Karyotypic aberrations of chromosome (chr) 6 embody duplications (trisomy 6 [+6]), deletions of its brief arm (del6p), uniparental disomy 6p (UPD6p), in addition to numerous microduplications and microdeletions. Lesions involving 6p are of explicit curiosity as they level towards HLA locus involvement, together with lack of heterozygosity and/or haploinsufficiency, as pathogenic drivers ensuing from deletions and UPD6p [1, 2]. Together with somatic mutations in numerous HLA alleles, deletions of varied sizes together with microdeletions of the HLA locus in addition to somatic UPD6p have been urged to outcome from immune stress and to behave as technique of escape hematopoiesis in immune-mediated aplastic anemia (AA) [3, 4]. Equally, our group described these lesions within the context of lack of graft versus leukemia (GvL) impact of mismatched donor HLA alleles following allogeneic hematopoietic stem cell transplant (alloHSCT) with subsequent relapse of myeloid leukemias [5]. We additional demonstrated that the enrichment of particular amino acids throughout the peptide-binding groove of HLA class II, particularly HLA-DRB1, impacts its interplay with the T-cell receptor (TCR) and therefore underlies the autoreactivity inherent to autoimmune AA [6].
Trisomy 6 has been noticed in myelodysplastic neoplasm (MDS) and acute myeloid leukemia (AML) and may happen as a sole genomic abnormality. It has additionally been reported in AA and different bone marrow failure problems [7]. Nevertheless, so far studies have been restricted to particular person or small case sequence, therefore precluding a complete evaluation of the scientific and molecular options of those sufferers. Particularly, the pathogenesis of duplication has not been clarified and will contain numerous potential oncogenes situated on chr 6, e.g., RAB44, ECT2L, amongst others. Alternatively, as with different chromosomal lesions, main drivers could also be situated on different chromosomes. As well as, the intrachromosomal disruption of the HLA locus by means of microdeletions or lack of perform mutations could presumably facilitate illness evolution and development. Whereas analyzing the HLA locus in AA, we noticed a number of circumstances with remoted +6 (iso +6), which served because the impetus for the formulation of this report in lieu of the affiliation of analogous occurrences in MDS and AML and the enigmatic scientific options and numerous pathogenesis of +6.
Along with circumstances from our clinics (N = 83), our search was complemented by iso +6 circumstances described within the literature (N = 54) to include further scientific information on iso +6 AA, MDS and AML [7] (metanalytic circumstances are summarized in Supplementary Tables S1 and S2). In whole, we have been capable of look at a big cohort of sufferers collected from a number of sources and recognized with MDS (n = 3971), AML (n = 6788), or AA (n = 706) for iso +6, non-isolated +6 (non-iso +6), and regular karyotype (NK) illness for comparisons (Fig. 1A and Supplementary Appendix) [8,9,10,11,12]. For chosen circumstances with +6 and wholesome controls (N = 8), we’ve additionally carried out expression evaluation utilizing bulk deep RNA-sequencing (RNA-seq) (Supplementary Appendix).
A Stream diagram of sufferers with trisomy 6 and aplastic anemia (AA), myelodysplastic neoplasm (MDS), or acute myeloid leukemia (AML) included in our research. B Comparability of baseline scientific traits of remoted trisomy 6 (iso +6)-related AA, MDS, and AML utilizing unpaired t-test for steady variables and Chi-square or Fisher’s actual take a look at for categorical variables.
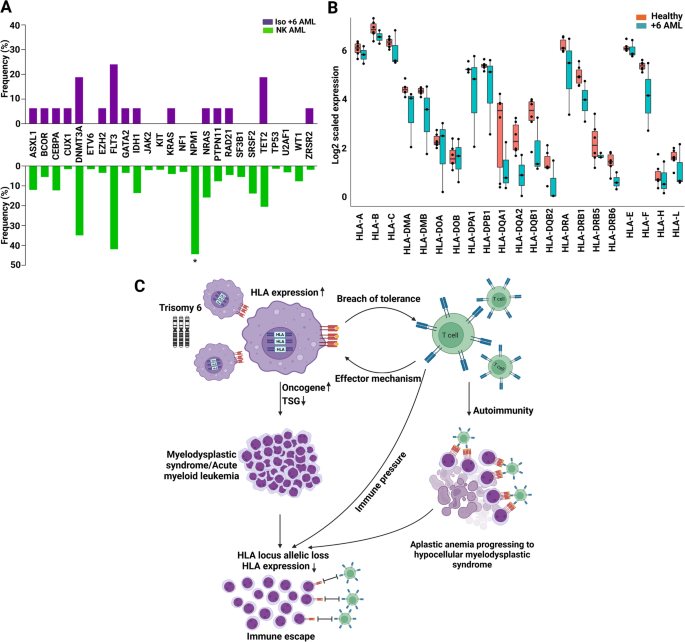
In whole, we recognized 94 +6 AML sufferers, of whom 48 had iso +6 and 46 non-iso +6. As well as, 16 iso +6 MDS and 14 iso +6 AA circumstances have been discovered (Fig. 1A and Supplementary Tables S1, S2). Of observe is that, the iso +6 AML circumstances from our cohort (n = 16) and the one reported within the literature (n = 32) exhibited the same scientific phenotype and survival traits (Supplementary Desk S3). Generally, iso +6 AML introduced at a youthful age of fifty vs 66.3 years (P < 0.0001) in comparison with NK AML. Iso +6 had analogous ontogenesis as NK AML (main AML – pAML – proportions of 89.4 vs 85.7%, P = 0.7), and exhibited the same hyperproliferative AML phenotype (median WBC depend 20.8 vs 14.4 × 109/L, P = 0.8). Focused next-generation sequencing demonstrated related co-mutational patterns to NK AML, together with FLT3 mutations, besides NPM1 mutations which weren’t present in iso +6 (0 vs 44.5% in NK AML, P = 0.0013; Fig. 2A). Remarkably, 5/16 iso +6 AML sufferers didn’t harbor any of the frequent myeloid mutations (Supplementary Desk S4). The remaining circumstances of iso +6 AML have been enriched for DNMT3A, FLT3, and TET2 however had much less TP53 mutations in comparison with non-iso +6 illness (Supplementary Desk S5). Regardless of the low mutational burden, iso +6 in AML sufferers had shorter general survival (median 22 vs 32.1 months; P = 0.04) when in comparison with NK AML (Supplementary Fig. S1); but, survival was not considerably completely different relative to non-iso +6 AML (22 vs 11 months, P = 0.1; Supplementary Fig. S1). By way of remedy, 2/2 sufferers who obtained 7 + 3 regimens adopted by cytarabine consolidation achieved remission, and 4/6 iso +6 AML sufferers efficiently underwent alloHSCT and remained relapse-free post-transplant (Supplementary Desk S3).
A Spectrum of mutations in trisomy 6 acute myeloid leukemia. The same mutational patterns throughout iso +6 AML and NK AML aside from NPM1 mutations that are much less frequent in remoted +6 AML. B The expression of HLA genes in trisomy 6 AML sufferers vs wholesome controls. C Scheme illustrating the proposed dichotomy “autoimmunity vs immune stress with HLA somatic loss” underlying the mechanisms of trisomy 6-related myeloid problems.
In MDS, iso +6 illness additionally introduced at a considerably youthful age vs NK MDS (median 43 vs 70 years, P < 0.0001). Apparently, iso +6 MDS manifested a definite scientific phenotype, notably characterised by profound anemia (median 8.4 vs 9.5 g/dL, P = 0.04) and hypocellular BM morphology (75% vs 9.5%, P = 0.0002) in comparison with NK MDS. Like AML, 3 of 4 iso +6 MDS sufferers with out there sequencing didn’t have any detectable mutations. Transformation price to AML was 37.5% with a median time to development of 5 months (IQR 2–33.5). The affiliation of +6 with pancytopenia and hypocellularity in 75% of the MDS circumstances is suggestive of a pathogenetic similarity with AA. Certainly, 2 of 14 sufferers with iso +6 AA finally remodeled to MDS.
When carried out, RNA-seq in +6 AML (n = 3) vs wholesome BM samples (n = 5) confirmed decrease HLA mRNA expression in +6 AML, together with 2/3 +6 AML circumstances with HLA expression ≤twentieth percentile of controls in 18 of 21 sequenced HLA genes, and even fully absent in 14 of the 21 HLA genes (Fig. 2B). We then analyzed HLA genes to seek for somatic hits in +6 MDS/AML (N = 8) utilizing our in-house developed HLA pipeline [6]. Whereas no mutations have been detected within the +6 MDS (n = 3) or +6 AML (n = 5) samples, but 2/5 +6 AML sufferers have been discovered to have allelic lack of HLA-DRB1 (Supplementary Desk S6). In distinction, not one of the 3 +6 MDS circumstances had any HLA lesions (Supplementary Desk S6). Each +6 AML sufferers with allelic loss have been pAML and had advanced karyotypes, comparable age (67 vs 58 years), related Hb (9.3 vs 9.4 g/dL) and platelet depend (16 vs 14 ok/uL), and a big share of BM blasts (79 vs 60%). Nevertheless, the primary affected person had considerably increased WBC depend (162.6 vs 5.3 ok/uL) and better variety of co-mutations (ASXL1, PRPF8, SRSF2 and TP53 vs BCOR and TP53, respectively). With regard to outcomes, each sufferers survived <1 month after analysis.
Our outcomes recommend that +6 could current as the only cytogenomic abnormality in AA with development to MDS as urged by the hypoproliferative phenotype harking back to prior AA. It’s doable that +6 through duplication of HLA locus could result in elevated expression of peptides triggering immune response in AA, e.g., as a part of tumor surveillance response. In distinction, +6 within the context of AML displays opposed danger presumably as a result of paucity of prognostically favorable NPM1 mutations. As well as, the absence of heightened HLA expression in sure +6 AML circumstances could contribute to the distinct scientific conduct of +6 in AML by means of HLA downregulation or segmental microdeletions as demonstrated in our research. Primarily based on these outcomes, one may speculate that an elevated copy of the HLA locus in AA and MDS could result in elevated autoantigen presentation past TCR activation threshold, presumably resulting in breach of tolerance, a mechanism which may be operative in AA and hypocellular MDS (Fig. 2C). Thus, clonal duplication by means of copy quantity features, +6, or UPD of the HLA locus in MDS could end in immune stress as seen in AA and GvL impact [13,14,15]. Certainly, we recognized frequent mixture options of pancytopenia and BM hypocellularity in iso +6 MDS on this sequence. In additional superior ailments, further triggers equivalent to deletions of tumor suppressor genes or acquisition of subclonal mutations may play a task, with development reflecting somatic stress to beat immune inhibition. Conversely, the allelic loss and decrease expression of HLA in +6 AML could suggest a dichotomy of mechanisms by which +6 contributes to superior leukemia vs bone marrow failure. Additional proof of allelic deletion of HLA genes in +6 AML factors towards immune escape and evasion, a mechanism already described in relapsing AML below immune stress [5] and probably contributing to the transformation of AA to malignant illness on this sequence.
To the very best of our data, we herein carried out probably the most complete clinico-genomic meta-analysis of the most important cohort ever reported of +6 in myeloid neoplasms, given its rarity within the literature. Our research consists of novel analyses of the contrasting, context-dependent position of HLA in mediating the pathogenesis of MDS and AML by predisposing to hyperimmunity and its somatic alIelic loss, respectively.