CAR T cells are much less efficient at killing myeloid leukemia than B-lineage leukemia
We first in contrast the anti-tumor exercise of CD33 CAR T cells towards AML and B-ALL cell strains that had been engineered to precise equal ranges of CD33 (U937CD33 and Nalm6CD33) (Fig. 1A). CD33 CAR T cells exhibited decrease cytotoxicity to U937CD33 than to Nalm6CD33 cells at numerous effector-to-target (E:T) ratios (Fig. 1B). Comparable outcomes had been noticed with CLL1 and CD123 CAR T cells (Fig. 1C). Nonetheless, there was no important distinction in T-cell counts and the apoptotic price after co-incubation with U937CD33 and Nalm6CD33 (Supplementary Fig. 1A, B). U937CD33-exposed CAR T expressed fewer activation markers CD25 and CD69, and effector molecules interferon-γ (IFN-γ), granzyme B, and interleukin-2 (IL-2) (Fig. 1D). We additional evaluated the cytotoxicity to completely different leukemia cell strains. The outcomes confirmed a decrease killing efficiency of CD33 CAR T to myeloid U937CD33 and THP-1CD33 than to lymphoid Nalm6CD33 and RajiCD33 cells. Apparently, no important distinction in cytotoxicity was noticed between RajiCD33 and HL60CD33 cells (Fig. 1E). The cytotoxicity of CD38 CAR T to major AML cells was decrease than to major ALL cells with equal ranges of CD38 antigen (Fig. 1F and Supplementary Fig. 1C).
A Consultant histograms displaying CD33 expression. B Cytolytic exercise of CD33 CAR T cells in vitro. n = 3 organic replicates with T cells from completely different donors per level. C Cytolytic exercise of CLL1 and CD123 CAR T cells in vitro, n = 3. D Share of CD25 and CD69, IFN-γ, Granzyme B (GZMB), and IL-2 in CAR T cells, n = 3. E Cytolytic exercise of CD33 CAR T cells towards completely different cell strains in vitro, n = 3. F Consultant histograms displaying CD38 expression on sorted major samples and CD38 CAR T cytolytic exercise in vitro, n = 4 in ALL group, n = 5 in AML group. G Schematic of mouse mannequin. NSG mice had been intravenously injected with tumor cells, adopted by 1 × 106 CAR or PCDH T cells 5 days later. Peripheral blood (PB), spleen (SP), and bone marrow (BM) cells had been collected from mice euthanized on days 3–5. H CAR T-cell counts in SP, BM, and PB from (G), n = 4 in SP and BM, n = 5 in PB. I Share of Annexin V+ of CAR T cells in spleen from (G), n = 3. J Share of CD25, GZMB, IFN-γ, and IL-2 of CAR T cells in spleen from (G), n = 3 in IL-2 expression, n = 4 in any other case. Ok Complete Naïve (N, CD45RA+CD62L+), central reminiscence (CM, CD45RA−CD62L+), effector reminiscence (EM, CD45RA−CD62L−) and effector (E, CD45RA+CD62L−) CAR T cells had been assigned to CD4+ and CD8+ CAR T cells in spleen, n = 3. L Share of PD-1, TIM-3, and TOX in CAR T cells in spleen, n = 5 in TOX, n = 3 in any other case. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests or a number of two-sided unpaired t checks had been used to evaluate significance in (B, C, D, F, H–L). One-way ANOVA was utilized in (E). All numbers outlined by “n” point out the variety of organic replicates with completely different human donors or mice. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
Subsequent, we evaluated the anti-tumor exercise of CD33 CAR T cells in NSG mice that had been pre-inoculated with both U937CD33 or Nalm6CD33 cells (Fig. 1G and Supplementary Fig. 1D). Though U937CD33 and Nalm6CD33 cells grew at completely different charges in mice, we noticed a quicker illness development and shorter survival in mice bearing U937CD33 cells at completely different preliminary CAR T-to-tumor cell ratios (Supplementary Fig. 1E). CAR T cells had been barely detectable in U937CD33 mice, however had been detectable in Nalm6CD33 mice 2 weeks after CAR T infusion (Supplementary Fig. 1F). These outcomes counsel a severely impaired exercise of CAR T cells in mice with myeloid leukemia.
We additional evaluated the affect of myeloid cells on CAR T-cell phenotype in vivo by creating situations the place mice had an similar tumor burden earlier than CAR T-cell infusion (Supplementary Fig. 1G, H). After 3–5 days, CAR T cells in mice bearing U937CD33 tumors had fewer cell counts, greater apoptotic charges, and decrease expression of CD25, granzyme B, IFN-γ, and IL-2 than in mice bearing Nalm6CD33 tumors (Fig. 1H–J and Supplementary Fig. 1I). Throughout an prolonged remark interval of almost 2 weeks earlier than the disappearance of CAR T cells in U937CD33 mice, we didn’t observe any variations within the effector/reminiscence subsets or exhaustion markers between the 2 teams (Fig. 1K, L).
CAR-T has an impaired effector program and faulty antigen receptor sign transduction in AML
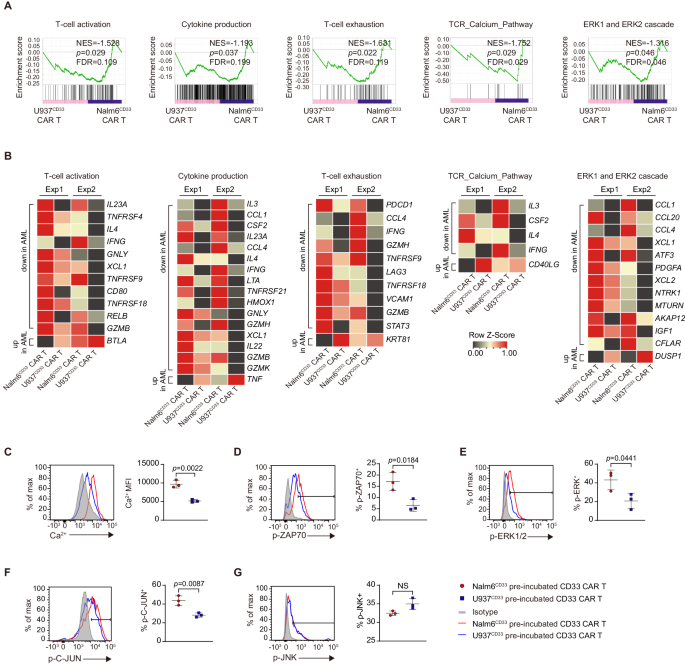
To analyze the mechanisms underlying CAR T cell incompetence to AML, we co-incubated CAR T cells with U937CD33 and Nalm6CD33 cells for two days after which sorted CD8+ CAR+ T cells to carry out RNA-seq (Supplementary Fig. 2A). Gene set enrichment evaluation (GSEA) revealed that U937CD33 co-incubated CAR T cells had a decrease degree of T cell activation, cytokine manufacturing, and T cell exhaustion than Nalm6CD33 co-incubated cells (Fig. 2A). Apparently, these cells exhibited diminished T cell receptor (TCR) calcium pathway and extracellular signal-regulated kinase (ERK) 1/2 cascade (Fig. 2A). Nonetheless, JNK-MAPK and nuclear issue of activated T cells (NFAT) signaling pathways had no important variations between the 2 teams (Supplementary Fig. 2B).
A, B GSEA outcomes from working RNAseq information of U937CD33 co-incubated- versus Nalm6CD33 co-incubated-CD33 CAR T cells (A). Nominal P values, FDR q values, and normalized enrichment rating (NES) had been calculated utilizing GSEA software program (Broad Institute). Warmth maps (B) indicating the expression of genes enriched in GSEA from (A) and the recognized associated genes not included within the GSEA gene set. The genes proven in heatmaps meet the parameters: fold change ≥ 1.5-fold in every of the 2 organic replicates. Every RNA pattern was pooled from three technical replicates with T cells from one donor, and we carried out experiment with two completely different donors, n = 2. C Depth of intracellular calcium in CAR T cells co-incubated with tumor cells, n = 3. Share of phosphorylated ZAP70 (D), phosphorylated ERK1/2 (E), phosphorylated C-JUN (F), and phosphorylated JNK (G) in CD33 CAR T cells pre-incubated with tumor cells and re-stimulated with U937 cells, n = 3. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests had been used to evaluate significance in (C–G). All numbers outlined by “n” point out the variety of organic replicates with completely different human donors. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
We then examined the particular genes differentially expressed in GSEA. U937CD33 co-incubated CAR T cells had decrease expression of cytokines IL3, IL4, IL22, and IL23A, cytotoxic molecules IFNg, GNLY, GZMB, and GZMH, and costimulatory molecules TNFRSF4 (OX40), TNFRSF9 (4-1BB), CD80, TNFRSF18 (GITR), and had decrease exhaustion-related genes together with PDCD1 and LAG3 (Fig. 2B). Regardless of a decrease degree of IL-2 protein in AML-co-incubated CAR T cells, IL2 mRNA confirmed no important change, suggesting that IL-2 could also be topic to post-transcriptional regulation. U937CD33 co-incubated CAR T cells additionally confirmed decreased expression of calcium signaling-related CSF213, and IFNg14, and the optimistic regulators of ERK signaling together with PDGFA15, NTRK116, MTURN17, IGF118, and CFLAR19, and elevated expression of DUSP120, a important phosphatase that inactivates ERK (Fig. 2B).
We subsequent assessed the exercise of antigen receptor downstream indicators. Upon antigen ligation to TCR, proximal lymphocyte-specific protein tyrosine kinase (LCK) and zeta-chain-associated protein kinase 70 (ZAP70) signaling are triggered, resulting in calcium mobilization, ERK, and JNK activation, and subsequent activation of transcription components similar to NFAT, FOS, and C-JUN21,22. Nonetheless, the phosphorylation of ERK in CAR T cells co-incubated with U937CD33 cells for 15–30 min just isn’t completely different from that of Nalm6CD33 cells, suggesting no instant defect in CAR T signaling after a brief AML publicity (Supplementary Fig. 2C). To realize additional perception into sign transduction, we pre-incubated CAR T cells with U937CD33 and Nalm6CD33 cells for 12 h after which sorted these cells and re-stimulated them with U937 cells. This two-step protocol is certainly to review the reprogramming occasions that happen inside CAR T cells through the preliminary 1-h co-incubated with myeloid and lymphoid tumor cells. We discovered that U937CD33 pre-incubated CAR T cells exhibited much less calcium inflow and decrease phosphorylated ZAP70 (p-ZAP70), ERK1/2 (p-ERK1/2), and C-JUN (p-C-JUN) proteins in comparison with Nalm6CD33 pre-incubated CAR T cells (Fig. 2C–F). Conversely, phosphorylated JNK protein didn’t present a distinction (Fig. 2G).
U937CD33 pre-incubated CAR T cells that obtained second stimulation with PMA or anti-CD3 had decrease p-ERK and p-C-JUN than Nalm6CD33 pre-incubated CAR T cells (Supplementary Fig. 2D, E). U937CD33 pre-incubated CAR T cells had decrease ranges of p-ZAP70 after anti-CD3 stimulation, however p-ZAP70 was not completely different after PMA stimulation, which bypassed TCR (Supplementary Fig. 2F). Complete ERK and C-JUN proteins in U937CD33 co-incubated CAR T cells are similar to or greater than these in Nalm6CD33 co-incubated CAR T cells, whereas complete ZAP70 protein was barely decrease (Supplementary Fig. 2G, H).
AML-expressed CD155 inhibits CAR T killing and attenuates the ERK signaling pathway
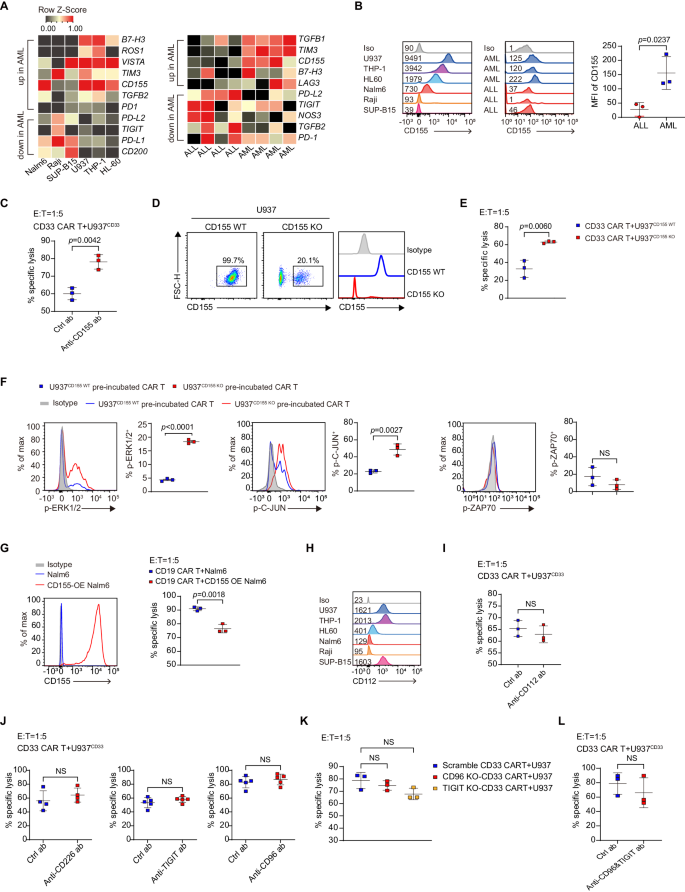
We subsequent explored which AML-derived components are answerable for the impaired perform of CAR T cells. Reactive oxygen species (ROS) and nitric oxide (NO), the recognized immunosuppressors23,24, had been expressed at diverse ranges throughout completely different AML and ALL cell strains (Supplementary Fig. 3A, C). Major AML samples confirmed greater expression of ROS however not NO (Supplementary Fig. 3B, D). Nonetheless, inhibition of NO and ROS didn’t enhance CAR-T cytotoxicity to U937CD33 cells (Supplementary Fig. 3E). To realize additional perception, we carried out transcriptome sequencing on a number of AML and ALL cell strains and first samples, which revealed greater expression of inhibitory ligands/receptors, together with B7-H3, TIM3, and CD155 in AML cell strains and first samples (Fig. 3A). As a result of B7-H325 and TIM326 had been inhibitory receptors, their expression on leukemia cells had been unlikely to ship an inhibitory sign in CAR T cells. Subsequently, we centered on CD155 which serves as an inhibitory ligand.
A Warmth maps displaying inhibitory genes which might be up- or down-regulated greater than two-fold. Major samples every had 4 organic replicates. B Histograms displaying CD155 expression of in tumor cell strains and first samples, and the MFI of CD155 in major samples, n = 3. C Cytolytic exercise of CD33 CAR T cells towards U937 cells within the presence of 500 ng/ml CD155 blocking antibody, n = 3. D Stream cytometric plots displaying knockout effectivity of CD155 on U937 cells. E Cytolytic exercise of CD33 CAR T cells towards U937CD155 WT and U937CD155 KO cells, n = 3. F Share of p-ERK1/2, p-C-JUN, and p-ZAP70 in CAR T cells pre-incubated with U937CD155 WT and U937CD155 KO cells and re-stimulated with U937 cells, n = 3. G Expression of CD155 in management and CD155-overexpressing (CD155 OE) Nalm6 cells, and CD19 CAR T cell cytolytic exercise towards them, n = 3. H Histograms displaying CD112 expression in AML and ALL cell strains. I Cytolytic exercise of CD33 CAR T cells towards U937 cells with 10 µg/ml CD112 antibody, n = 3. J Cytolytic exercise of CD33 CAR T cells towards U937 cells with 10 µg/ml CD226, 50 µg/ml TIGIT, or 20 µg/ml CD96 antibody, n = 4 in CD226 antibody group, n = 5 in any other case. Ok Cytolytic exercise of scramble-, CD96 knockout (CD96 KO), and TIGIT knockout (TIGIT KO)-CD33 CAR T cells towards U937 cells, n = 3. The scrambled CD33 CAR T had been electroporated with non-targeting sgRNAs. L Cytolytic exercise of CD33 CAR T cells towards U937 cells with each 50 µg/ml TIGIT and 20 µg/ml CD96 antibody, n = 3. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests had been used to evaluate significance in (B, C, E–G, I, J, L). One-way ANOVA was utilized in (Ok). All numbers outlined by “n” point out the variety of organic replicates with completely different human donors. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
Constantly, AML cell strains and first samples had greater ranges of CD155 protein than lymphoid controls (Fig. 3B). CD155 blocking antibody enhanced the killing exercise of CAR T cells towards U937 cells (Fig. 3C). When CD155 was knocked out in U937 cells, the killing exercise of CAR T cells was partially restored (Fig. 3D, E), and the degrees of p-ERK and p-C-JUN elevated, whereas p-ZAP70 confirmed no change (Fig. 3F and Supplementary Fig. 4A). Nonetheless, CD155 didn’t have an effect on the overall ERK, C-JUN, or ZAP70 protein expression (Supplementary Fig. 4B). Moreover, overexpression of CD155 in Nalm6 cells decreased the cytotoxicity of CD19 CAR T cells (Fig. 3G).
CD155 and the intently associated CD112 are ligands for stimulatory receptor CD226 and inhibitory receptors TIGIT and CD9627,28. CD112 expression diverse throughout AML and ALL cell strains, though its expression was greater in U937 than in Nalm6 (Fig. 3H). Blocking CD112 didn’t have an effect on the cytotoxicity of CD33 CAR T to U937 (Fig. 3I). Nonetheless, antibody blockade or knockout of TIGIT, CD96, or CD226 didn’t have an effect on the cytotoxicity of CD33 CAR T cells (Fig. 3J, Ok and Supplementary Fig. 4C, D, F). Moreover, blocking each TIGIT and CD96 concurrently had no impact on CD33 CAR T cell-cytotoxicity (Fig. 3L and Supplementary Fig. 4E).
Overexpression of C-JUN restores CAR T perform in AML
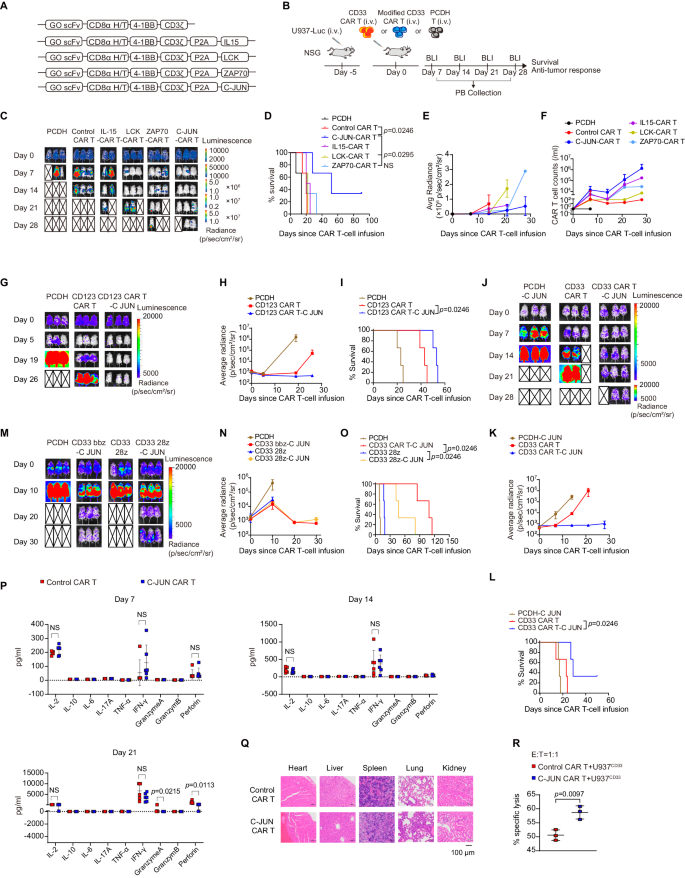
We subsequent sought to outline a method to revive CAR T perform in AML. As a result of the blockade of TIGIT and CD96 was unable to rescue the performance of CD33 CAR T cells, we sought to revive CAR T perform by overexpressing key antigen receptor signaling components to enhance therapeutic efficacy. LCK and ZAP70 present T cell receptor proximal signaling for antigen downstream sign initiation and transduction29. C-JUN and C-Fos are members of the AP-1 advanced, which regulates T cell activation and effector perform30. Thus, we developed CD33 CAR constructs overexpressing LCK, ZAP70, C-JUN, C-Fos, and IFN-γ, and that overexpressing IL-15, a cytokine recognized to potentiate CAR T therapeutic exercise in numerous tumor fashions31,32,33, as a management (Fig. 4A, B and Supplementary Fig. 5A, B). The exercise of CAR T cells overexpressing IL-15, LCK, ZAP70, and C-JUN, however not C-Fos and IFN-γ, improved at numerous levels (Fig. 4C–E and Supplementary Fig. 5C, D). CAR T cells overexpressing IL-15 confirmed enhanced growth in mice however sadly didn’t management tumor development as successfully as different molecules (Fig. 4C–F). CAR T cells overexpressing LCK successfully managed tumor development at an early stage, however the impact was not as long-lasting as that of ZAP70 or C-JUN, whereas overexpression of C-JUN confirmed the best impact in bettering tumor management and prolonging mouse survival (Fig. 4C–E). C-JUN overexpression didn’t have an effect on the CAR transduction price (Supplementary Fig. 6A–D), and C-JUN CAR T cells confirmed superior efficacy regardless of completely different transduction charges in several experimental batches (Supplementary Fig. 6E, F). C-JUN impact was recapitulated in CD123 CAR T cells (Fig. 4G-I).
A Schematic of CAR constructs (gemtuzumab ozogamicin, GO). B Schematic of mouse mannequin. NSG mice obtained U937 cells intravenously adopted by CAR T cells or management PCDH T cells 5 days later. C Consultant bioluminescence imaging of mouse mannequin after CAR T remedy, n = 2 in IL15-CAR T group, n = 3 in different teams. Survival curve (D) and quantification of tumor burden (E) as indicated by common radiance (p/sec/cm²/sr) of (C), n = 2 in IL15-CAR T group, n = 3 in different teams. F CAR T-cell counts in PB, information are summarized from two impartial experiments, n = 3 in PCDH and IL15-CAR T group, n = 4 in LCK-CAR T and ZAP70-CAR T group, n = 5 in Management CAR T and C-JUN-CAR T group. G, J, M Consultant bioluminescence imaging of mouse mannequin after CAR T remedy, n = 3. H, Ok, N Quantification of (G, J, M) displaying the tumor burden as indicated by common radiance (p/sec/cm²/sr), n = 3. (I, L, O) Survival curve of NSG mice in (G, J, M), n = 3. P Cytokine ranges in tail blood collected on days 7, 14, and 21 post-infusion, n = 6. Q Pathological evaluation of the center, liver, spleen, lung, and kidney of the consultant two mice within the management and C-JUN CAR T-treatment group at their terminal stage by utilizing HE staining. Magnification, 200×. R Cytolytic exercise of management and C-JUN CAR T cells towards U937 cells in vitro, CAR T cells had been sorted from the mice spleen, n = 3. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests or a number of two-sided unpaired t checks had been used to evaluate significance in (P, R). Survival curves had been in contrast utilizing the log-rank Mantel-Cox take a look at in (D, I, L, O). All numbers outlined by “n” point out the variety of organic replicates with completely different mice. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
C-JUN overexpression alone in non-CAR T cells couldn’t get rid of tumors or delay survival (Fig. 4J–L). We subsequent evaluated C-JUN affect on CAR T cells with completely different costimulation domains and noticed that mice within the CD33 bbz-C-JUN and CD33 28z-C-JUN CAR T cell remedy teams had comparable results in enhancing tumor management in contrast with non-C-JUN CAR T cells (Fig. 4M–O and Supplementary Fig. 6H). In C-JUN CAR T-treated mice, regardless of enhanced anti-tumor exercise, CAR T cells steadily decreased over time (Supplementary Fig. 6I), resulting in tumor development and eventual demise in a proportion of mice. C-JUN CAR T cells didn’t induce a big improve in cytokine ranges in tail blood (Fig. 4P), and pathological analysis of significant organs revealed no important inflammatory cell infiltration, suggesting that C-JUN CAR T didn’t trigger inflammatory organ toxicity in mice mannequin (Fig. 4Q).
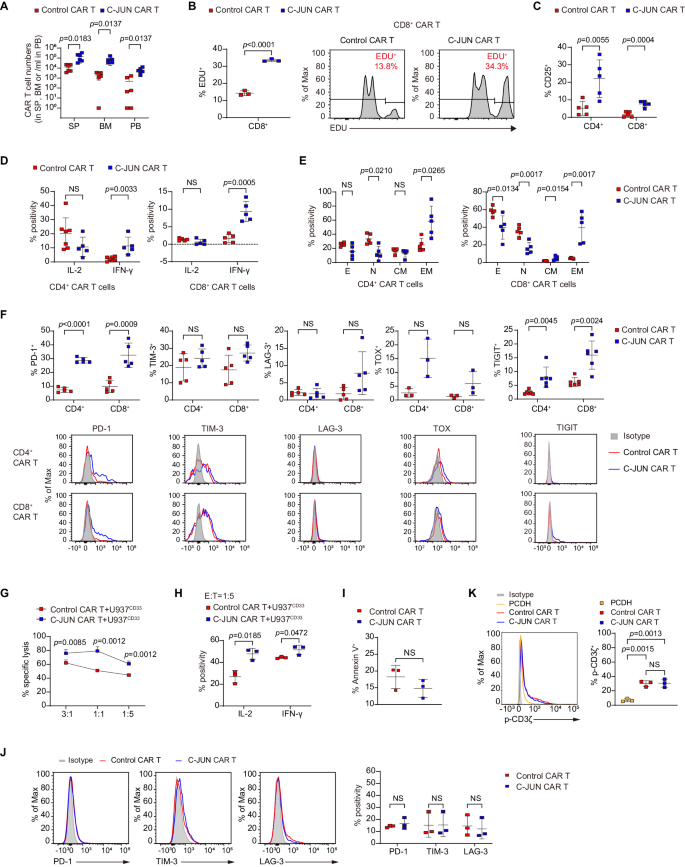
We subsequent investigated the impact of C-JUN overexpression on CAR T-cell growth and performance in vivo. C-JUN CAR T cells sorted from U937-bearing mice exhibited larger cytotoxicity towards U937 than management CAR T cells (Fig. 4R). In comparison with management teams, C-JUN CAR T cells had greater in vivo numbers and confirmed the next proliferative price (Fig. 5A, B). They expressed greater IL-2 receptor (CD25) and IFN-γ (Fig. 5C, D). Nonetheless, no important distinction within the expression of IL-2 was noticed (Fig. 5D). Contrasting to regulate cells which had been primarily naïve subpopulations, C-JUN CAR T cells had been primarily enriched in effector reminiscence subpopulations inside the CD4+ subset and enriched in each effector reminiscence and central reminiscence subpopulations inside the CD8+ subset (Fig. 5E and Supplementary Fig. 6J). C-JUN CAR T cells didn’t present considerably modified apoptosis (Supplementary Fig. 6K) or alleviated exhaustion (Fig. 5F).
A CAR T cell counts in SP, BM, and PB collected from day 10 to twenty post-infusion, n = 6. B Share of EDU+ cells in CD8+ CAR T cells, n = 3. C Share of CD25+ cells in CAR T cells, n = 5. D Share of IL-2 and IFN-γ in CAR T cells, n = 7 in CD4+ CAR T cells within the management CAR T group, n = 5 in any other case. E Share of naïve (N, CD45RA+CD62L+), central reminiscence (CM, CD45RA−CD62L+), effector reminiscence (EM, CD45RA−CD62L−), and effector (E, CD45RA+CD62L−) cells in CAR T cells, n = 5. F Share of PD-1, TIM-3, LAG-3, TOX, and TIGIT in CAR T cells, n = 3 in TOX, n = 6 in TIGIT, n = 5 in any other case. G Cytolytic exercise of management and C-JUN CAR T cells towards U937 cells in vitro, CAR T cells had been cocultured with U937 cells on the indicated E:T ratios, n = 3 replicates per level, consultant of three donors. H Share of IL-2 and IFN-γ in CAR T cells cocultured with U937 cells in vitro, n = 3. I Share of Annexin V+ in management and C-JUN CAR T cells cultured alone, n = 3. J Share of PD-1, TIM-3, and LAG-3 in management and C-JUN CAR T cells cultured alone, n = 3. Ok Expression of phosphorylated CD3ζ in management CAR-T, C-JUN CAR-T, and PCDH (non-CAR transduction) T cells cultured alone, n = 3. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests or a number of two-sided unpaired t checks had been used to evaluate significance in (A–J). One-way ANOVA was utilized in (Ok). All numbers outlined by “n” point out the variety of organic replicates with completely different human donors or mice. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
C-JUN CAR T cells maintained in vitro additionally exhibited enhanced killing skill and produced extra IL-2 and IFN-γ when co-incubated with U937 (Fig. 5G, H). We subsequent examined whether or not C-JUN overexpression had any impact on the tonic sign in CAR T cells with out antigen stimulation. We discovered no important variations between management CAR T cells and C-JUN CAR T cells cultured alone by way of apoptosis and inhibitory receptors (PD-1, TIM-3, LAG-3) (Fig. 5I, J). There was a rise in CD3ζ phosphorylation (p-CD3ζ) in each management CAR T cells and C-JUN CAR T cells in comparison with PCDH T cells, however no distinction was discovered between management CAR T cells and C-JUN CAR T cells (Fig. 5K). It must be famous that each CAR and TCR can induce CD3ζ phosphorylation, so these outcomes counsel that C-JUN overexpression had little impact on the general CD3ζ signaling downstream of each TCR and CAR within the absence of overt antigen stimulation.
Overexpression of C-JUN upregulates costimulatory molecules and cytokines and reactivates ERK
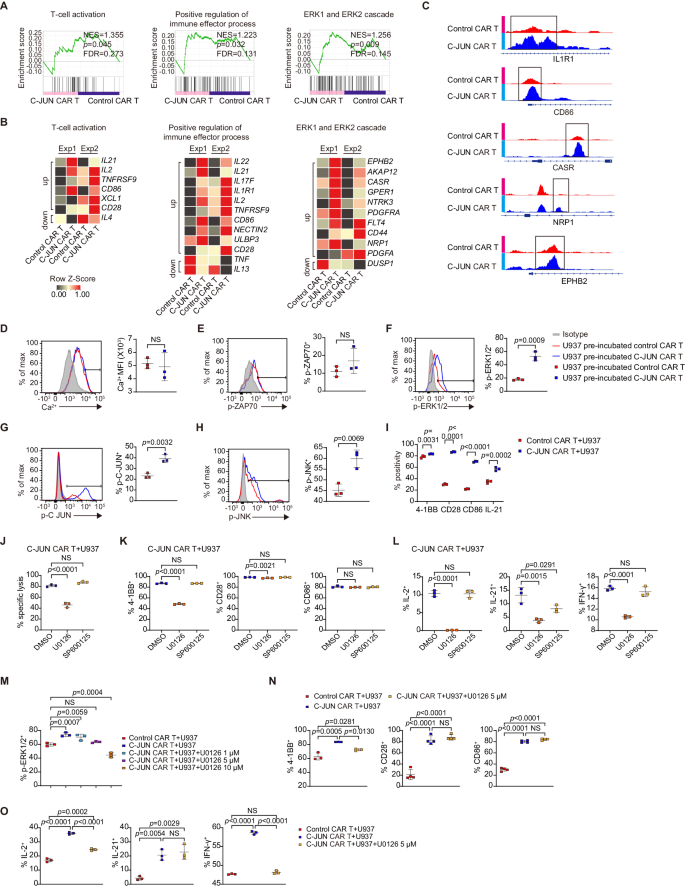
To know the mechanisms underlying the enhancing impact of C-JUN, we co-incubated CAR T with U937 cells for two days, after which a transcriptome evaluation was carried out on management or C-JUN CAR T cells (Supplementary Fig. 7A). GSEA revealed that C-JUN CAR T cells exhibited greater ranges of T cell activation, optimistic regulation of immune effector course of, and ERK1/2 cascade (Fig. 6A). Nonetheless, no important adjustments in JNK-MAPK, NFAT, TCR calcium, or exhaustion pathways had been noticed (Supplementary Fig. 7B).
A, B GSEA outcomes from RNAseq of U937 co-incubated- management versus C-JUN CAR T (A). Nominal P values, FDR q values, and NES had been calculated utilizing GSEA software program (Broad Institute). Warmth maps (B) indicating the expression of genes enriched in GSEA from (A) and the recognized associated genes not within the GSEA gene set. The genes in heatmaps meet: fold change ≥ 1.5-fold in every of the 2 organic replicates. Every RNA pattern was pooled from three technical replicates with T cells from one donor, and we carried out experiment with two completely different donors, n = 2. C Differentially accessible areas of indicated genes from ATAC-seq evaluation. D Depth of intracellular calcium in CAR T cells, n = 3. Expression of p-ZAP70 (E), p-ERK1/2 (F), p-C-JUN (G), and p-JNK (H) in CAR T cells pre-incubated with U937 and re-stimulated with U937, n = 3. I Share of 4-1BB, CD28, CD86, and IL-21 in CAR T cells cocultured with U937, n = 3. J Cytolytic exercise of C-JUN CAR T cells towards U937 with 50 µM U0126 or 50 µM SP600125, n = 3. Share of 4-1BB, CD28, CD86 (Ok), and IL-2, IL-21, IFN-γ (L) in T cells from (J), n = 3. (M) Share of p-ERK1/2 in U937 pre-incubated-control CAR T, C-JUN CAR T, and C-JUN CAR T with 1, 5, 10 µM U0126, n = 3. Share of 4-1BB, CD28, and CD86 (N) and IL-2, IL-21, and IFN-γ (O) in U937-co-incubated management CAR T, C-JUN CAR T, and C-JUN CAR T with 5 µM U0126, n = 4 in CD28 and CD86 expression, n = 3 in any other case. For all bar plots, information are proven as imply ± SD. Assays had been carried out on day 10 after T-cell preliminary activation. Two-sided unpaired t-tests or a number of two-sided unpaired t checks had been used to evaluate significance in (D–I). One-way ANOVA was utilized in (J–O). All numbers outlined by “n” point out the variety of organic replicates with completely different human donors. Information are consultant of two impartial experiments. NS not important. Supply information are offered within the Supply Information file.
Examination of particular genes in C-JUN CAR T cells revealed greater ranges of transcripts for cytokines IL2, IL17F, IL21, and IL22, cytokine receptor IL1R1, cytotoxicity mediator ULBP334, and costimulatory molecules TNFRSF9 (4-1BB), CD28, and CD86 (Fig. 6B). IFNg mRNA can be elevated in C-JUN CAR T cells, however no more than 1.5 fold (Supplementary Fig. 7C). As well as, C-JUN CAR T cells had greater expression of CASR35, EPHB236, NRP137, PDGFA15, and PDGFRA38, upstream optimistic regulators of ERK1/2 activation (Fig. 6B). Notably, exhaustion-related genes had been principally upregulated or unchanged in C-JUN CAR T cells (Supplementary Fig. 7C).
In chromatin accessibility evaluation, C-JUN CAR T cells had considerably extra distinctive accessible chromatin areas (13192 peaks) than management cells (7529 peaks). Notably, C-JUN CAR T confirmed elevated chromatin accessibility close to genes associated to T cell effector perform, together with IL1R1 and CD86, and the regulators of ERK activation, similar to CASR, NRP1, and EPHB2 (Fig. 6C).
Sign transduction exercise was then analyzed. After 12 h pre-incubation and 15–30 min re-stimulation with U937 cells, C-JUN CAR T cells had considerably greater ranges of p-ERK and p-C-JUN than management CAR T cells, whereas p-JNK confirmed solely a slight improve, and no variations in calcium inflow or p-ZAP70 had been noticed (Fig. 6D–H). We additionally carried out a re-stimulation with U937CD33KO (damaging management), PMA, or anti-CD3. The outcomes confirmed that C-JUN CAR T cells re-stimulated with PMA or anti-CD3 or had greater p-ERK1/2, p-C-JUN, and p-JNK (solely with anti-CD3) ranges than management CAR T cells (Supplementary Fig. 7D). C-JUN overexpression didn’t change complete ERK and JNK protein ranges, however elevated C-JUN protein degree in CAR T cells which have been co-incubated for 12 h with U937 (Supplementary Fig. 7E). Subsequently, the elevated p-ERK and p-JNK had been unlikely a results of elevated complete protein ranges, however could as a substitute be attributable to an elevated price of phosphorylation induced by upstream activating indicators.
After confirming that C-JUN CAR T cells expressed greater ranges of 4-1BB, CD28, CD86, and IL-21 protein (Fig. 6I), we investigated whether or not the elevated p-ERK1/2 and p-JNK had been concerned within the improved cytotoxicity and upregulation of costimulatory molecules/cytokines in C-JUN CAR T cells. The MEK1/2 inhibitor U0126, however not the JNK inhibitor SP600125, abolished the improved killing capability (Fig. 6J). At a excessive focus of fifty µM, U0126 suppressed 4-1BB, CD28, IL-2, IL-21, and IFN-γ protein expression, whereas SP600125 didn’t have an effect on their protein ranges, apart from IL-21 (Fig. 6K, L). Moreover, at a excessive focus of fifty µM, U0126 suppressed the expression of 4-1BB, IL-2, IL-21, and IFN-γ in management CD33 CAR T cells. This implies that ERK is required for the expression of 4-1BB, IL-2, and IFN-γ, whereas JNK and ERK could also be concerned within the expression of IL-21 (Supplementary Fig. 8A). To additional assess whether or not this elevation of co-stimulatory molecules and cytokines is a results of C-JUN-mediated improve of ERK exercise or a direct C-JUN transcriptional regulation, we additional titrate down U1026 to a focus of 5 µM to deliver p-ERK ranges in C-JUN CAR-T to the identical degree of management CAR T cells (Fig. 6M). On this situation, U0126 solely suppressed 4-1BB, IL-2, and IFN-γ, however not CD28, CD86, and IL-21 protein (Fig. 6N, O and Supplementary Fig. 8B). These outcomes collectively counsel the elevated 4-1BB, IL-2, and IFN-γ is probably going depending on C-JUN-mediated improve of ERK exercise, however the elevated CD28, CD86, and IL-21 protein is fairly depending on the direct C-JUN-mediated transcriptional activation. This implies that the benefit of C-JUN lies on the synergistic motion of suggestions reactivation of ERK and its direct transcription issue exercise.
Security of C-JUN-overexpressing CD33 CAR T cells in sufferers with r/r AML
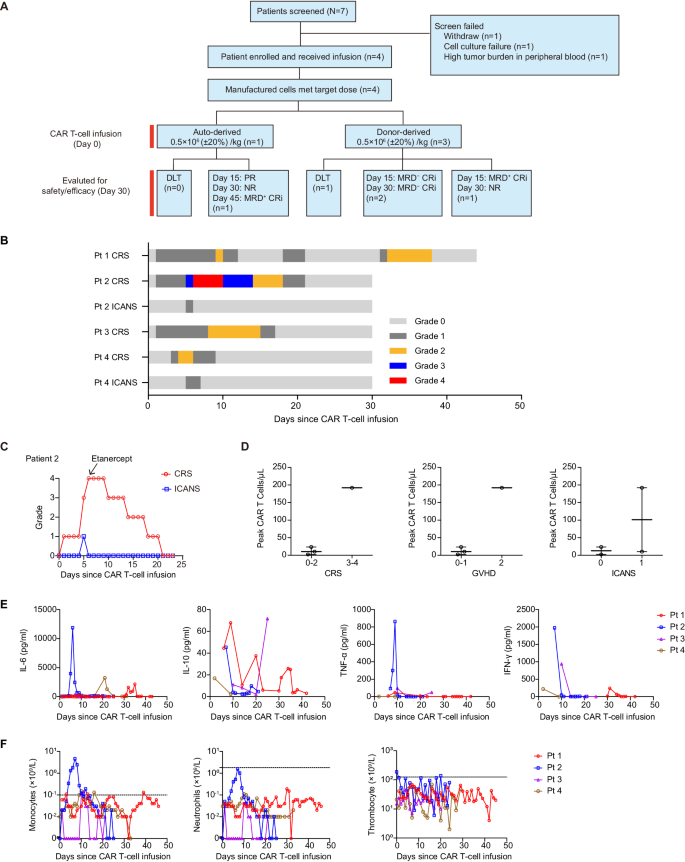
We initiated a Section I trial to guage security and efficacy of optimized CD33 CAR T cells overexpressing C-JUN in sufferers with r/r AML. Of the 7 sufferers screened, 4 had been enrolled (Fig. 7A). Two sufferers had been male and two had been feminine. The median age of the 4 sufferers enrolled was 9.5 years (vary, 3–12). These sufferers had skilled morphologic relapse with out extramedullary illness and had obtained 2–6 strains of prior therapies, and three had prior transplantation. The baseline and outcomes are in Desk 1. All 4 sufferers obtained lymphodepletion earlier than infusion. 4 sufferers obtained a dose of 0.5 × 106 (±20%) CAR T cells/kg. These cells had been sourced both autologous (the primary) or from earlier transplantation donors (sufferers 2, 3, and 4).
A CONSORT diagram of the medical trial. B Swimmer plot (n = 4) demonstrating the incidence of CRS and ICANS after the infusion of CAR T cells. Every bar represents a person affected person. The severity is indicated by completely different colours. C CRS and ICANS administration with etanercept in affected person 2, n = 1. D The height numbers of CAR T cells in PB of sufferers in keeping with the grade of CRS, GVHD, and ICANS. Every dot represents one affected person, n = 4. E Kinetics of serum cytokines for all sufferers within the first 30 days after CAR T-cell infusion. Every line represents one affected person, n = 4. F Kinetics of monocyte, neutrophil, and thrombocyte counts in PB at completely different time factors post-infusion. Every line represents one affected person, n = 4. CRS cytokine launch syndrome, ICANS immune effector cell-associated neurotoxicity syndrome, GVHD Graft-versus-Host Illness. Supply information are offered within the Supply Information file.
Three sufferers skilled grade 1–2 CRS and one skilled grade 4. The median onset was day 1 post-infusion (vary, 1–3), and the median length was 11 days (vary, 6–20) (Fig. 7B, D). One affected person additionally skilled grade 2 CRS on days 2–8 post-second-infusion (Fig. 7B). Two sufferers obtained tocilizumab, and all 4 obtained corticosteroids and nonsteroidal anti-inflammatory medicine for CRS. The affected person with grade 4 CRS offered with fever, dyspnea, and hypotension and didn’t reply to tocilizumab or corticosteroids, and developed dose-limiting toxicity (DLT), and his serum IL-6, IFN-γ, and TNF-α ranges had been comparatively greater than others (Fig. 7E). Because of elevated TNF-α, etanercept was administered, leading to aid of signs (Fig. 7C). The subsequent two sufferers additionally obtained etanercept to regulate CRS.
Two sufferers skilled grade 1 neurotoxicity on day 5 that lasted 1–2 days (Fig. 7B, D). Two sufferers developed grade 1–2 graft-versus-host illness (GVHD) (Fig. 7D). All 4 sufferers skilled grade 2–4 neutropenia, monocytopenia, and thrombocytopenia (Fig. 7F). One affected person developed sepsis, efficiently handled with antibiotics.
The examine has been halted as a result of security issues (DLT and extreme an infection), and the choice was made by the IRB primarily based on “Termination Standards” within the examine protocol.
Exercise of C-JUN-overexpressing CD33 CAR T cells in sufferers with r/r AML
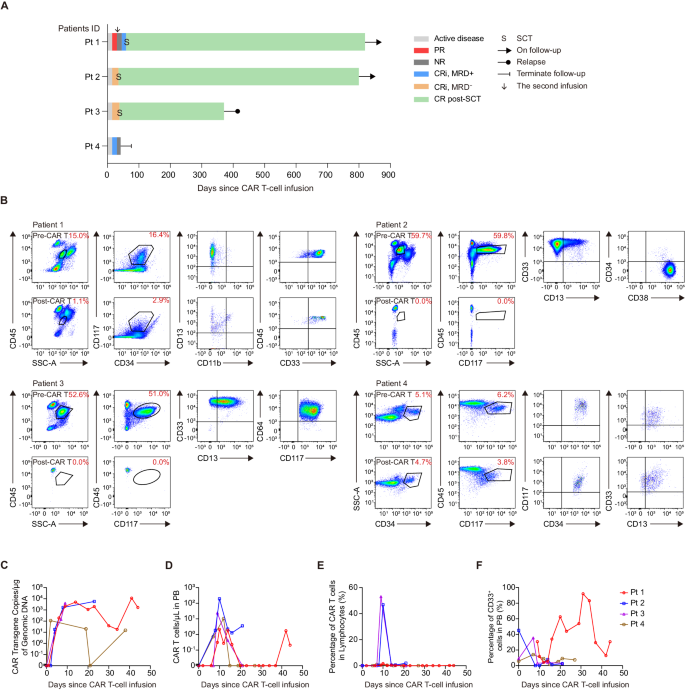
One affected person underwent tumor burden evaluation after lymphodepletion and earlier than infusion, and lymphodepletion didn’t utterly get rid of tumor cells (16.86% in BM). The remaining three sufferers refused to bear a biopsy after lymphodepletion. Two sufferers with excessive baseline tumor burden achieved minimal residual disease-negative (MRD−) CR with incomplete hematologic restoration (CRi) on day 15, which was maintained till day 30 (Fig. 8A, B). Affected person 4 achieved MRD+ CRi on day 15 however didn’t obtain CR on day 30 (Fig. 8A, B). Affected person 1 achieved partial remission (PR) on day 15, however illness rebounded and was assessed as no response (NR) on day 30. This affected person underwent a second infusion and achieved MRD+ CRi after 15 days (Fig. 8A, B). We additionally noticed myelosuppression after CD33 CAR T cell remedy (Supplementary Fig. 9A). The CR/CRi sufferers underwent SCT at 35, and 38 days after infusion or at 29 days after the second infusion, respectively (Fig. 8A). By cutoff, two sufferers remained disease-free and alive for greater than 2 years. One affected person skilled a transient CD33+ illness relapse after 1 12 months, the affected person was subsequently handled with Selinexor and Artesunate, reached remission, and underwent transplantation (Supplementary Fig. 9B).
A Swimmer plot (n = 4) displaying affected person responses. Every bar represents a person affected person. Responses had been decided on day 15 and day 30 and had been indicated by completely different colours. Bars with strong arrows signify sufferers in an ongoing follow-up. B Dot plots indicating blasts within the BM samples earlier than CAR T-cell infusion (affected person 1 at 21 days, affected person 2 at 72 days, affected person 3 at 12 days, affected person 4 at 12 days) and on day 30 post-infusion (besides affected person 1 who was analyzed on day 15 publish the second CAR T-cell infusion), as decided by movement cytometry. Kinetics of CAR vector transgene copies (C), CAR T cell counts (D), proportion of CAR T cells on lymphocytes (E), and proportion of CD33+ cells in PB (F) of particular person sufferers at completely different time factors post-infusion, as decided by quantitative PCR and movement cytometry. Every line represents one affected person, n = 4. CR full remission, CRi full remission with incomplete hematologic restoration, DLT dose-limiting toxicity, MRD minimal residual illness, NR no response, PR partial remission, Pt affected person, SCT stem cell transplantation. Supply information are offered within the Supply Information file.
CAR T cells expanded considerably in all sufferers. The 2 single-dose CRi sufferers had peak transgene ranges of 5744 and 3966 copies/μg, respectively (Fig. 8C). The NR affected person had a comparatively low peak transgene degree (109 copies/μg). The opposite affected person had a peak transgene degree of 4999 copies/μg after the primary infusion and a peak degree of 11,350 copies/μg after the second infusion (Fig. 8C). The 2 single-dose CRi sufferers had peak CAR T-cell counts of 192 and 23.5 cells/μl, respectively. The NR affected person had a peak rely of 10.3 cells/μl. The twice-infusion affected person achieved a peak rely of two.12 and 1.73 cells/μl after the primary and second infusion, respectively (Fig. 8D). As well as, the single-dose CRi sufferers had the next proportion of CAR T cells (46.8% and 53.0%) amongst lymphocytes (Fig. 8E). As anticipated, peripheral CD33+ myeloid cells decreased (Fig. 8F).