4 (ACTN4)
ACTN4, a member of the spectrin gene superfamily, is a cytoskeletal protein crucially linked to cell motility, most cancers invasion, and metastasis. Through the transition from androgen-dependent to androgen-independent PCa, ACTN4 expression escalates, stimulating the transcription of genes related to proliferation and metastasis (S. Park et al., 2020). Differentiating protein expression within the serum exosomes of sufferers with CRPC, Ishizuya famous a particular elevation solely in ACTN4 ranges in CRPC sufferers in comparison with these present process ADT. Notably, downregulating ACTN4 expression utilizing RNA interference considerably curtailed the proliferation and invasion of PCa cells (Ishizuya et al., 2020). Managed by the upstream zinc finger protein CTCF, ACTN4 is upregulated by way of the lncRNA HOXA11-AS/miR-518b signaling pathway, fostering PCa proliferation and migration (Xing et al., 2020). These research illustrate that ACTN4 upregulation propels the proliferation and motility of PCa cells; nevertheless, no efficient and focused medicine for ACTN4 at the moment exist.
4 Actin filament nucleation in PCa
4.1 Formin household proteins
Formins play a pivotal position in actin nucleation and filament meeting, essential in numerous mobile features similar to cell division, migration, and adhesion. INF2, a member of the formin subfamily, emerges as a PCa-linked regulator, impacting tumor development by way of various pathways (Zhao et al., 2022). Generally mutated in main PCa, the SPOP gene triggers aberrant polyubiquitination of INF2, disrupting INF2’s facilitation of mitochondrial fission and fostering PCa improvement (Jin et al., 2017). Investigating Formin subfamily member DAAM2 gene mutations in Androgen Insensitivity Syndrome, Knerr highlighted their potential contribution to AR inactivation and PCa development. DAAM2-mediated actin meeting drives AR clustering in response to dihydrotestosterone, inducing transcriptionally energetic droplet formation and doubtlessly influencing PCa development in direction of androgen deprivation resistance (Knerr et al., 2023). Silencing of Formin subfamily member DIAPH3 stimulates the formation and shedding of extracellular vesicles in PCa cells, hastening tumorigenesis and metastasis in vivo by modulating the tumor microenvironment (Kim et al., 2014). Notably, DIAPH3 inhibition of oncosome formation in EVs is counteracted by the lack of DIAPH3 gene expression in metastatic PCa cells (Di Vizio et al., 2009). Depletion of DIAPH3, as recognized by Reis-Sobreiro et al., prompts nuclear instability and confers an amoeboid-like phenotype to PCa cells, fostering tumor cell propagation and migration (Morley et al., 2014; Reis-Sobreiro et al., 2018). In a contrasting impact, DIAPH3 knockdown or silencing heightens susceptibility to paclitaxel and epothilone B medicine (Morley et al., 2015; Lin and Windhorst, 2016). Thus, the intricate involvements of varied Formin subfamily members in PCa development, encompassing ubiquitination, mutations, and gene loss, place them as very important regulators and potential therapeutic targets for impeding tumor cell proliferation and migration.
4.2 Actin-related protein 2/3 advanced (Arp2/3 advanced)
The Arp2/3 advanced is a widely known actin nucleation protein consisting of ARP2, ARP3, and ARPC1 to ARPC5 (Goley and Welch, 2006). Along with ABPs like Formin, N-WASP, WAVE1, and Cortactin, the Arp2/3 advanced collectively regulates the formation of secondary protrusions from main protrusions (Yang and Svitkina, 2011; Giri et al., 2013). This course of allows PCa cells to reinforce their migration and invasion skills by growing invasive pseudopods and degrading the extracellular matrix (Desai et al., 2008). Evaluation of databases signifies that ARPC1A can independently predict lymph node metastasis and illness prognosis in PCa (N. Xu et al., 2020). Practical in vitro experiments have revealed that Sign Transducer and Activator of Transcription 3 (STAT3) performs a task in regulating the transcription of ARPC1A. Lowering ARPC1A expression promotes ferroptosis, finally resulting in lowered cell viability and invasion capabilities of PCa cells (Ji et al., 2022). Suppression of ARPC1B expression impedes the migration and invasion of PCa cells, inflicting cell cycle arrest within the G2/M section. Assessing sufferers’ scientific prognosis might be achieved by inspecting the mix of ARPC1B with PTEN or ERG (Gamallat et al., 2022). Krüppel-like issue 4 (KLF4) acts as a transcriptional activator of ARPC5, selling PCa development by way of the activation and upregulation of ARPC5 by way of the Notch and Wnt pathways (Qu et al., 2023). The aforementioned research present preliminary insights into the numerous roles of ARPC1A, ARPC1B, and ARPC5 inside the Arp2/3 advanced in PCa, underscoring their significance in cell migration and invasion. Concentrating on the exercise of the Arp2/3 advanced with particular medicine could current novel therapeutic approaches for PCa therapy.
4.3 WASP/WAVE household proteins
The Wiskott-Aldrich Syndrome Protein (WASP) and WASP Household Verprolin Homologous Protein (WAVE) household proteins, encompassing subtypes like WAVE1, WAVE2, WAVE3, WASP, and N-WASP, act as scaffold proteins in activating the ARP 2/3 advanced (Takenawa and Suetsugu, 2007). Fernando and Moazzam have reported a powerful correlation between WAVE1, WAVE3, and the invasiveness of metastatic PCa cells (Fernando et al., 2008; 2010; Moazzam et al., 2015). The WASP/WAVE proteins are thought-about potential therapeutic targets towards PCa, with N-WASP believed to be a stronger activator of the ARP 2/3 advanced in comparison with Cortactin (Burianek and Soderling, 2013; Mughees et al., 2021). These household proteins are regulated by upstream Rho GTPase protein alerts, triggering ARP 2/3 advanced activation, actin cytoskeleton reworking, and the formation of invadopodia (Weeks et al., 2016; Rana et al., 2021). Hebbrecht has developed nanobodies that focus on N-WASP, modulating the N-WASP-Arp2/3 advanced interplay, thereby lowering invasive pseudopod formation and matrix degradation throughout PCa invasion (Hebbrecht et al., 2017). Collapsin Response Mediator Protein-1 (CRMP1) acts as a tumor suppressor and reveals lowered expression in superior PCa tissues. CRMP1 has the aptitude to bind to WAVE1 and the ARP 2/3 advanced, inhibiting EMT and the invasive potential of PCa cells (Cai et al., 2017). The WAVE Regulatory Advanced (WRC), essential for activating ARP 2/3, consists of proteins similar to ABI2 and WAVE2. PIM1 kinase can phosphorylate ABI2, enhancing WRC exercise and selling actin dynamics to drive PCa invasion (Jensen et al., 2023). By concentrating on the pivotal position of the WASP/WAVE household proteins in actin nucleation branching, the event of particular inhibitors or antagonists holds promise. As an illustration, Peterson recognized that cyclomodulin 187–1 selectively targets N-WASP, disrupting the interplay between WASP/WAVE household proteins and the ARP 2/3 advanced, presenting a possible therapeutic technique for PCa (Peterson et al., 2001). Determine 5 illustrates the roles and mechanisms of nucleation proteins in PCa.
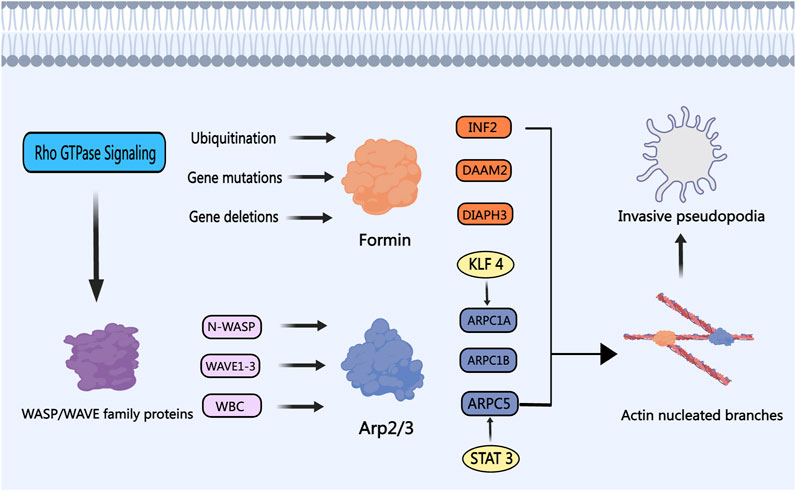
Determine 5. The Function of Nucleation Proteins in PCa. The WASP/WAVE household proteins are modulated by upstream signaling elements, notably the Rho GTPase protein household, which in flip prompts the nucleation exercise of the Arp2/3 advanced. Transcription activators like STAT3 and KLF4 improve the Arp2/3 advanced exercise, fostering the event of invasive and metastatic phenotypes in PCa cells. Members of the Formin subfamily, together with INF2, DAAM2, and DIAPH3, bear ubiquitination, gene mutation, and gene loss in PCa, respectively. Depletion of Formins ends in the event of invasive pseudopodia, thereby selling PCa cell invasion and metastasis.
5 Actin filament meeting and elongation in PCa
5.1 eEF1A
The eEF1A household proteins, comprising eEF1A1 and eEF1A2, play a task in regulating actin filament meeting in PCa development. In each cultured PCa cells and tissues, eEF1A2 demonstrates elevated ranges of mRNA and protein in comparison with eEF1A1 (Scaggiante et al., 2012). Conversely, eEF1A1 is notably expressed in osteoblasts neighboring metastatic tumor cells (Rehman et al., 2012). Enhanced eEF1A1 expression has been recognized in PCa sufferers with Gleason scores of seven to eight, correlating positively with unfavorable scientific outcomes. Notably, the GT75 DNA aptamer developed by Bosutti et al. targets the eEF1A1-actin advanced in invasive PCa cells, resulting in lowered cell viability and elevated cell dying, suggesting eEF1A1 as a possible therapeutic goal for superior PCa (Bosutti et al., 2022). In a examine screening PCa resistance-related genes to metformin, Chen et al. noticed that metformin resistance in PCa is linked to eEF1A1 activation (Chen et al., 2020). Consequently, therapeutic methods aiming to disrupt the interplay between eEF1A and actin could show useful in inhibiting actin filament meeting and PCa cell migration.
5.2 Tropomyosin (TPM)
Tropomyosin encompasses two most important subtypes, TPM1 and TPM2, which work together with actin to assemble a steady cytoskeleton. In PCa cells, each TPM1 and TPM2, often known as tumor suppressor genes, exhibit suppressed expression ranges (Assinder et al., 2010). The lowered expression of TPM2 in PCa affected person tumor tissues is considerably correlated with a much less favorable prognosis, suggesting that downregulation of TPM2 predicts poor outcomes for PCa sufferers (Varisli, 2013). TPM2 inhibits androgen-independent proliferation, invasion, and the expansion of subcutaneous xenograft tumors in PCa cells by activating the Hippo pathway and suppressing downstream goal genes of YAP1 (Wu et al., 2023). Subsequently, concentrating on the expression or practical modulation of TPM2 could possibly be an efficient method to hinder PCa development. Moreover, TPM1 expression is downregulated in PCa cells, doubtlessly linked to exosomal miR-183. miR-183 enhances the proliferation and invasion of PCa cells by lowering TPM1 expression (Dai and Gao, 2021). Future methods involving the upregulation of TPM or the activation of the Hippo pathway by way of gene modifying applied sciences to exert its anti-cancer results could maintain promise as efficient therapeutic avenues.
5.3 Vasodilatation stimulates phosphoprotein (VASP)
VASP, a member of the Ena/VASP protein household, performs a vital position in initiating PCa migration by way of phosphorylation. Nitric oxide (NO) in prostate cells can activate the cGMP/PKG signaling pathway, leading to VASP phosphorylation (Prepare dinner and Haynes, 2007). Lysophosphatidic acid (LPA), a bioactive lipid related to motility and invasive properties in numerous most cancers cell strains, induces VASP phosphorylation upon activation of LPA receptors, a pivotal step in lamellipodia formation and cell migration (Hasegawa et al., 2008). Microcystin-LR (MC-LR), a possible human carcinogen, promotes microfilament rearrangement and cell invasion in PCa cells by enhancing VASP, Ezrin, and ERK phosphorylation (X. Zhang et al., 2022). Given the importance of VASP phosphorylation in PCa migration, therapeutic methods concentrating on molecules like NO, LPA, and MC-LR to inhibit or antagonize VASP phosphorylation, or immediately block VASP phosphorylation, provide promising avenues for therapeutic interventions.
5.4 Profilin
Profilin, a extensively distributed actin filament-binding protein with a molecular weight of round 15 kDa, performs a significant position in regulating actin filament polymerization and elongation dynamics together with cofilin (FU et al., 2008). These proteins collectively affect numerous processes in tumor development, together with migration and invasion of PCa cells. Usually, invasive tumors exhibit low Profilin expression, resulting in mitotic defects, chromosomal instability, and heightened malignancy in tumor cells (Scotto et al., 2023). Present analysis in PCa predominantly focuses on Profilin subtypes PFN1 and PFN2. Research point out that elevated PFN1 expression enhances the effectiveness of chemotherapy medicine and chemotherapy-induced cell dying (Saurav and Manna, 2022). Cathepsin X, recognized as a PCa promoter, could hinder clathrin binding by cleaving PFN1 (Pečar and Kos, 2015). The compromised operate of PFN1 because of cathepsin X motion ends in elevated motility and invasiveness of PCa cells (Pečar et al., 2013). Eicosapentaenoic acid (EPA) reduces the acetylation stage of PFN1, affecting its localization and impeding PCa cell migration and invasion by inhibiting lamellipodia or filopodia formation (C. He et al., 2024). Moreover, by way of interplay with AR, the transcription issue OCT1 enhances signaling pathways linked to PCa development, with PFN2 recognized as a goal gene of OCT1 in AR-negative PCa cells. Knockdown of PFN2 considerably inhibits tumor cell progress, proposing PFN2 concentrating on as a possible therapeutic avenue for CRPC (Obinata et al., 2022). Determine 6 illustrates the roles and mechanisms of ABPs in PCa.
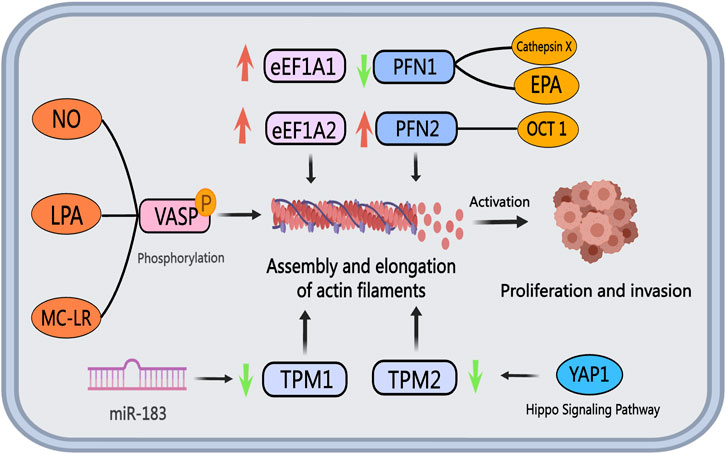
Determine 6. The position of actin-binding proteins in PCa. The eEF1A household proteins, eEF1A1, and eEF1A2, show upregulation in PCa cells and tissues. Tropomyosin, performing as a tumor suppressor gene, reveals diminished expression ranges of TPM1 and TPM2 in PCa, facilitating most cancers cell invasion and metastasis. Extracellular vesicle miR-183 downregulates TPM1 expression, whereas lowered TPM2 expression impacts the Hippo pathway and downstream YAP1 goal genes. NO, LPA, and MC-LR promote VASP phosphorylation, influencing cytoskeletal reworking and fostering PCa invasion and metastasis by modulating actin filament meeting and elongation. Cathepsin X and EPA inhibit PFN1 expression, whereas OCT1 boosts PFN2 expression, leading to elevated tumor cell proliferation and invasion capability.
6 Actin filament capping in PCa
6.1 Capping proteins
Capping proteins bind to the barbed ends of actin filaments, facilitating fast filament progress and influencing cell motility. In PCa, CapG expression is notably elevated in comparison with matched non-cancerous prostate tissue, impacting cell proliferation and apoptosis by way of the activation of the Caspase 6/Caspase 9/Bcl-2/p-Akt/Akt signaling pathway (T. Li et al., 2016). The heterodimeric type of CapZ, consisting of CAPZA1 and CAPZB2, acts as a substrate for the oncogenic PIM1 kinase. Phosphorylation of CAPZ by PIM1 diminishes its actin-capping operate, enhancing PCa cell adhesion and migration (Santio et al., 2020). Concentrating on the elevated expression and phosphorylation of capping proteins in PCa presents a possible therapeutic method.
6.2 Gelsolin (GSN)
Gelsolin, a member of the gelsolin superfamily, primarily features in severing and capping actin filaments, thereby regulating the cell cytoskeleton. Performing as an AR co-activator, GSN reveals excessive expression in late-stage PCa immune to endocrine remedy (Culig et al., 2005). Oelrich et al. efficiently inhibited the IL6-mediated neuroendocrine differentiation in NEPC cells by concentrating on and lowering GSN expression (Oelrich et al., 2021). NEPC cells secrete neurotensin, which boosts PCa cell invasiveness by activating neurotensin receptors and fostering GSN-related processes (Hashimoto et al., 2015). Subsequently, GSN performs a essential position in regulating the neuroendocrine differentiation in PCa. Concentrating on the interaction between GSN and AR holds promise as a possible therapeutic technique for PCa (Nishimura et al., 2003).
7 Actin binding in PCa
Transgelin (TAGLN) is a protein that modulates actin polymerization, essential for stabilizing and gelating the actin cytoskeleton. Proteomic and bioinformatic analyses point out that TAGLN is usually a helpful diagnostic biomarker for benign prostatic hyperplasia and PCa (Kmeťová Sivoňová et al., 2021; Su et al., 2023). TAGLN is usually linked to tumor suppression, with experiences of its downregulation in PCa tissue (Prasad et al., 2010). The lower in TAGLN expression disrupts the actin cytoskeleton, a pivotal think about PCa development. TRAF6-mediated proteasomal degradation contributes to TAGLN downregulation, enhancing PCa cell proliferation and suppressing migration by activating the NF-κB and Myc signaling pathways (Wen et al., 2021). Nonetheless, conflicting outcomes from numerous research on TAGLN and its mobile detection exist. Elements like TGF-β1 and kallikrein-related peptidase-4 (KLK4) induce CAF-like behaviors, growing TAGLN expression in prostate stromal cell strains, thereby triggering extracellular matrix reworking and invasive metastasis (Untergasser et al., 2005; Kryza et al., 2017). These findings spotlight the differential expression of TAGLN in PCa tissues and the particular mechanisms involving regulatory targets like TRAF6, NF-κB, and Myc. In essence, gene regulation of TAGLN or interventions in upstream and downstream signaling targets could assist in restoring actin cytoskeleton stability, thus impeding PCa development. The related ABPs in PCa and their members of the family are detailed in Desk 2.
8 Dialogue and prospect
PCa stands as one of the vital prevalent malignant ailments amongst males globally, posing important challenges like androgen deprivation resistance and bone metastasis in superior therapy levels. These challenges intricately tie again to the roles of ABPs in governing the actin cytoskeleton. Current analysis illustrates that ABPs not solely affect the cytoskeleton stability but in addition regulate androgen signaling pathways, thus modulating the migration, invasion, and proliferation of PCa cells. It’s essential to notice that ABPs exhibit various features quite than singular roles. Varied ABPs doubtless play distinct elements within the pathogenesis and development of PCa. As an illustration, cofilin acts in each severing actin filaments and governing actin polymerization and depolymerization. This text summarizes 14 documented ABPs linked to PCa, laying a basis for conceptualizing subsequent PCa therapy methods.
Nonetheless, this examine primarily focuses on identified ABPs related to PCa, with nonetheless restricted complete understanding relating to the particular mechanisms of interplay between ABPs and the actin cytoskeleton. Moreover, the molecular mechanisms of different ABPs not but reported in PCa stay obscure, which could constrain our holistic comprehension of PCa pathogenesis. Whereas some ABPs associated to PCa have been preliminarily explored, quite a few unanswered questions persist. Future investigations ought to delve deeper into the interplays between ABPs and the actin cytoskeleton, elucidating their exact roles in PCa pathogenesis and metastasis. Such in-depth insights will facilitate the event of focused therapy approaches directed at ABPs to extra successfully fight the present challenges in PCa administration.
Creator contributions
FF: Writing–unique draft, Writing–evaluate and modifying. YY: Writing–evaluate and modifying. BZ: Visualization, Writing–evaluate and modifying. YL: Visualization, Writing–evaluate and modifying. LW: Visualization, Writing–evaluate and modifying. JY: Visualization, Writing–evaluate and modifying. QZ: Conceptualization, Funding acquisition, Mission administration, Supervision, Writing–evaluate and modifying.
Funding
The creator(s) declare that monetary help was acquired for the analysis, authorship, and/or publication of this text. This analysis was funded by Joint Funds of the Nationwide Pure Science Basis of China (Grant No. U20A20408); Nationwide Pure Science Basis of China (Grant No. 82074450); the Self-discipline Development Mission “Jiebangguashuai” of Hunan College of Chinese language Drugs (Grant No. 22JBZ016).
Battle of curiosity
The authors declare that the analysis was carried out within the absence of any industrial or monetary relationships that could possibly be construed as a possible battle of curiosity.
Writer’s be aware
All claims expressed on this article are solely these of the authors and don’t essentially characterize these of their affiliated organizations, or these of the writer, the editors and the reviewers. Any product that could be evaluated on this article, or declare that could be made by its producer, isn’t assured or endorsed by the writer.
References
Ahmed, T., Shea, Ok., Masters, J. R. W., Jones, G. E., and Wells, C. M. (2008). A PAK4-LIMK1 pathway drives prostate most cancers cell migration downstream of HGF. Cell. Sign. 20 (7), 1320–1328. doi:10.1016/j.cellsig.2008.02.021
PubMed Summary | CrossRef Full Textual content | Google Scholar
Assinder, S., and Cole, N. (2011). Does TGF-β induced formation of actin stress fibres reinforce Smad dependent TGF-β signalling within the prostate? Med. Hypotheses 76 (6), 802–804. doi:10.1016/j.mehy.2011.02.021
PubMed Summary | CrossRef Full Textual content | Google Scholar
Assinder, S. J., Au, E., Dong, Q., and Winnick, C. (2010). A novel splice variant of the beta-tropomyosin (TPM2) gene in prostate most cancers. Mol. Carcinog. 49 (6), 525–531. doi:10.1002/mc.20626
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bai, W., Ren, J.-S., Xia, M., Zhao, Y., Ding, J.-J., Chen, X., et al. (2023). Concentrating on FSCN1 with an oral small-molecule inhibitor for treating ocular neovascularization. J. Transl. Med. 21 (1), 555. doi:10.1186/s12967-023-04225-0
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bedolla, R. G., Wang, Y., Asuncion, A., Chamie, Ok., Siddiqui, S., Mudryj, M. M., et al. (2009). Nuclear versus cytoplasmic localization of filamin A in prostate most cancers: immunohistochemical correlation with metastases. Clin. Most cancers Res. Official J. Am. Assoc. Most cancers Res. 15 (3), 788–796. doi:10.1158/1078-0432.CCR-08-1402
PubMed Summary | CrossRef Full Textual content | Google Scholar
Beghein, E., Devriese, D., Van Hoey, E., and Gettemans, J. (2018). Cortactin and fascin-1 regulate extracellular vesicle launch by controlling endosomal trafficking or invadopodia formation and performance. Sci. Rep. 8, 15606. doi:10.1038/s41598-018-33868-z
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bosutti, A., Dapas, B., Grassi, G., Bussani, R., Zanconati, F., Giudici, F., et al. (2022). Excessive eEF1A1 protein ranges mark aggressive prostate cancers and the in vitro concentrating on of eEF1A1 reveals the eEF1A1–actin advanced as a brand new potential goal for remedy. Int. J. Mol. Sci. 23 (8), 4143. doi:10.3390/ijms23084143
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bravo-Cordero, J. J., Magalhaes, M. A. O., Eddy, R. J., Hodgson, L., and Condeelis, J. (2013). Features of cofilin in cell locomotion and invasion. Nat. Rev. Mol. Cell Biol., 14 (7), 405–415. doi:10.1038/nrm3609
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bulut, G., Hong, S.-H., Chen, Ok., Beauchamp, E. M., Rahim, S., Kosturko, G. W., et al. (2012). Small molecule inhibitors of ezrin inhibit the invasive phenotype of osteosarcoma cells. Oncogene 31 (3), 269–281. doi:10.1038/onc.2011.245
PubMed Summary | CrossRef Full Textual content | Google Scholar
Burianek, L. E., and Soderling, S. H. (2013). Beneath lock and key: spatiotemporal regulation of WASP household proteins coordinates separate dynamic mobile processes. Seminars Cell and Dev. Biol. 24 (4), 258–266. doi:10.1016/j.semcdb.2012.12.005
PubMed Summary | CrossRef Full Textual content | Google Scholar
Cahuzac, M., Langlois, P., Péant, B., Fleury, H., Mes-Masson, A.-M., and Saad, F. (2022). Pre-activation of autophagy impacts response to olaparib in prostate most cancers cells. Commun. Biol. 5 (1), 251. doi:10.1038/s42003-022-03210-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
Cai, G., Wu, D., Wang, Z., Xu, Z., Wong, Ok.-B., Ng, C.-F., et al. (2017). Collapsin response mediator protein-1 (CRMP1) acts as an invasion and metastasis suppressor of prostate most cancers by way of its suppression of epithelial-mesenchymal transition and reworking of actin cytoskeleton group. Oncogene 36 (4), 546–558. doi:10.1038/onc.2016.227
PubMed Summary | CrossRef Full Textual content | Google Scholar
Castoria, G., Auricchio, F., and Migliaccio, A. (2017). Extranuclear companions of androgen receptor: on the crossroads of proliferation, migration, and neuritogenesis. FASEB J. Official Publ. Fed. Am. Soc. Exp. Biol. 31 (4), 1289–1300. doi:10.1096/fj.201601047R
PubMed Summary | CrossRef Full Textual content | Google Scholar
Castoria, G., D’Amato, L., Ciociola, A., Giovannelli, P., Giraldi, T., Sepe, L., et al. (2011). Androgen-induced cell migration: position of androgen receptor/filamin A affiliation. PloS One 6 (2), e17218. doi:10.1371/journal.pone.0017218
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chen, B., Zhang, C., Wang, Z., Chen, Y., Xie, H., Li, S., et al. (2019). Mechanistic insights into Nav1.7-dependent regulation of rat prostate most cancers cell invasiveness revealed by toxin probes and proteomic evaluation. FEBS J. 286 (13), 2549–2561. doi:10.1111/febs.14823
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chen, J., Huang, Y., Tang, Z., Li, M., Ling, X., Liao, J., et al. (2020a). Genome-scale CRISPR-cas9 transcriptional activation screening in metformin resistance associated gene of prostate most cancers. Entrance. Cell Dev. Biol. 8, 616332. doi:10.3389/fcell.2020.616332
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chen, L., Cai, J., Huang, Y., Tan, X., Guo, Q., Lin, X., et al. (2020b). Identification of cofilin-1 as a novel mediator for the metastatic potentials and chemoresistance of the prostate most cancers cells. Eur. J. Pharmacol. 880, 173100. doi:10.1016/j.ejphar.2020.173100
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chen, Z., Wang, J., Lu, Y., Lai, C., Qu, L., and Zhuo, Y. (2022). Ezrin expression in circulating tumor cells is a predictor of prostate most cancers metastasis. Bioengineered 13 (2), 4076–4084. doi:10.1080/21655979.2021.2014710
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chuan, Y.-C., Iglesias-Gato, D., Fernandez-Perez, L., Cedazo-Minguez, A., Pang, S.-T., Norstedt, G., et al. (2010). Ezrin mediates c-Myc actions in prostate most cancers cell invasion. Oncogene 29 (10), 1531–1542. doi:10.1038/onc.2009.442
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chuan, Y.-C., Pang, S.-T., Cedazo-Minguez, A., Norstedt, G., Pousette, A., and Flores-Morales, A. (2006). Androgen induction of prostate most cancers cell invasion is mediated by ezrin. J. Biol. Chem. 281 (40), 29938–29948. doi:10.1074/jbc.M602237200
PubMed Summary | CrossRef Full Textual content | Google Scholar
Coleman, R. E., Croucher, P. I., Padhani, A. R., Clézardin, P., Chow, E., Fallon, M., et al. (2020). Bone metastases. Nat. Rev. Dis. Prim. 6 (1), 83. doi:10.1038/s41572-020-00216-3
PubMed Summary | CrossRef Full Textual content | Google Scholar
Collazo, J., Zhu, B., Larkin, S., Martin, S. Ok., Pu, H., Horbinski, C., et al. (2014). Cofilin drives cell-invasive and metastatic responses to TGF-β in prostate most cancers. Most cancers Res. 74 (8), 2362–2373. doi:10.1158/0008-5472.CAN-13-3058
PubMed Summary | CrossRef Full Textual content | Google Scholar
Prepare dinner, A.-L. M., and Haynes, J. M. (2007). Phosphorylation of the PKG substrate, vasodilator-stimulated phosphoprotein (VASP), in human cultured prostatic stromal cells. Nitric Oxide Biol. Chem. 16 (1), 10–17. doi:10.1016/j.niox.2006.09.003
PubMed Summary | CrossRef Full Textual content | Google Scholar
Culig, Z., Steiner, H., Bartsch, G., and Hobisch, A. (2005). Mechanisms of endocrine therapy-responsive and -unresponsive prostate tumours. Endocrine-Associated Most cancers 12 (2), 229–244. doi:10.1677/erc.1.00775a
PubMed Summary | CrossRef Full Textual content | Google Scholar
Dai, Y., and Gao, X. (2021). Inhibition of most cancers cell-derived exosomal microRNA-183 suppresses cell progress and metastasis in prostate most cancers by upregulating TPM1. Most cancers Cell Int. 21 (1), 145. doi:10.1186/s12935-020-01686-x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Darnel, A. D., Behmoaram, E., Vollmer, R. T., Corcos, J., Bijian, Ok., Sircar, Ok., et al. (2009). Fascin regulates prostate most cancers cell invasion and is related to metastasis and biochemical failure in prostate most cancers. Clin. Most cancers Res. Official J. Am. Assoc. Most cancers Res. 15 (4), 1376–1383. doi:10.1158/1078-0432.CCR-08-1789
PubMed Summary | CrossRef Full Textual content | Google Scholar
Davila, M., Frost, A. R., Grizzle, W. E., and Chakrabarti, R. (2003). LIM kinase 1 is crucial for the invasive progress of prostate epithelial cells: implications in prostate most cancers. J. Biol. Chem. 278 (38), 36868–36875. doi:10.1074/jbc.M306196200
PubMed Summary | CrossRef Full Textual content | Google Scholar
Davila, M., Jhala, D., Ghosh, D., Grizzle, W. E., and Chakrabarti, R. (2007). Expression of LIM kinase 1 is related to reversible G1/S section arrest, chromosomal instability and prostate most cancers. Mol. Most cancers 6, 40. doi:10.1186/1476-4598-6-40
PubMed Summary | CrossRef Full Textual content | Google Scholar
Desai, B., Ma, T., and Chellaiah, M. A. (2008). Invadopodia and matrix degradation, a brand new property of prostate most cancers cells throughout migration and invasion. J. Biol. Chem. 283 (20), 13856–13866. doi:10.1074/jbc.M709401200
PubMed Summary | CrossRef Full Textual content | Google Scholar
Di Donato, M., Giovannelli, P., Barone, M. V., Auricchio, F., Castoria, G., and Migliaccio, A. (2021a). A small peptide concentrating on the ligand-induced androgen receptor/filamin a interplay inhibits the invasive phenotype of prostate most cancers cells. Cells 11 (1), 14. doi:10.3390/cells11010014
PubMed Summary | CrossRef Full Textual content | Google Scholar
Di Donato, M., Giovannelli, P., Cernera, G., Di Santi, A., Marino, I., Bilancio, A., et al. (2015). Non-genomic androgen motion regulates proliferative/migratory signaling in stromal cells. Entrance. Endocrinol. 5, 225. doi:10.3389/fendo.2014.00225
Di Donato, M., Zamagni, A., Galasso, G., Di Zazzo, E., Giovannelli, P., Barone, M. V., et al. (2021b). The androgen receptor/filamin A posh as a goal in prostate most cancers microenvironment. Cell Demise Dis. 12 (1), 127. doi:10.1038/s41419-021-03402-7
PubMed Summary | CrossRef Full Textual content | Google Scholar
Di Vizio, D., Kim, J., Hager, M. H., Morello, M., Yang, W., Lafargue, C. J., et al. (2009). Oncosome formation in prostate most cancers: affiliation with a area of frequent chromosomal deletion in metastatic illness. Most cancers Res. 69 (13), 5601–5609. doi:10.1158/0008-5472.CAN-08-3860
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fares, J., Fares, M. Y., Khachfe, H. H., Salhab, H. A., and Fares, Y. (2020). Molecular ideas of metastasis: an indicator of most cancers revisited. Sign Transduct. Goal. Ther. 5, 28. doi:10.1038/s41392-020-0134-x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Feng, Y., Huang, Z., Lu, F., Tune, L., Liu, R., Zhang, Y., et al. (2024). 8-Br-cGMP prompts HSPB6 and will increase the antineoplastic exercise of quinidine in prostate most cancers. Cell Demise Discov. 10 (1), 90. doi:10.1038/s41420-024-01853-3
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fernando, H. S., Sanders, A. J., Kynaston, H. G., and Jiang, W. G. (2008). WAVE1 is related to invasiveness and progress of prostate most cancers cells. J. Urology 180 (4), 1515–1521. doi:10.1016/j.juro.2008.06.004
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fernando, H. S., Sanders, A. J., Kynaston, H. G., and Jiang, W. G. (2010). WAVE3 is related to invasiveness in prostate most cancers cells. Urol. Oncol. 28 (3), 320–327. doi:10.1016/j.urolonc.2008.12.022
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fu, Y.-M., Yu, Z.-X., Lin, H., Fu, X., and Meadows, G. G. (2008). Selective amino acid restriction differentially impacts the motility and directionality of DU145 and PC3 prostate most cancers cells. J. Cell. Physiology 217 (1), 184–193. doi:10.1002/jcp.21490
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fuse, M., Nohata, N., Kojima, S., Sakamoto, S., Chiyomaru, T., Kawakami, Ok., et al. (2011). Restoration of miR-145 expression suppresses cell proliferation, migration and invasion in prostate most cancers by concentrating on FSCN1. Int. J. Oncol. 38 (4), 1093–1101. doi:10.3892/ijo.2011.919
PubMed Summary | CrossRef Full Textual content | Google Scholar
Gai, W.-T., Yu, D.-P., Wang, X.-S., and Wang, P.-T. (2016). Anti-cancer impact of ursolic acid prompts apoptosis by way of ROCK/PTEN mediated mitochondrial translocation of cofilin-1 in prostate most cancers. Oncol. Lett. 12 (4), 2880–2885. doi:10.3892/ol.2016.5015
PubMed Summary | CrossRef Full Textual content | Google Scholar
Gamallat, Y., Zaaluk, H., Kish, E. Ok., Abdelsalam, R., Liosis, Ok., Ghosh, S., et al. (2022). ARPC1B is related to deadly prostate most cancers and its inhibition decreases cell invasion and migration in vitro. Int. J. Mol. Sci. 23 (3), 1476. doi:10.3390/ijms23031476
PubMed Summary | CrossRef Full Textual content | Google Scholar
Giri, A., Bajpai, S., Trenton, N., Jayatilaka, H., Longmore, G. D., and Wirtz, D. (2013). The Arp2/3 advanced mediates multigeneration dendritic protrusions for environment friendly three-dimensional most cancers cell migration. FASEB J. Official Publ. Fed. Am. Soc. Exp. Biol. 27 (10), 4089–4099. doi:10.1096/fj.12-224352
PubMed Summary | CrossRef Full Textual content | Google Scholar
Griner, N. B., Younger, D., Chaudhary, P., Mohamed, A. A., Huang, W., Chen, Y., et al. (2015). ERG oncoprotein inhibits ANXA2 expression and performance in prostate most cancers. Mol. Most cancers Res. MCR 13 (2), 368–379. doi:10.1158/1541-7786.MCR-14-0275-T
PubMed Summary | CrossRef Full Textual content | Google Scholar
Harrison, G. M., Davies, G., Martin, T. A., Jiang, W. G., and Mason, M. D. (2002). Distribution and expression of CD44 isoforms and Ezrin throughout prostate cancer-endothelium interplay. Int. J. Oncol. 21 (5), 935–940. doi:10.3892/ijo.21.5.935
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hasegawa, Y., Murph, M., Yu, S., Tigyi, G., and Mills, G. B. (2008). Lysophosphatidic acid (LPA)-induced vasodilator-stimulated phosphoprotein mediates lamellipodia formation to provoke motility in PC-3 prostate most cancers cells. Mol. Oncol. 2 (1), 54–69. doi:10.1016/j.molonc.2008.03.009
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hashimoto, Ok., Kyoda, Y., Tanaka, T., Maeda, T., Kobayashi, Ok., Uchida, Ok., et al. (2015). The potential of neurotensin secreted from neuroendocrine tumor cells to advertise gelsolin-mediated invasiveness of prostate adenocarcinoma cells. Laboratory Investigation; a J. Tech. Strategies Pathology 95 (3), 283–295. doi:10.1038/labinvest.2014.165
PubMed Summary | CrossRef Full Textual content | Google Scholar
He, C., Chen, X., Chen, Y., Solar, J., Qi, M., Rocha, S., et al. (2024). International acetylome profiling signifies EPA impedes however OA promotes prostate most cancers motility by way of altered acetylation of PFN1 and FLNA. Proteomics, e2300393. doi:10.1002/pmic.202300393
PubMed Summary | CrossRef Full Textual content | Google Scholar
He, G., Li, M., Fang, L., Xu, L., Huang, X., Zheng, L., et al. (2020). N-Myc induces the tumor development of prostate most cancers by regulating FSCN1. Oncol. Rep. 44 (5), 2265–2274. doi:10.3892/or.2020.7775
PubMed Summary | CrossRef Full Textual content | Google Scholar
He, X., Gao, Q., Qiang, Y., Guo, W., and Ma, Y. (2017). Cucurbitacin E induces apoptosis of human prostate most cancers cells by way of cofilin-1 and mTORC1. Oncol. Lett. 13 (6), 4905–4910. doi:10.3892/ol.2017.6086
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hebbrecht, T., Van Audenhove, I., Zwaenepoel, O., Verhelle, A., and Gettemans, J. (2017). VCA nanobodies goal N-WASp to cut back invadopodium formation and functioning. PloS One 12 (9), e0185076. doi:10.1371/journal.pone.0185076
PubMed Summary | CrossRef Full Textual content | Google Scholar
Herrlich, P., Morrison, H., Sleeman, J., Orian-Rousseau, V., König, H., Weg-Remers, S., et al. (2000). CD44 acts each as a growth- and invasiveness-promoting molecule and as a tumor-suppressing cofactor. Ann. N. Y. Acad. Sci. 910, 106–118. ; dialogue 118-120. doi:10.1111/j.1749-6632.2000.tb06704.x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hirokawa, Y. S., Kanayama, Ok., Kagaya, M., Shimojo, N., Uchida, Ok., Imai, H., et al. (2020). SOX11-induced lower in vimentin and a rise in prostate most cancers cell migration attributed to cofilin exercise. Exp. Mol. Pathology 117, 104542. doi:10.1016/j.yexmp.2020.104542
PubMed Summary | CrossRef Full Textual content | Google Scholar
Huang, F., Wang, P., and Wang, X. (2018). Thapsigargin induces apoptosis of prostate most cancers by way of cofilin-1 and paxillin. Oncol. Lett. 16 (2), 1975–1980. doi:10.3892/ol.2018.8833
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ishizuya, Y., Uemura, M., Narumi, R., Tomiyama, E., Koh, Y., Matsushita, M., et al. (2020). The position of actinin-4 (ACTN4) in exosomes as a possible novel therapeutic goal in castration-resistant prostate most cancers. Biochem. Biophysical Res. Commun. 523 (3), 588–594. doi:10.1016/j.bbrc.2019.12.084
PubMed Summary | CrossRef Full Textual content | Google Scholar
Izdebska, M., Zielińska, W., Hałas-Wiśniewska, M., and Grzanka, A. (2020). Involvement of actin and actin-binding proteins in carcinogenesis. Cells 9 (10), 2245. doi:10.3390/cells9102245
PubMed Summary | CrossRef Full Textual content | Google Scholar
Jensen, C. C., Clements, A. N., Liou, H., Ball, L. E., Bethard, J. R., Langlais, P. R., et al. (2023). PIM1 phosphorylates ABI2 to reinforce actin dynamics and promote tumor invasion. J. Cell Biol. 222 (6), e202208136. doi:10.1083/jcb.202208136
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ji, J., Li, H., Wang, W., Yuan, B., and Shen, T. (2022). ARPC1A is regulated by STAT3 to inhibit ferroptosis and promote prostate most cancers development. Hum. Cell 35 (5), 1591–1601. doi:10.1007/s13577-022-00754-w
PubMed Summary | CrossRef Full Textual content | Google Scholar
Jin, X., Wang, J., Gao, Ok., Zhang, P., Yao, L., Tang, Y., et al. (2017). Dysregulation of INF2-mediated mitochondrial fission in SPOP-mutated prostate most cancers. PLoS Genet. 13 (4), e1006748. doi:10.1371/journal.pgen.1006748
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kadzik, R. S., Homa, Ok. E., and Kovar, D. R. (2020). F-actin cytoskeleton community self-organization by way of competitors and cooperation. Annu. Rev. Cell Dev. Biol. 36, 35–60. doi:10.1146/annurev-cellbio-032320-094706
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kiełb, P., Kowalczyk, Ok., Gurwin, A., Nowak, Ł., Krajewski, W., Sosnowski, R., et al. (2023). Novel histopathological biomarkers in prostate most cancers: implications and views. Biomedicines 11 (6), 1552. doi:10.3390/biomedicines11061552
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kim, J., Morley, S., Le, M., Bedoret, D., Umetsu, D. T., Di Vizio, D., et al. (2014). Enhanced shedding of extracellular vesicles from amoeboid prostate most cancers cells: potential results on the tumor microenvironment. Most cancers Biol. Ther. 15 (4), 409–418. doi:10.4161/cbt.27627
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kmeťová Sivoňová, M., Tatarková, Z., Jurečeková, J., Kliment, J., Híveš, M., Lichardusová, L., et al. (2021). Differential profiling of prostate tumors versus benign prostatic tissues by utilizing a 2DE-MALDI-TOF-based proteomic method. Neoplasma 68 (1), 154–164. doi:10.4149/neo_2020_200611N625
PubMed Summary | CrossRef Full Textual content | Google Scholar
Knerr, J., Werner, R., Schwan, C., Wang, H., Gebhardt, P., Grötsch, H., et al. (2023). Formin-mediated nuclear actin at androgen receptors promotes transcription. Nature 617 (7961), 616–622. Article 7961. doi:10.1038/s41586-023-05981-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kryza, T., Silva, L. M., Bock, N., Fuhrman-Luck, R. A., Stephens, C. R., Gao, J., et al. (2017). Kallikrein-related peptidase 4 induces cancer-associated fibroblast options in prostate-derived stromal cells. Mol. Oncol. 11 (10), 1307–1329. doi:10.1002/1878-0261.12075
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lampe, J. B., Desai, P. P., Tripathi, A. Ok., Sabnis, N. A., Chen, Z., Ranjan, A. P., et al. (2023). Cabazitaxel-loaded nanoparticles scale back the invasiveness in metastatic prostate most cancers cells: past the classical taxane operate. Pharmaceutics 15 (2), 662. doi:10.3390/pharmaceutics15020662
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lawson, C. D., Peel, S., Jayo, A., Corrigan, A., Iyer, P., Baxter Dalrymple, M., et al. (2022). Nuclear fascin regulates most cancers cell survival. eLife 11, e79283. doi:10.7554/eLife.79283
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lee, M.-H., Kundu, J. Ok., Chae, J.-I., and Shim, J.-H. (2019). Concentrating on ROCK/LIMK/cofilin signaling pathway in most cancers. Archives Pharmacal Res. 42 (6), 481–491. doi:10.1007/s12272-019-01153-w
PubMed Summary | CrossRef Full Textual content | Google Scholar
Leitão, C., Matos, B., Roque, F., Herdeiro, M. T., and Fardilha, M. (2022). The affect of way of life on prostate most cancers: a street to the invention of latest biomarkers. J. Clin. Med. 11 (10), 2925. doi:10.3390/jcm11102925
PubMed Summary | CrossRef Full Textual content | Google Scholar
Li, P., Wang, Z., Li, S., and Wang, L. (2021). Circ_0006404 accelerates prostate most cancers development by way of regulating miR-1299/CFL2 signaling. OncoTargets Ther. 14, 83–95. doi:10.2147/OTT.S277831
Li, T., Hong, X., Zhao, J., Teng, Y., Zheng, J., Chen, H., et al. (2016). Gelsolin-like actin-capping protein is related to affected person prognosis, mobile apoptosis and proliferation in prostate most cancers. Biomarkers Med. 10 (12), 1251–1260. doi:10.2217/bmm-2016-0186
PubMed Summary | CrossRef Full Textual content | Google Scholar
Li, X., Han, X., Wei, P., Yang, J., and Solar, J. (2020). Knockdown of lncRNA CCAT1 enhances sensitivity of paclitaxel in prostate most cancers by way of regulating miR-24-3p and FSCN1. Most cancers Biol. Ther. 21 (5), 452–462. doi:10.1080/15384047.2020.1727700
PubMed Summary | CrossRef Full Textual content | Google Scholar
Li, Z., Shi, J., Zhang, N., Zheng, X., Jin, Y., Wen, S., et al. (2022). FSCN1 acts as a promising therapeutic goal within the blockade of tumor cell motility: a evaluate of its operate, mechanism, and scientific significance. J. Most cancers 13 (8), 2528–2539. doi:10.7150/jca.67977
PubMed Summary | CrossRef Full Textual content | Google Scholar
Li, Z., Tao, Y., Gao, Z., Peng, S., Lai, Y., Li, Ok., et al. (2023). SYTL2 promotes metastasis of prostate most cancers cells by enhancing FSCN1-mediated pseudopodia formation and invasion. J. Transl. Med. 21, 303. doi:10.1186/s12967-023-04146-y
PubMed Summary | CrossRef Full Textual content | Google Scholar
Liu, H., Zhang, Y., Li, L., Cao, J., Guo, Y., Wu, Y., et al. (2021). Fascin actin-bundling protein 1 in human most cancers: promising biomarker or therapeutic goal? Mol. Ther. Oncolytics 20, 240–264. doi:10.1016/j.omto.2020.12.014
PubMed Summary | CrossRef Full Textual content | Google Scholar
Liu, T., Mendes, D. E., and Berkman, C. E. (2014). Extended androgen deprivation results in overexpression of calpain 2: implications for prostate most cancers development. Int. J. Oncol. 44 (2), 467–472. doi:10.3892/ijo.2013.2196
PubMed Summary | CrossRef Full Textual content | Google Scholar
Loy, C. J., Sim, Ok. S., and Yong, E. L. (2003). Filamin-A fraction localizes to the nucleus to control androgen receptor and coactivator features. Proc. Natl. Acad. Sci. U. S. A. 100 (8), 4562–4567. doi:10.1073/pnas.0736237100
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lu, L. I., Fu, N. I., Luo, X. U., Li, X.-Y., and Li, X.-P. (2015). Overexpression of cofilin 1 in prostate most cancers and the corresponding scientific implications. Oncol. Lett. 9 (6), 2757–2761. doi:10.3892/ol.2015.3133
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ma, S. C., Chen, R., Yang, T. N., Zhang, F., and Zhao, D. (2020). Baicalein inhibits the proliferative exercise of human prostate most cancers cell line PC3 by downregulating Ezrin. J. Biol. Regul. Homeost. Brokers 34 (3), 885–892. doi:10.23812/20-145-A-44
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mahaveer Chand, N., Tekumalla, P. Ok., Rosenberg, M. T., Dobi, A., Ali, A., Miller, G. M., et al. (2024). Filamin A is a prognostic serum biomarker for differentiating benign prostatic hyperplasia from prostate most cancers in caucasian and african American males. Cancers 16 (4), 712. doi:10.3390/cancers16040712
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mardilovich, Ok., Gabrielsen, M., McGarry, L., Orange, C., Patel, R., Shanks, E., et al. (2015). Elevated LIM kinase 1 in nonmetastatic prostate most cancers displays its position in facilitating androgen receptor nuclear translocation. Mol. Most cancers Ther. 14 (1), 246–258. doi:10.1158/1535-7163.MCT-14-0447
PubMed Summary | CrossRef Full Textual content | Google Scholar
McGrath, M. J., Binge, L. C., Sriratana, A., Wang, H., Robinson, P. A., Pook, D., et al. (2013). Regulation of the transcriptional coactivator FHL2 licenses activation of the androgen receptor in castrate-resistant prostate most cancers. Most cancers Res. 73 (16), 5066–5079. doi:10.1158/0008-5472.CAN-12-4520
PubMed Summary | CrossRef Full Textual content | Google Scholar
Merino, F., Pospich, S., and Raunser, S. (2020). In the direction of a structural understanding of the transforming of the actin cytoskeleton. Seminars Cell and Dev. Biol. 102, 51–64. doi:10.1016/j.semcdb.2019.11.018
PubMed Summary | CrossRef Full Textual content | Google Scholar
Miller, Ok. D., Nogueira, L., Devasia, T., Mariotto, A. B., Yabroff, Ok. R., Jemal, A., et al. (2022). Most cancers therapy and survivorship statistics, 2022. CA A Most cancers J. Clin. 72 (5), 409–436. doi:10.3322/caac.21731
Misra, U. Ok., Deedwania, R., and Pizzo, S. V. (2005). Binding of activated alpha2-macroglobulin to its cell floor receptor GRP78 in 1-LN prostate most cancers cells regulates PAK-2-dependent activation of LIMK. J. Biol. Chem. 280 (28), 26278–26286. doi:10.1074/jbc.M414467200
PubMed Summary | CrossRef Full Textual content | Google Scholar
Moazzam, M., Ye, L., Solar, P.-H., Kynaston, H., and Jiang, W. G. (2015). Knockdown of WAVE3 impairs HGF induced migration and invasion of prostate most cancers cells. Most cancers Cell Int. 15, 51. doi:10.1186/s12935-015-0203-3
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mooso, B. A., Vinall, R. L., Tepper, C. G., Savoy, R. M., Cheung, J. P., Singh, S., et al. (2012). Enhancing the effectiveness of androgen deprivation in prostate most cancers by inducing Filamin A nuclear localization. Endocrine-Associated Most cancers 19 (6), 759–777. doi:10.1530/ERC-12-0171
PubMed Summary | CrossRef Full Textual content | Google Scholar
Moretti, R. M., Mai, S., Montagnani Marelli, M., Rizzi, F., Bettuzzi, S., and Limonta, P. (2011). Molecular mechanisms of the antimetastatic exercise of nuclear clusterin in prostate most cancers cells. Int. J. Oncol. 39 (1), 225–234. doi:10.3892/ijo.2011.1030
PubMed Summary | CrossRef Full Textual content | Google Scholar
Morley, S., Hager, M. H., Pollan, S. G., Knudsen, B., Di Vizio, D., and Freeman, M. R. (2014). Buying and selling in your spindles for blebs: the amoeboid tumor cell phenotype in prostate most cancers. Asian J. Androl. 16 (4), 530–535. doi:10.4103/1008-682X.122877
PubMed Summary | CrossRef Full Textual content | Google Scholar
Morley, S., You, S., Pollan, S., Choi, J., Zhou, B., Hager, M. H., et al. (2015). Regulation of microtubule dynamics by DIAPH3 influences amoeboid tumor cell mechanics and sensitivity to taxanes. Sci. Rep. 5, 12136. doi:10.1038/srep12136
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mu, D., Zhou, G., Li, J., Su, B., and Guo, H. (2018). Ursolic acid prompts the apoptosis of prostate most cancers by way of ROCK/PTEN mediated mitochondrial translocation of cofilin-1. Oncol. Lett. 15 (3), 3202–3206. doi:10.3892/ol.2017.7689
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mughees, M., Bano, F., and Wajid, S. (2021). Mechanism of WASP and WAVE household proteins within the development of prostate most cancers. Protoplasma 258 (4), 683–693. doi:10.1007/s00709-021-01608-2
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ngalame, N. N. O., Makia, N. L., Waalkes, M. P., and Tokar, E. J. (2016). Mitigation of arsenic-induced acquired most cancers phenotype in prostate most cancers stem cells by miR-143 restoration. Toxicol. Appl. Pharmacol. 312, 11–18. doi:10.1016/j.taap.2015.12.013
PubMed Summary | CrossRef Full Textual content | Google Scholar
Nishimura, Ok., Ting, H.-J., Harada, Y., Tokizane, T., Nonomura, N., Kang, H.-Y., et al. (2003). Modulation of androgen receptor transactivation by gelsolin: a newly recognized androgen receptor coregulator. Most cancers Res. 63 (16), 4888–4894.
Obinata, D., Funakoshi, D., Takayama, Ok., Hara, M., Niranjan, B., Teng, L., et al. (2022). OCT1-target neural gene PFN2 promotes tumor progress in androgen receptor-negative prostate most cancers. Sci. Rep. 12 (1), 6094. doi:10.1038/s41598-022-10099-x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Oelrich, F., Junker, H., Stope, M. B., Erb, H. H. H., Walther, R., Venz, S., et al. (2021). Gelsolin governs the neuroendocrine transdifferentiation of prostate most cancers cells and suppresses the apoptotic equipment. Anticancer Res. 41 (8), 3717–3729. doi:10.21873/anticanres.15163
PubMed Summary | CrossRef Full Textual content | Google Scholar
Paller, C., Pu, H., Begemann, D. E., Wade, C. A., Hensley, P. J., and Kyprianou, N. (2019). TGF-β receptor I inhibitor enhances response to enzalutamide in a pre-clinical mannequin of superior prostate most cancers. Prostate 79 (1), 31–43. doi:10.1002/professionals.23708
PubMed Summary | CrossRef Full Textual content | Google Scholar
Panigrahi, G. Ok., Praharaj, P. P., Kittaka, H., Mridha, A. R., Black, O. M., Singh, R., et al. (2019). Exosome proteomic analyses determine inflammatory phenotype and novel biomarkers in African American prostate most cancers sufferers. Most cancers Med. 8 (3), 1110–1123. doi:10.1002/cam4.1885
PubMed Summary | CrossRef Full Textual content | Google Scholar
Park, S., Kang, M., Kim, S., An, H.-T., Gettemans, J., and Ko, J. (2020). α-Actinin-4 promotes the development of prostate most cancers by way of the akt/GSK-3β/β-catenin signaling pathway. Entrance. Cell Dev. Biol. 8, 588544. doi:10.3389/fcell.2020.588544
PubMed Summary | CrossRef Full Textual content | Google Scholar
Pečar, F. U., Jevnikar, Z., Rojnik, M., Doljak, B., Fonović, M., Jamnik, P., et al. (2013). Profilin 1 as a goal for cathepsin X exercise in tumor cells. PLoS ONE 8 (1), e53918. doi:10.1371/journal.pone.0053918
PubMed Summary | CrossRef Full Textual content | Google Scholar
Pečar, F. U., and Kos, J. (2015). Cathepsin X cleaves Profilin 1 C-terminal Tyr139 and influences clathrin-mediated endocytosis. PloS One 10 (9), e0137217. doi:10.1371/journal.pone.0137217
PubMed Summary | CrossRef Full Textual content | Google Scholar
Pérez-Martínez, F. C., Carrión, B., Lucío, M. I., Rubio, N., Herrero, M. A., Vázquez, E., et al. (2012). Enhanced docetaxel-mediated cytotoxicity in human prostate most cancers cells by way of knockdown of cofilin-1 by carbon nanohorn delivered siRNA. Biomaterials 33 (32), 8152–8159. doi:10.1016/j.biomaterials.2012.07.038
PubMed Summary | CrossRef Full Textual content | Google Scholar
Peterson, J. R., Lokey, R. S., Mitchison, T. J., and Kirschner, M. W. (2001). A chemical inhibitor of N-WASP reveals a brand new mechanism for concentrating on protein interactions. Proc. Natl. Acad. Sci. U. S. A. 98 (19), 10624–10629. doi:10.1073/pnas.201393198
PubMed Summary | CrossRef Full Textual content | Google Scholar
Prasad, P. D., Stanton, J.-A. L., and Assinder, S. J. (2010). Expression of the actin-associated protein transgelin (SM22) is decreased in prostate most cancers. Cell Tissue Res. 339 (2), 337–347. doi:10.1007/s00441-009-0902-y
PubMed Summary | CrossRef Full Textual content | Google Scholar
Qian, L., Wang, Y., Xiong, Y., Ren, H., Liu, S., Chen, D., et al. (2023). LINC01002 targets miR-650/FLNA pathway to suppress prostate most cancers development. Urol. Int. 107 (5), 526–538. doi:10.1159/000529947
PubMed Summary | CrossRef Full Textual content | Google Scholar
Qie, Y., Wang, L., Du, E., Chen, S., Lu, C., Ding, N., et al. (2020). TACC3 promotes prostate most cancers cell proliferation and restrains main cilium formation. Exp. Cell Res. 390 (2), 111952. doi:10.1016/j.yexcr.2020.111952
PubMed Summary | CrossRef Full Textual content | Google Scholar
Qu, G., Zhang, Y., Duan, H., Tang, C., Yang, G., Chen, D., et al. (2023). ARPC5 is transcriptionally activated by KLF4, and promotes cell migration and invasion in prostate most cancers by way of up-regulating ADAM17: ARPC5 serves as an oncogene in prostate most cancers. Apoptosis 28 (5), 783–795. doi:10.1007/s10495-023-01827-3
PubMed Summary | CrossRef Full Textual content | Google Scholar
Rana, P. S., Alkrekshi, A., Wang, W., Markovic, V., and Sossey-Alaoui, Ok. (2021). The position of WAVE2 signaling in most cancers. Biomedicines 9 (9), 1217. doi:10.3390/biomedicines9091217
PubMed Summary | CrossRef Full Textual content | Google Scholar
Reggi, E., Kaiser, S., Sahnane, N., Uccella, S., La Rosa, S., and Diviani, D. (2024). AKAP2-anchored protein phosphatase 1 controls prostatic neuroendocrine carcinoma cell migration and invasion. Biochimica Biophysica Acta (BBA) – Mol. Foundation Dis. 1870 (1), 166916. doi:10.1016/j.bbadis.2023.166916
Rehman, I., Evans, C. A., Glen, A., Cross, S. S., Eaton, C. L., Down, J., et al. (2012). iTRAQ identification of candidate serum biomarkers related to metastatic development of human prostate most cancers. PloS One 7 (2), e30885. doi:10.1371/journal.pone.0030885
PubMed Summary | CrossRef Full Textual content | Google Scholar
Reis-Sobreiro, M., Chen, J.-F., Novitskaya, T., You, S., Morley, S., Steadman, Ok., et al. (2018). Emerin deregulation hyperlinks nuclear form instability to metastatic potential. Most cancers Res. 78 (21), 6086–6097. doi:10.1158/0008-5472.CAN-18-0608
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ren, S., Ouyang, D.-Y., Saltis, M., Xu, L.-H., Zha, Q.-B., Cai, J.-Y., et al. (2012). Anti-proliferative impact of 23,24-dihydrocucurbitacin F on human prostate most cancers cells by way of induction of actin aggregation and cofilin-actin rod formation. Most cancers Chemother. Pharmacol. 70 (3), 415–424. doi:10.1007/s00280-012-1921-z
PubMed Summary | CrossRef Full Textual content | Google Scholar
Salimi, R., Bandaru, S., Devarakonda, S., Gökalp, S., Ala, C., Alvandian, A., et al. (2018). Blocking the cleavage of filamin A by calpain inhibitor decreases tumor cell progress. Anticancer Res. 38 (4), 2079–2085. doi:10.21873/anticanres.12447
PubMed Summary | CrossRef Full Textual content | Google Scholar
Santio, N. M., Vainio, V., Hoikkala, T., Mung, Ok. L., Lång, M., Vahakoski, R., et al. (2020). PIM1 accelerates prostate most cancers cell motility by phosphorylating actin capping proteins. Cell Commun. Sign. CCS 18, 121. doi:10.1186/s12964-020-00618-6
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sarantelli, E., Mourkakis, A., Zacharia, L. C., Stylianou, A., and Gkretsi, V. (2023). Fascin-1 in most cancers cell metastasis: outdated target-new insights. Int. J. Mol. Sci. 24 (14), 11253. doi:10.3390/ijms241411253
PubMed Summary | CrossRef Full Textual content | Google Scholar
Saurav, S., and Manna, S. Ok. (2022). Elevated expression of Profilin potentiates chemotherapeutic agent-mediated tumour regression. Br. J. Most cancers 126 (10), 1410–1420. doi:10.1038/s41416-021-01683-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
Savoy, R. M., Chen, L., Siddiqui, S., Melgoza, F. U., Durbin-Johnson, B., Drake, C., et al. (2015). Transcription of Nrdp1 by the androgen receptor is regulated by nuclear Filamin A in prostate most cancers. Endocrine-Associated Most cancers 22 (3), 369–386. doi:10.1530/ERC-15-0021
PubMed Summary | CrossRef Full Textual content | Google Scholar
Scaggiante, B., Dapas, B., Bonin, S., Grassi, M., Zennaro, C., Farra, R., et al. (2012). Dissecting the expression of EEF1A1/2 genes in human prostate most cancers cells: the potential of EEF1A2 as an indicator for prostate transformation and development. Br. J. Most cancers 106 (1), 166–173. doi:10.1038/bjc.2011.500
PubMed Summary | CrossRef Full Textual content | Google Scholar
Scotto, D. C. F., Russo, S., Muyas, F., Mangini, M., Garribba, L., Pazzaglia, L., et al. (2023). Profilin 1 deficiency drives mitotic defects and reduces genome stability. Commun. Biol. 6, 9. doi:10.1038/s42003-022-04392-8
PubMed Summary | CrossRef Full Textual content | Google Scholar
Shao, Q.-Q., Zhang, T.-P., Zhao, W.-J., Liu, Z.-W., You, L., Zhou, L., et al. (2016). Filamin A: insights into its actual position in cancers. Pathology Oncol. Res. POR 22 (2), 245–252. doi:10.1007/s12253-015-9980-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
Shishkin, S., Eremina, L., Pashintseva, N., Kovalev, L., and Kovaleva, M. (2017). Cofilin-1 and different ADF/cofilin superfamily members in human malignant cells. Int. J. Mol. Sci. 18 (1), 10. doi:10.3390/ijms18010010
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sizemore, S., Cicek, M., Sizemore, N., Ng, Ok. P., and Casey, G. (2007). Podocalyxin will increase the aggressive phenotype of breast and prostate most cancers cells in vitro by way of its interplay with ezrin. Most cancers Res. 67 (13), 6183–6191. doi:10.1158/0008-5472.CAN-06-3575
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sousa-Squiavinato, A. C. M., and Morgado-Díaz, J. A. (2024). A glimpse into cofilin-1 position in most cancers remedy: a possible goal to enhance scientific outcomes? Biochimica Biophysica Acta (BBA) – Rev. Most cancers 1879 (2), 189087. doi:10.1016/j.bbcan.2024.189087
Stark, T. W., Hensley, P. J., Spear, A., Pu, H., Strup, S. S., and Kyprianou, N. (2017). Predictive worth of epithelial-mesenchymal-transition (EMT) signature and PARP-1 in prostate most cancers radioresistance. Prostate 77 (16), 1583–1591. doi:10.1002/professionals.23435
PubMed Summary | CrossRef Full Textual content | Google Scholar
Su, Z., Wang, G., and Li, L. (2023). CHRDL1, NEFH, TAGLN and SYNM as novel diagnostic biomarkers of benign prostatic hyperplasia and prostate most cancers. Most cancers Biomarkers Sect. A Dis. Markers 38 (2), 143–159. doi:10.3233/CBM-230028
Sundram, V., Chauhan, S. C., Ebeling, M., and Jaggi, M. (2012). Curcumin attenuates β-catenin signaling in prostate most cancers cells by way of activation of protein kinase D1. PloS One 7 (4), e35368. doi:10.1371/journal.pone.0035368
PubMed Summary | CrossRef Full Textual content | Google Scholar
Turpin, A., Delliaux, C., Guardian, P., Chevalier, H., Escudero-Iriarte, C., Bonardi, F., et al. (2023). Fascin-1 expression is related to neuroendocrine prostate most cancers and immediately suppressed by androgen receptor. Br. J. Most cancers 129 (12), 1903–1914. doi:10.1038/s41416-023-02449-x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Untergasser, G., Gander, R., Lilg, C., Lepperdinger, G., Plas, E., and Berger, P. (2005). Profiling molecular targets of TGF-beta1 in prostate fibroblast-to-myofibroblast transdifferentiation. Mech. Ageing Dev. 126 (1), 59–69. doi:10.1016/j.mad.2004.09.023
PubMed Summary | CrossRef Full Textual content | Google Scholar
Vakhrusheva, A. V., Murashko, A. V., Trifonova, E. S., Efremov, Y. M., Timashev, P. S., and Sokolova, O. S. (2022). Function of actin-binding proteins within the regulation of mobile mechanics. Eur. J. Cell Biol. 101 (3), 151241. doi:10.1016/j.ejcb.2022.151241
PubMed Summary | CrossRef Full Textual content | Google Scholar
Valdman, A., Fang, X., Pang, S.-T., Nilsson, B., Ekman, P., and Egevad, L. (2005). Ezrin expression in prostate most cancers and benign prostatic tissue. Eur. Urol. 48 (5), 852–857. doi:10.1016/j.eururo.2005.03.013
PubMed Summary | CrossRef Full Textual content | Google Scholar
Van Audenhove, I., Boucherie, C., Pieters, L., Zwaenepoel, O., Vanloo, B., Martens, E., et al. (2014). Stratifying fascin and cortactin operate in invadopodium formation utilizing inhibitory nanobodies and focused subcellular delocalization. FASEB J. Official Publ. Fed. Am. Soc. Exp. Biol. 28 (4), 1805–1818. doi:10.1096/fj.13-242537
Varisli, L. (2013). Identification of latest genes downregulated in prostate most cancers and investigation of their results on prognosis. Genet. Check. Mol. Biomarkers 17 (7), 562–566. doi:10.1089/gtmb.2012.0524
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wang, Y., Kreisberg, J. I., Bedolla, R. G., Mikhailova, M., deVere White, R. W., and Ghosh, P. M. (2007). A 90 kDa fragment of filamin A promotes Casodex-induced progress inhibition in Casodex-resistant androgen receptor constructive C4-2 prostate most cancers cells. Oncogene 26 (41), 6061–6070. doi:10.1038/sj.onc.1210435
PubMed Summary | CrossRef Full Textual content | Google Scholar
Warner, H., Franciosa, G., van der Borg, G., Coenen, B., Faas, F., Koenig, C., et al. (2024). Atypical cofilin signaling drives dendritic cell migration by way of the extracellular matrix by way of nuclear deformation. Cell Rep. 43 (3), 113866. doi:10.1016/j.celrep.2024.113866
PubMed Summary | CrossRef Full Textual content | Google Scholar
Weeks, H. P., Sanders, A. J., Kynaston, H. G., and Jiang, W. G. (2016). The affiliation between WAVE1 and -3 and the ARP2/3 advanced in PC 3 cells. Anticancer Res. 36 (3), 1135–1142.
Weibo, X., Junkai, C., Xinyi, D., and Junqing, H. (2017). Lengthy non-coding RNA PCAT-1 contributes to tumorigenesis by regulating FSCN1 by way of miR-145-5p in prostate most cancers. Biomed. Pharmacother. = Biomedecine Pharmacother. 95, 1112–1118. doi:10.1016/j.biopha.2017.09.019
Wen, F., Solar, X., Solar, C., Dong, Z., Jia, G., Bao, W., et al. (2021). TAGLN is downregulated by TRAF6-mediated proteasomal degradation in prostate most cancers cells. Mol. Most cancers Res. 19 (7), 1113–1122. doi:10.1158/1541-7786.MCR-20-0513
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wo, Q., Zhang, D., Hu, L., Lyu, J., Xiang, F., Zheng, W., et al. (2019). Lengthy noncoding RNA SOX2-OT facilitates prostate most cancers cell proliferation and migration by way of miR-369-3p/CFL2 axis. Biochem. Biophysical Res. Commun. 520 (3), 586–593. doi:10.1016/j.bbrc.2019.09.108
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wong, S. Y., Haack, H., Kissil, J. L., Barry, M., Bronson, R. T., Shen, S. S., et al. (2007). Protein 4.1B suppresses prostate most cancers development and metastasis. Proc. Natl. Acad. Sci. U. S. A. 104 (31), 12784–12789. doi:10.1073/pnas.0705499104
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wu, Z., Ge, L., Ma, L., Lu, M., Tune, Y., Deng, S., et al. (2023). TPM2 attenuates development of prostate most cancers by blocking PDLIM7-mediated nuclear translocation of YAP1. Cell and Biosci. 13, 39. doi:10.1186/s13578-023-00993-w
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xiao, P., Ma, T., Zhou, C., Xu, Y., Liu, Y., and Zhang, H. (2016). Anticancer impact of docetaxel induces apoptosis of prostate most cancers by way of the cofilin-1 and paxillin signaling pathway. Mol. Med. Rep. 13 (5), 4079–4084. doi:10.3892/mmr.2016.5000
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xing, Z., Li, S., Liu, Z., Zhang, C., and Bai, Z. (2020). CTCF-induced upregulation of HOXA11-AS facilitates cell proliferation and migration by concentrating on miR-518b/ACTN4 axis in prostate most cancers. Prostate 80 (5), 388–398. doi:10.1002/professionals.23953
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xu, N., Chen, S.-H., Lin, T.-T., Cai, H., Ke, Z.-B., Dong, R.-N., et al. (2020). Improvement and validation of hub genes for lymph node metastasis in sufferers with prostate most cancers. J. Cell. Mol. Med. 24 (8), 4402–4414. doi:10.1111/jcmm.15098
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xu, X., Xu, S., Wan, J., Wang, D., Pang, X., Gao, Y., et al. (2023). Disturbing cytoskeleton by engineered nanomaterials for enhanced most cancers therapeutics. Bioact. Mater. 29, 50–71. doi:10.1016/j.bioactmat.2023.06.016
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, A. C., Rasul, R., Golden, A., and Feuerstein, M. A. (2021a). Incidence and mortality tendencies of metastatic prostate most cancers: surveillance, Epidemiology, and Finish Outcomes database evaluation. Can. Urological Assoc. J. = J. De l’Affiliation Des Urologues Du Can. 15 (12), E637–E643. doi:10.5489/cuaj.7173
Zhang, N., Bian, Q., Gao, Y., Wang, Q., Shi, Y., Li, X., et al. (2023a). The position of fascin-1 in human urologic cancers: a promising biomarker or therapeutic goal? Technol. Most cancers Res. Deal with. 22, 15330338231175733. doi:10.1177/15330338231175733
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, X., Fang, J., Chen, S., Wang, W., Meng, S., and Liu, B. (2019). Nonconserved miR-608 suppresses prostate most cancers development by way of RAC2/PAK4/LIMK1 and BCL2L1/caspase-3 pathways by concentrating on the three’-UTRs of RAC2/BCL2L1 and the coding area of PAK4. Most cancers Med. 8 (12), 5716–5734. doi:10.1002/cam4.2455
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, X., Yi, X., Zhang, Q., Tang, Y., Lu, Y., Liu, B., et al. (2022). Microcystin-LR induced microfilament rearrangement and cell invasion by activating ERK/VASP/ezrin pathway in DU145 cells. Toxicon 210, 148–154. doi:10.1016/j.toxicon.2022.02.023
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, Y., Meng, X., Ma, Z., Solar, Z., and Wang, Z. (2023b). Affiliation of androgen-receptor gene mutations with the copy variety of androgen-receptor silk protein A posh and glutathione-S-transferases T1 and M1 in prostate most cancers sufferers. Genet. Res. 2023, 5956951. doi:10.1155/2023/5956951
Zhang, Y.-G., Niu, J.-T., Wu, H.-W., Si, X.-L., Zhang, S.-J., Li, D.-H., et al. (2021b). Actin-binding proteins as potential biomarkers for continual inflammation-induced most cancers prognosis and remedy. Anal. Cell. Pathol. Amst. 2021, 6692811. doi:10.1155/2021/6692811
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhao, Y., Zhang, H., Wang, H., Ye, M., and Jin, X. (2022). Function of formin INF2 in human ailments. Mol. Biol. Rep. 49 (1), 735–746. doi:10.1007/s11033-021-06869-x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhu, B., Fukada, Ok., Zhu, H., and Kyprianou, N. (2006). Prohibitin and cofilin are intracellular effectors of reworking progress issue beta signaling in human prostate most cancers cells. Most cancers Res. 66 (17), 8640–8647. doi:10.1158/0008-5472.CAN-06-1443
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zu, S., Ma, W., Xiao, P., Cui, Y., Ma, T., Zhou, C., et al. (2015). Analysis of docetaxel-sensitive and docetaxel-resistant proteomes in PC-3 cells. Urol. Int. 95 (1), 114–119. doi:10.1159/000351263
PubMed Summary | CrossRef Full Textual content | Google Scholar