Leukemic cells induce international adjustments within the stromal cells upon in vitro co-culture
Now we have beforehand reported that stromal cells induce drug resistance in leukemic cells utilizing an in vitro stromal-leukemic cell co-culture system2,3,23. To review the impact of leukemic cells on these stromal cells, we used the identical system the place a standard stromal cell line (HS-5) was co-cultured with leukemic cells (NB4) for 48 h and a worldwide gene expression profile (GEP) of the stromal cell was studied (Fig. 1a). The stromal cells had been flow-characterized utilizing CD73 (a marker for stromal cells) for enumeration of residual leukemic cells. The co-cultured stromal cells had been proven to be greater than 90% purity (CD73 +) earlier than transcriptomic evaluation. (Supplementary Fig. 1a, b). The GEP urged that the transcriptomic profile of stromal cells in co-culture differed from the management (stromal cells alone), indicating that the leukemic cells alter the stromal cells throughout co-culture. We noticed that round 9046 genes had been differentially regulated (Fig. 1b). Additional pathway evaluation of the GEP utilizing the DAVID database urged an enrichment of bone formation and mobile differentiation of stromal cells in the direction of osteoblastic lineage. Together with these pathways, there was vital enrichment for Wnt signaling, BMP signaling, cell adhesion molecules and cytokines (Fig. 1c). These findings had been additional confirmed by GSEA evaluation, the place enrichment of osteoblast differentiation was famous (Fig. 1d) and the differential expression of genes was represented (Fig. 1e). These information had been additionally validated by an impartial RNA sequencing experiment by which the stromal cell line was co-cultured with one other leukemic cell line (U937) (Supplementary Desk S2).
(a) Schematic illustration of co-culture between stromal cells (HS-5) and leukemic cells (NB4). The stromal cells had been subjected to GEP evaluation on the finish of 48 h. (b) Volcano plot exhibiting the differentially regulated genes in stromal cells upon leukemic cell interplay. (c) DAVID pathway evaluation confirmed an enrichment of osteoblast differentiation-related genes and Wnt signaling pathways. (d) Gene Set Enrichment Evaluation (GSEA) evaluation indicated an enrichment of osteoblast differentiation pathway in stromal cells upon co-culture. (e) Warmth map demonstrates osteoblast-associated (differentially regulated) genes enriched in stromal cells upon co-culture with leukemic cells (n = 3).
Stromal cells are primed for osteoblast differentiation by the leukemic cells.
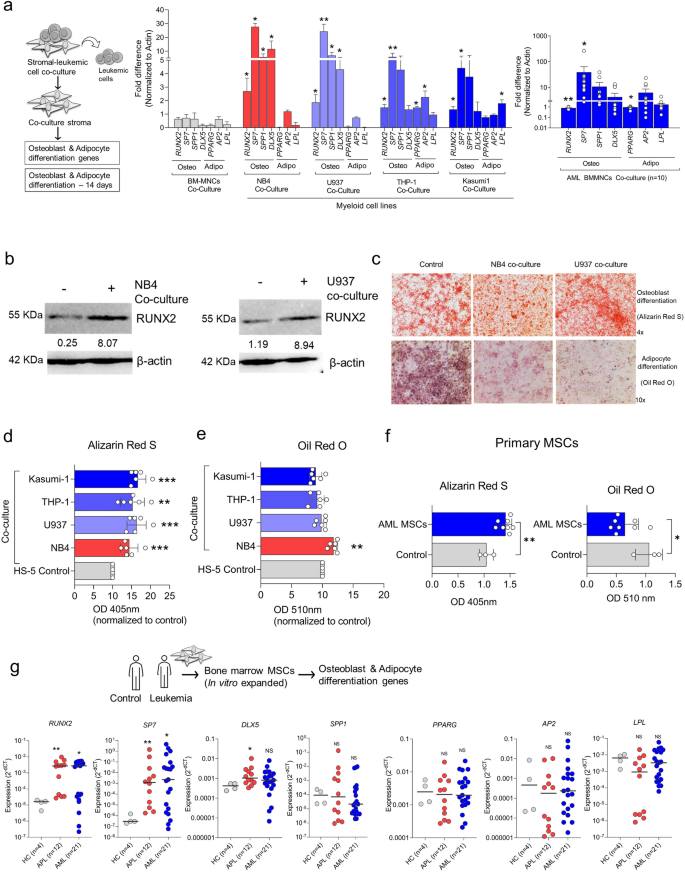
To validate the GEP evaluation, we carried out QPCR and western blot assays to confirm genes concerned in osteoblast differentiation. We famous a major upregulation of genes concerned in osteoblast differentiation (RUNX2, SP7 & DLX5) with out altering the expression of genes concerned in adipocyte differentiation (PPARG, AP2 & LPL)) when the stromal cells had been co-cultured with myeloid leukemic cell strains and first cells (Fig. 2a; Supplementary Fig. 1c). The expression of those genes was additionally evaluated in leukemic cells as a damaging management (Supplementary Fig. 1d). In corroboration with QPCR evaluation, western blot additionally demonstrated a rise in RUNX2 (osteogenic transcription issue) in stromal cells throughout co-culture with leukemic cells (NB4 and U937) (Fig. 2b). For additional validation utilizing a practical assay, the stromal cells had been co-cultured with totally different leukemic cells and subjected to differentiation assay. The stromal cells co-cultured with leukemic cell strains demonstrated considerably elevated osteoblast differentiation, as indicated by Alizarin Pink S staining (Fig. 2c,d). Nevertheless, there have been no adjustments in differentiation in the direction of adipocytes, as proven by Oil Pink O staining (Fig. 2e), with minimal heterogeneity throughout the cells. In distinction, the first leukemic cells couldn’t prime the stromal cell line (HS-5) in the direction of osteoblast/ adipocyte differentiation, not like leukemic cell strains (Supplementary Fig. 2a).
(a) QPCR evaluation of osteogenic (RUNX2, SP7, SPP1 & DLX5) and adipogenic genes (PPARG, AP2 & LPL) in stromal cells co-cultured with totally different myeloid leukemic cell strains (NB4, U937, THP-1 and Kasumi-1) and first AML cells AML BM-MNCs (n = 10) compared to wholesome management bone marrow mono-nuclear cells (BM-MNCs) cells co-cultured for 48 h and expression of genes had been evaluated by real-time PCR assay (n = 4). Statistical significance was calculated compared to management BM-MNCs co-culture. (b) Immunoblot demonstrates an elevated expression of RUNX2 (an osteogenic transcription issue) within the stromal cells co-cultured with leukemic cells (NB4 and U937) for 48 h. (c) Microscopic image demonstrating the differentiation of stromal cells into osteoblast and adipocytes after co-culturing with leukemic cells NB4 and U937 cells for 48 h, adopted by differentiation of stromal cells for 14 days, and the cells had been stained with Alizarin Pink S (osteoblast) and Oil Pink O (adipocytes). The dimensions bar is 50 µm. (d) Quantifying osteoblast differentiation of stromal cells submit co-culture with totally different myeloid leukemic cells and their colorimetric detection of Alizarin Pink S staining utilizing absorbance at 405 nm (n = 5). (e) Quantifying adipocyte differentiation of stromal cells submit leukemic cell co-culture adopted by colorimetric detection of Oil Pink O staining utilizing absorbance at 510 nm (n = 5). (f) Quantifying the osteoblast and adipocyte differentiation potential of major AML MSCs (n = 8). (g) QPCR evaluation of osteogenic and adipogenic genes in major stromal cells expanded from the bone marrow of wholesome management (HC) (n = 4), APL (n = 12), AML (n = 21) (*—P = 0.05, **—P = 0.001, ***—P = 0.0001, ns—not vital).
To substantiate our findings from cell line fashions, we used major stromal cells expanded from the bone marrow of sufferers with acute leukemia (APL and AML). Subjecting these major stromal cells to differentiation assays, the AML stroma demonstrated a major enhance in osteoblast differentiation with a simultaneous lower in adipocyte differentiation potential (Fig. 2f), suggesting that the stromal cells are primed to osteoblast differentiation by the leukemic cells. We additionally famous that osteoblast differentiation-associated genes (RUNX2, SP7 & DLX5) had been considerably upregulated throughout these cells. Nevertheless, the adipocyte differentiation-associated genes displayed no change within the expression of those genes when in comparison with wholesome controls (Fig. 2g). Further analysis of osteocalcin (a marker for bone formation) ranges suggests a considerably elevated expression in stromal cells post-leukemic co-culture (Supplementary Fig. 2b). Screening of cell adhesion molecules within the co-cultured stromal cells additionally suggests adhesion molecules associated to osteoblast lineage had been upregulated (Supplementary Fig. 3).
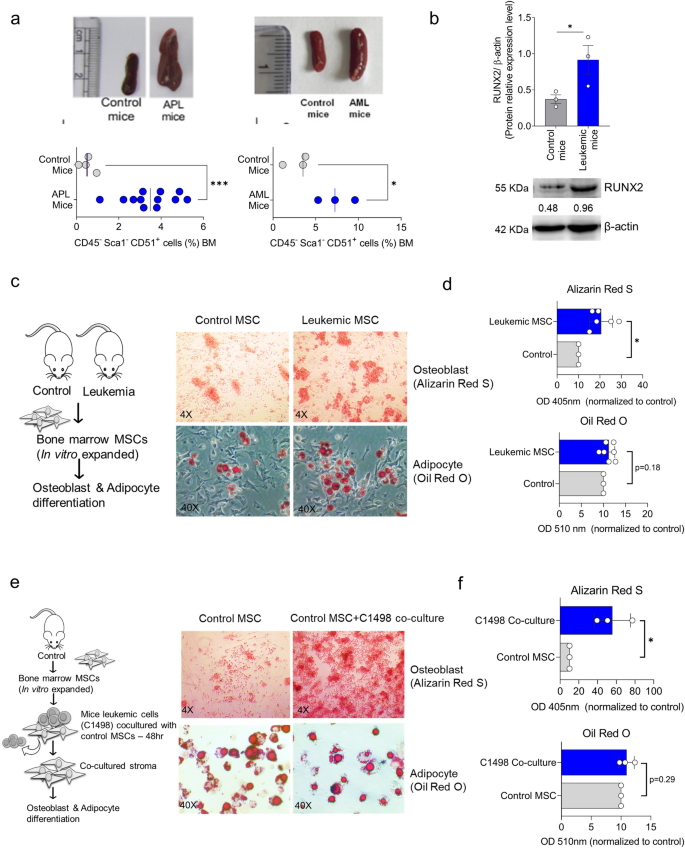
Our in vitro findings had been additional validated in vivo utilizing two totally different leukemic mice fashions [MRP8-PML-RAR transgenic APL mice model (FVB/N strain) and C1498 murine AML cell line model (C57BL/6 strain)]. There was a major enhance in osteoblast proportion (CD45− Sca1− CD51+) within the bone marrow of leukemic mice in comparison with the management (Fig. 3a). The in vitro expanded mice stroma additionally demonstrated an elevated expression of the osteoblast marker RUNX2, as proven by western blot evaluation (Fig. 3b). The differentiation potential of mouse stromal cells exhibited a major enhance in osteoblast differentiation in comparison with the management, whereas the extent of adipocyte differentiation was not substantial (Fig. 3c,d). As well as, we co-cultured the mouse leukemic cell line (C1498) with stromal cells expanded from management mice and evaluated their differentiation potential. Apparently, the management stromal cells co-cultured with leukemic cells additionally confirmed an elevated osteoblast differentiation (Fig. 3e,f). The info recommend that leukemic cells prime the stromal cells to decide to differentiation in the direction of the osteoblast lineage.
(a) APL and AML mice mannequin exhibiting splenomegaly in comparison with management in addition to osteoblast proportion (CD45− Sca1− CD51+) cells in bone marrow in comparison with management mice APL (n = 12) AML (n = 3) on day 28. (b) Immunoblot evaluation with relative quantification of RUNX2 in bone marrow stromal cells expanded from management and leukemic mice (n = 3). (c) Workflow and microscopic image of Alizarin Pink S staining (osteoblast differentiation) and Oil Pink O staining (adipocyte differentiation) of the expanded stroma after 14 days of differentiation of leukemic MSCs and management MSCs. The dimensions bar is 50 µm. (d) Quantification of Alizarin Pink S and Oil Pink O staining demonstrated a considerable enhance in osteoblast differentiation (Alizarin Pink S staining). In distinction, the adipocyte differentiation (Oil Pink O) confirmed an insignificant variation in management MSCs and leukemic MSCs. e) The MSC expanded from management mice and was co-cultured with mice leukemic cell line C1498 for 48 h, adopted by induction for 14 days. The dimensions bar is 50 µm. The co-cultured MSCs confirmed an elevated osteoblast differentiation with out vital change in adipocyte differentiation, as demonstrated within the microscopic image and colorimetric evaluation (f). (*—P = 0.05, ns—not vital).
Osteoblasts differentiated stroma gives superior safety to leukemic cells in comparison with undifferentiated or adipocyte differentiated stromal cells
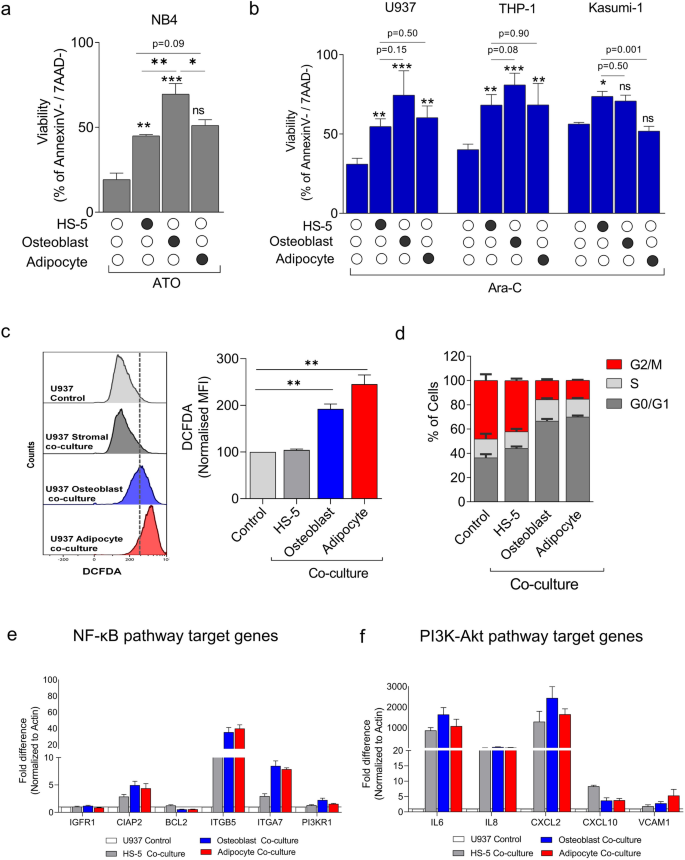
Since we noticed that leukemic cells prime the stromal cells in the direction of osteoblastic lineage, we explored the affect of those primed stromal cells by differentiating them into osteoblast and adipocyte lineages and evaluating their chemo-protective capacity. In the direction of this, stromal cells (HS-5) had been differentiated into osteoblast and adipocytes, adopted by co-culture with leukemic cells within the presence of chemotherapeutic medication similar to arsenic trioxide (ATO) for promyelocytic cells NB4, daunorubicin (DNR) and cytarabine (Ara-C) for myeloid leukemic cells (U937, THP-1 and Kasumi-1). The viability was assessed on the finish of 48 h (Fig. 4a,b). The osteoblast differentiated stroma gives enhanced safety in leukemic cells towards ATO and Ara-C in comparison with adipocyte-differentiated and undifferentiated stromal cells. Although the distinction was not statistically vital, the pattern towards enhance was constant and reproducible throughout the leukemic cells evaluated. Nevertheless, there was a marginal heterogeneity throughout cells, with a reasonable impact within the presence of DNR. (Supplementary Fig. 4). The co-cultured leukemic cells had an elevated ROS, and so they had been within the G0/ G1 quiescence section of the cell cycle when co-cultured with osteoblast and adipocyte differentiated stroma in comparison with undifferentiated controls (Fig. 4c,d). These co-cultured leukemic cells probably rely upon cell survival signaling pathways like NF-κB and PI3K-Akt signaling pathway (Fig. 4e,f) demonstrating an elevated expression of its goal genes, which is according to our earlier findings in APL and AML2,3. The outcomes point out that leukemic cells, upon stromal co-culture, alter the stromal cells in the direction of the osteoblast lineage to create a drug-resistant area of interest for survival.
(a) Osteoblast differentiated stroma induces a major protecting impact towards arsenic trioxide ATO (2 µM) in malignant promyelocytes (NB4) in comparison with undifferentiated stroma (HS-5) and adipocyte differentiated cells (n = 5). Statistical significance was calculated by evaluating the viability of leukemic cells handled with drug alone and leukemic cells co-cultured with totally different feeder layers, adopted by comparability with osteoblast differentiated and undifferentiated cells (b) Osteoblasts and adipocytes induce a major protecting impact towards cytarabine (Ara-C) (400 ng/ml) in leukemic cells (U937, THP-1 and Kasumi-1) whereas adipocytes give marginal safety in comparison with monoculture. Viability was assessed in leukemic cells utilizing an Annexin V/7AAD package by stream cytometry after 48 h of drug remedy (n = 5). (c) Analysis of ROS ranges in leukemic cells submit 24 h of co-culture with stromal cells utilizing DCFDA by stream cytometry. (n = 3). (d) Cell cycle evaluation in leukemic cells submit 24 h of co-culture utilizing propidium iodide staining by stream cytometry (n = 6). e) QPCR evaluation of NF-κB pathway goal genes in leukemic cells submit 48 h of co-culture with differentiated and undifferentiated stromal cells in comparison with management U937 alone (n = 3). f) QPCR evaluation of PI3K-Akt pathway goal genes (n = 3). (*—P = 0.05, **—P = 0.001, ***—P = 0.0001, ns—not vital).
Wnt signaling is dysregulated in stroma upon co-culture with leukemic cells
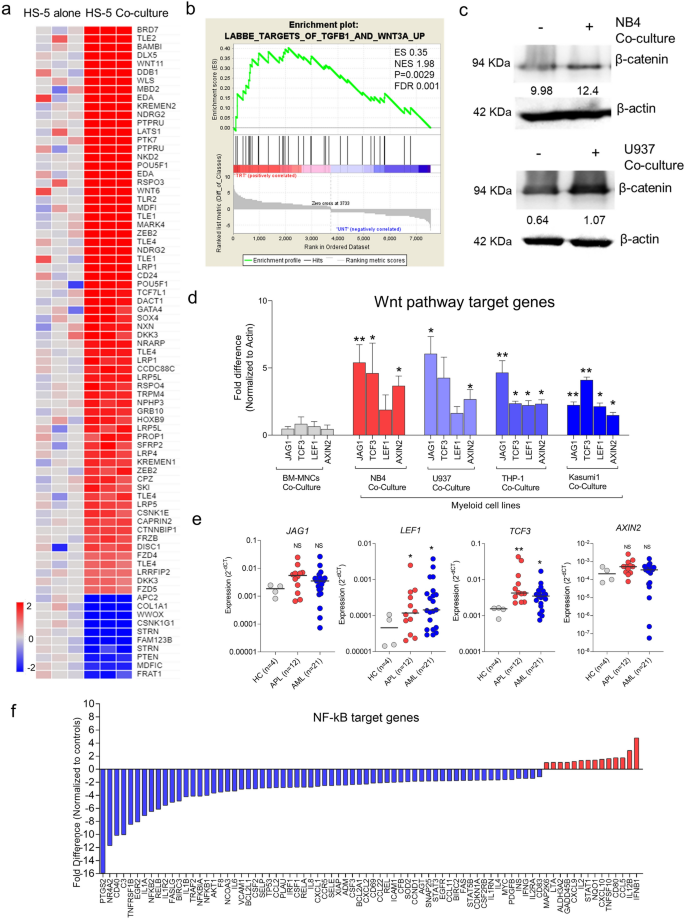
A earlier report means that the osteoblast priming of stromal cells by leukemic cells primarily includes BMP signaling14. Our gene expression profiling additionally confirmed that Wnt signaling was enriched in stromal cells upon co-culture with leukemic cells (Fig. 5a,b). It has been established that Wnt signaling is important for stroma cells to decide to osteoblast lineage within the presence of BMP signaling and, in its absence, stromal cells decide to chondrocyte lineage24. Subsequently, it was essential to guage the position of canonical Wnt signaling in stromal cells in the direction of drug resistance. The Wnt signaling is activated in co-cultured stromal cells by the stabilization of β-catenin protein, which was confirmed by western blot evaluation (Fig. 5c), and its goal genes (JAG1, LEF1, TCF3 & AXIN2) was confirmed by QPCR in stromal cells upon co-culture with totally different myeloid leukemic cells (Fig. 5d). This in vitro information was additional validated in major stromal cells from APL and AML, demonstrating a major enhance in expression of LEF1 and TCF3, with a reasonable enhance in JAG1 & AXIN2 suggestive of variation may very well be particular person and context-dependent (Fig. 5e). Proof means that Wnt activation can antagonize the NF-κB signaling pathway25,26. To judge this, we carried out an NF-κB goal gene array and located that many of the NF-κB goal genes had been downregulated in stromal cells upon co-culture with leukemic cells (NB4) (Fig. 5f). These findings recommend that the Wnt signaling pathway is activated in stromal cells upon co-culture with leukemic cells.
(a) Heatmap exhibiting dysregulation of Wnt signaling in stromal cells upon co-culture with leukemic cells from GEP evaluation validated by GSEA enrichment evaluation (b). (c) Immunoblot exhibiting stabilization of β-catenin protein within the stromal cells upon co-culture with leukemic cells (NB4 and U937) for 48 h. (d) QPCR validation of Wnt signaling genes in stromal cells co-cultured with totally different leukemic cell strains for 48 h (n = 3). (e) QPCR evaluation of Wnt signaling genes in major MSCs from APL (n = 12) and AML (n = 21) compared to HC (n = 4). (f) Actual-time PCR array for NF-κB goal genes exhibiting a down-regulation of genes managed by NF-κB in stomal cells co-cultured with leukemic cells (NB4) (n = 3) for 48 h. (*—P = 0.05, **—P = 0.001, ns—not vital).
Inhibition of Wnt signaling in vitro and in vivo reduces the drug resistance phenotype in AML
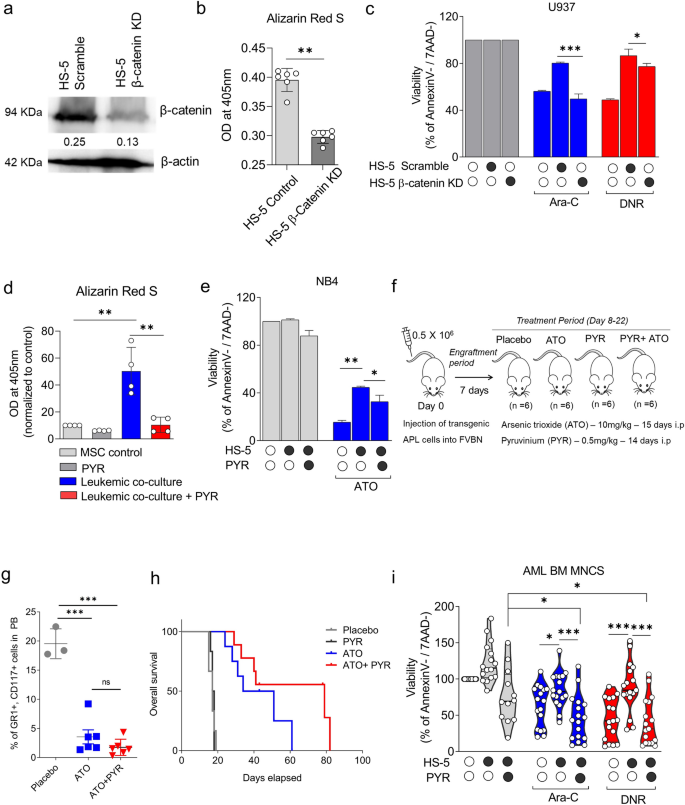
Since Wnt signaling was upregulated in co-cultured stromal cells, we hypothesized it might induce drug resistance in leukemic cells. We knocked down the β-catenin RNA by the shRNA technique (Fig. 6a). When the HS-5 cells had been knocked down with β-catenin, there was no change within the proliferation of cells (Supplementary Fig. 5), and there was a major lower in osteoblast differentiation upon induction, as evidenced by much less Alizarin Pink S staining (Fig. 6b). These cells have a considerably much less cytoprotective impact on leukemic cells from AML chemotherapeutic medication (Ara-C & DNR) when in comparison with scramble HS-5 (Fig. 6c). This was additional confirmed by Wnt chemical inhibitors. We used an anthelmintic drug, pyrvinium pamoate (PYR), a identified Wnt signaling pathway inhibitor27. PYR remedy inhibits Wnt signaling in vitro and in vivo (Supplementary Fig. 6) and reduces osteoblast differentiation in stromal cells throughout leukemic co-culture (Fig. 6d), thereby reverses the drug resistance in leukemic cells (Fig. 6e). PYR pre-treatment in stromal cells abrogated the drug resistance and overcomes the stroma-mediated protecting impact in myeloid leukemic cells U937, THP-1 and Kasumi-1 as evidenced by Annexin V apoptosis assay and time-lapse microscopic evaluation in U937 demonstrates the elevated apoptotic cells within the mixture group (PYR + Ara-C) when in comparison with PYR alone at 48 h (Supplementary Fig. 7). The info was additional confirmed by trypan blue staining viability evaluation (Supplementary Fig. 8). PYR additionally confirmed an additive impact with ATO, Ara-C & DNR in NB4 and U937, respectively, with a synergy rating of lower than 10 within the presence or absence of stromal cells when the synergy was evaluated utilizing the SynergyFinder device28 (Supplementary Fig. 9). The info validates our speculation that the Wnt signaling is upregulated throughout leukemic cell interplay, which confers drug resistance to leukemic cells towards chemotherapeutic brokers.
(a) Immunoblot evaluation demonstrating the knockdown (KD) efficacy of β-catenin in stromal cells in comparison with management. (b) Alizarin Pink S staining demonstrates much less osteoblast differentiation in HS-5 β-catenin KD cells (n = 6). (c) HS-5 β-catenin KD cells reversed the drug resistance in leukemia cells when co-cultured with the knockdown cells in comparison with scrambled HS-5 cells as proven by annexin V apoptosis assay for 48 h (n = 4). (d) Inhibition of the Wnt signaling pathway utilizing a identified Wnt inhibitor and an anthelmintic drug, pyrvinium pamoate (PYR), overcomes osteoblast differentiation, as evident by Alizarin Pink S staining on MSCs co-cultured with leukemic cells U937 and differentiated to osteoblast for 14 days with PYR at focus 0.1 µM (n = 4). (e) PYR pre-treatment reversed drug resistance in leukemic cells (NB4) upon pre-treatment with PYR, and the viability of leukemic cells was assessed after 48 h (n = 3). (f) Workflow of APL mice mannequin on remedy with PYR and ATO. (g) PYR, when mixed with ATO, diminished the leukemic burden (GR1+ CD117+ cells) within the APL mice mannequin as evaluated by stream cytometry evaluation in peripheral blood on day 28. (h) Mixture of PYR with ATO in APL mice extended the survival of the mice in comparison with placebo or both of the brokers handled alone (n = 6). For survival, a log-rank take a look at was employed to calculate significance. (i) PYR pre-treatment successfully overcomes the stromal cell-mediated drug resistance together with Ara-C and DNR in AML BM MNCs (n = 17). Every dot represents a affected person pattern. (*—P = 0.05, **—P = 0.001, ***—P = 0.0001, ns—not vital).
Lastly, our in vitro outcomes had been validated in vivo utilizing a transgenic APL mice mannequin, the place an elevated osteoblast proportion was noticed in leukemic mice. Combining PYR with the usual chemotherapeutic drug ATO (Fig. 6f) might considerably scale back the tumor burden in APL mice (Fig. 6g) and enhance survival in comparison with ATO or PYR alone remedy (Fig. 6 h). The efficacy of PYR was additionally evaluated in mice earlier than or after leukemic cell engraftment. In keeping with our in vitro findings, the APL mice handled with PYR resulted in decreased leukemic burden (Supplementary Fig. 10a, b) in addition to osteoblast proportion in bone marrow (Supplementary Fig. 10c). As well as, the PYR remedy additionally extended the survival of mice from a median of 23 days (placebo) to 34 days (when PYR was administered earlier than leukemic engraftment) and by 29 days (when PYR was administered submit leukemic engraftment together with ATO) (Supplementary Fig. 10d). Nevertheless, the mixture and the dose utilized for the experiments weren’t poisonous to the conventional mice, as evidenced by their survival over three months (Supplementary Fig. 10e). As well as, PYR was additionally capable of overcome the stromal cell-mediated resistance in major AML cells together with cytarabine and daunorubicin (Fig. 6i).
Impression of lymphoid leukemic cells on stromal cell differentiation and drug resistance
To judge the potential of lymphoid leukemic cells within the differentiation of stromal cells, we repeated all of the experiments in lymphoid leukemic cell strains SUP B15 (B-ALL), Jurkat E6.1 (T-ALL) and first ALL cells. Like myeloid leukemic cells, the lymphoid leukemic cells additionally exhibited a major upregulation of osteoblast differentiation-associated genes throughout co-culture (Supplementary Fig. 11a,b), together with elevated expression of osteocalcin (Supplementary Fig. 11c). As well as, the practical assay, stromal cell differentiation with ALL cells co-cultured stroma, exhibited a major differentiation in the direction of osteoblast/ adipocyte lineage (Supplementary Fig. 11d,e). Nevertheless, the first ALL cells couldn’t induce differentiation (Supplementary Fig. 11f). Equally, the first stroma from ALL sufferers didn’t present a rise in osteoblast differentiation. Nonetheless, it considerably diminished adipocyte lineage regardless of a considerable upregulation of osteoblast differentiation genes (RUNX2, SP7 & DLX5) (Supplementary Fig. 11g,h).
In keeping with AML information, the ALL-co-cultured stroma additionally confirmed a major upregulation of Wnt goal genes (TCF3, LEF1 & AXIN2) with elevated expression of AXIN2 in SUP B15 (Supplementary Fig. 12). Nevertheless, the differentiated stromal cells had a variable impact on inducing resistance in each ALL cell strains used on this research (Supplementary Fig. 13). Conversely, within the major ALL cell co-culture, PYR pre-treatment of stromal cells overcomes the resistance exhibited by the stromal cells (Supplementary Fig. 14). Collectively, the info demonstrates that lymphoid leukemic cells, in distinction to myeloid leukemic cells, have an analogous however much less outstanding priming impact on stromal cells to induce the osteoblast differentiation bias and creating drug resistance area of interest.