Chemokine evaluation of Human GBM favors MDSC recruitment over lymphocyte infiltration
Upon examination of human glioma tumors for CD3 protein expression offered by way of the Human Protein Atlas, we discovered that > 80% of tumor specimens evaluated are detrimental for lymphocyte infiltrates (Supplementary Fig. S1a), corroborating prior literature describing the lymphocyte-replete nature of those tumors20. To determine if a paucity of lymphocyte chemotactic elements is a contributing facet, human glioma samples had been screened through chemokine proteome array. Of those, CXCL4, CXCL7, CXCL8 (IL-8), CXCL16, LCF (IL-16), TIG-2, and midkine (MDK) emerged as probably the most considerable secreted chemokines detected (Fig. 1a, b), the place these ligands play a big function in recruiting myeloid-derived suppressor cells within the context of gliomagenesis21. Notably, chemotactic elements that favor lymphocyte recruitment had been poorly expressed, together with CXCL9 (MIG) and MIP-1α/β (CCL3/CCL4) (Fig. 1a, b). CXCL9 is a robust attractant identified to induce the migration, differentiation, and activation of cytotoxic T lymphocytes22. Expression of CXCL9 has been proven to correlate with anti-tumor immune exercise and is predictive of response to ICB in a number of stable tumors23. We hypothesized that AAV supply and restoration of CXCL9 as a “call-and-receive” sign for T lymphocytes throughout the tumor microenvironment (TME) would improve their recruitment and infiltration within the tumor.
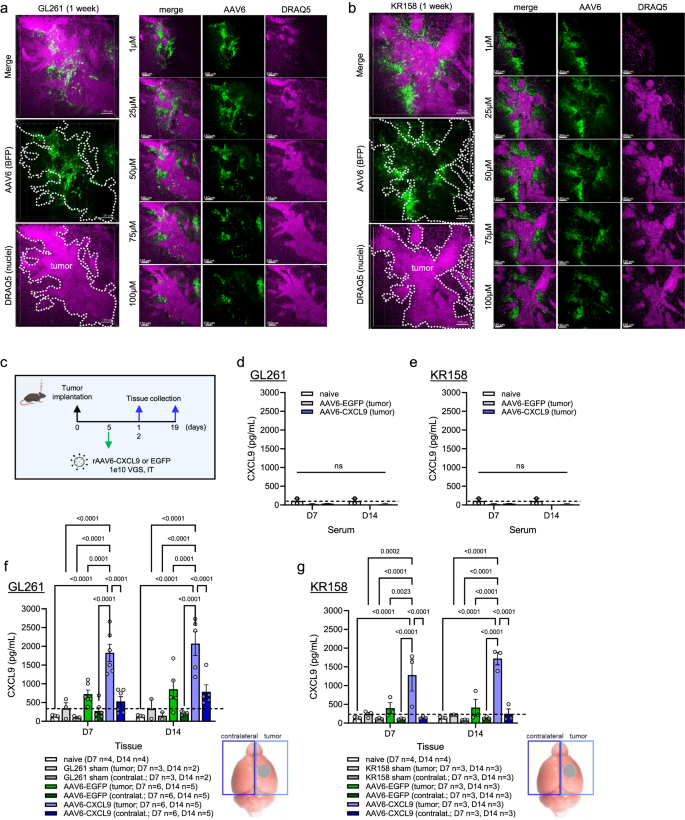
a Immunoblots of GBM samples exhibiting sign depth of 31 chemokines (n = 8). CXCL9 (undetected) is printed in pink. b Field-whisker plots of cumulative relative protein expression of immunoblots proven in panel (a). c Recombinant AAV6 vector design encoding CXCL9 and the fluorescent reporter gene RFP. d 3D IHC of RFP-labeled GL261 tumor tissue collected 1 week following AAV6-EGFP injection. The highest row depicts 3D rendering at 10 x magnification, scale bar 200 µm. AAV6 transduced cells are proven in inexperienced, GFAP in pink, RFP+ tumor cells in grey, and DAPI nuclear stain in darkish blue. 2nd and third rows depict 2D digital zoom as outlined by the yellow dashed line within the prime row to reinforce mobile decision. Voxel-based co-localization between AAV6 and GFAP (2nd row) and AAV6 and tumor cells (third row) is proven as a separate channel (yellow or pink). Consultant photos chosen from n = 5. e 3D IHC of AAV6-EGFP transduction in age-matched naïve management mice. The highest row depicts 3D rendering at 10x magnification, scale bar 200 µm. AAV6 transduced cells are proven in inexperienced, GFAP in pink, and DAPI nuclear stain in darkish blue. 2nd row depicts 2D digital zoom as outlined by the yellow dashed line within the prime row to reinforce mobile decision. Voxel-based co-localization between AAV6 and GFAP is proven as a separate channel (yellow). Consultant photos chosen from n = 3. f Field-whisker quantitative abstract of voxel-based AAV6 co-localization with both tumor (GL261, n = 5) or astrocytes in every tumor-bearing (n = 3) and naïve mice (n = 3). g Field-whisker plot of circulate cytometry quantitation of AAV6 (EGFP + ) co-localization with both tumor cells (GL261, RFP + ) or astrocytes (GFAP + , RFP-) at 3-, 5-, and seven days submit AAV6 transduction as illustrated within the schematic. Two-way extraordinary ANOVA statistical evaluation carried out evaluating % transduction between tumor and astrocytes throughout matched time factors, n = 6 per time level. P-values ≤ 0.05 are thought-about statistically vital. Field-whisker plots show the field starting from the primary to the third quartile, the middle median worth, and the whiskers lengthen from every quartile to the minimal and most values. Supply information are offered as a Supply Knowledge File.
Traditionally, focused transduction of most cancers with AAV has confirmed difficult regardless of elegant efforts in capsid evolution research and capsid engineering. To realize enough transgene expression, transduction of both tumor cells or tumor-associated stroma is probably going essential, the place intra-tumoral supply would decrease the potential for systemic toxicities and reduce off track homing of T cells. To determine an applicable AAV capsid for focusing on glioma tumors, we carried out an in vitro capsid display screen. Enhanced inexperienced fluorescent protein (EGFP) was encoded into an AAV2 single stranded vector, using the non-cell-autonomous, constitutively lively CBA promoter to drive transgene expression (AAVn-EGFP). These constructs had been pseudotyped into 29 distinctive capsids as beforehand described24. Transduction in 15 distinctive glioma fashions, together with major human25 and murine xenografts, was assessed through EGFP relative fluorescence expression (Supplementary Fig. S1b). AAV6 was chosen for additional examination because it demonstrates reasonable to excessive transduction in almost all fashions examined and is additional substantiated by wonderful CNS transduction in different research24,26. AAV6 capsids encoding every CXCL9, EGFP, blue fluorescent protein (BFP), and empty vector management (Fig. 1c, Supplementary Fig. S1c–e) had been designed for additional testing. Quantitative circulate cytometric analysis of EGFP expression in three distinct syngeneic murine GBM fashions 72 h following AAV6-EGFP transduction reveals reasonable transduction of GL261 and KR158, with > 25% of cells constructive for the transgene right now level, with decrease transduction noticed in CT-2A cells (<20%) (Supplementary Fig. S1f).
AAV6 transduces tumor-reactive astrocytes in vivo in preclinical fashions of GBM
Whereas we display good focusing on of glioma cells in an in vitro setting, AAV6 has additionally been reported able to focusing on different cell populations within the CNS24,26. These research have largely been carried out within the context of naïve mice or in fashions of neurodegenerative illness, which can or will not be straight relevant to CNS malignancy. To outline AAV6 tropism in murine GBM in vivo, AAV6-EGFP (Supplementary Fig. S1c) was intratumorally injected into established intracranial KR158 and GL261 tumors. EGFP expression was detected 1 week following tumor transduction in each fashions (Supplementary Fig. S2a, b), nevertheless the morphological look and contiguous distribution of transduced cells counsel that AAV6 focused cells are doubtless tumor-associated, and never most cancers cells straight. Microglia and tumor-associated macrophages are reported to comprise a big mobile proportion of glioma tumors27, and so we sought to determine if AAV6 was focusing on both inhabitants. KR158 and GL261 had been implanted into CCR2RFPCX3CR1GFP (B6.129(Cg)-Cx3cr1tm1Litt Ccr2tm2.1lfc/JernJ) twin reporter mice, the place microglia may be recognized through GFP expression, and bone marrow-derived inflammatory cells through RFP expression28,29. Tumors had been evaluated by 3D IHC for viral transduction 1 week following intratumor injection with AAV6 encoding a BFP reporter (Supplementary Fig. S1d). In each tumor mannequin methods, we discovered no co-localization between BFP and both RFP or GFP, indicating that neither microglia nor tumor-associated macrophages are the principal goal of AAV6 transduction (Supplementary Fig. S3a, b). To evaluate the diploma of AAV6 transduction particularly in tumor cells we implanted mice with RFP-labeled KR158 or GL261 cells. Tumors had been evaluated by 3D IHC for viral transduction 1 week following intratumor injection with AAV6-EGFP. Resected tumors had been immuno-labeled in opposition to glial fibrillary acidic protein (GFAP) to detect astrocytes, one other candidate tumor-associated cell inhabitants. Each tumor fashions reveal a excessive diploma of overlap between GFAP (pink) and EGFP (inexperienced), with minimal overlap between tumor cells (grey) and EGFP (Fig. 1d, Supplementary Fig. S3c), indicating that EGFP-positive cells are doubtless astrocytes. Voxel-based co-localization algorithms to quantitate EGFP co-localization with every tumor or astrocytes verify astrocytes because the principal cell goal of AAV6 transduction, accounting for ~ 60–70% of EGFP-positive cells in each GL261 (Fig. 1f) and KR158 (Supplementary Fig. S3d) intracranial tumors. As a result of tumor presence can stimulate totally different activation states in astrocytes that will trigger them to be roughly prone to viral transduction30,31, we additionally evaluated CNS tropism of AAV6 in age-matched naïve mice. AAV6 was equally environment friendly at transducing astrocytes in naïve animals as proven by co-localization between GFAP immunostaining and EGFP transgene expression (Fig. 1e, f).
One of many distinctive options of AAV gene transduction is that it hardly ever integrates into the host genome. Following uncoating within the host nucleus, single-stranded genomes are transformed to double-stranded multimeric round concatemeric episomes32. As such, AAV transgene expression can persist long-term in post-mitotic cells. As a result of tumor cells bear speedy cell division, it might be potential that transgene expression is misplaced over time by way of sequential dilution of episomes handed right down to daughter cells. To discover this, we evaluated AAV6-EGFP transgene expression longitudinally throughout early time factors in mice harboring RFP labeled GL261 cells, which display the very best transduction effectivity in vitro (Supplementary Fig. S1f). Tumors had been resected 3, 5, and seven days following AAV6-EGFP intra-tumor injection as outlined in Fig. 1g, and EGFP transgene expression in tumors and astrocytes was measured by circulate cytometry. Even at early time factors, AAV6 predominantly transduces astrocytes recognized as GFAP + RFP- (70–80% EGFP+ cells), with restricted EGFP expression noticed in RFP+ tumor cells (<15%) (Fig. 1g, Supplementary Fig. S4a). By 7 days submit intratumor viral injection, lower than 5% of EGFP+ cells on common had been RFP+ tumor cells, the place astrocytes constantly comprised 70–80% of EGFP+ cells at every time level. These information point out that AAV6 extra selectively transduces astrocytes in vivo, with restricted and transient expression in tumor cells.
AAV6 transgene sign distribution and sturdiness in GBM
Subsequent, we examined the distribution of transgene sign in each the GL261 and KR158 tumor fashions through 3D IHC to raised perceive the avidity of AAV6 for tumor-associated versus distal astrocytes following direct intra-tumor injection. BFP transgene expression was noticed in a peritumoral sample in and across the tumor physique in each mannequin methods (Fig. 2a, b), redolent of glial scar formation present in human mind malignancies33. As CXCL9 is a small, secreted chemokine, we wished to find out if sign expression was nonetheless focal to the tumor or could possibly be detected in contralateral mind and/or systemically. The entire mind was collected at 1 and a pair of weeks following AAV6-CXCL9 or AAV6-EGFP intratumor injection as outlined in Fig. 2c. Cerebellar tissue was eliminated, and remaining tissue was dissected into the tumor containing and contralateral hemispheres. Serum was collected following peripheral blood attracts taken from the posterior vena cava. Mind tissue and serum had been additionally collected from non-transduced (sham) tumor controls, and naïve (non-tumor bearing) controls to determine CXCL9 baseline values. Serum ranges of CXCL9 following intratumor supply of AAV6-CXCL9 measured utilizing excessive sensitivity ELISA assay didn’t exceed these noticed in naïve controls (Fig. 2nd, e). Within the mind, elevated CXCL9 expression was selectively detected within the tumor bearing hemisphere transduced with AAV-CXCL9, with minimal sign noticed within the contralateral hemisphere in each GL261 and KR158 mannequin methods (Fig. 2f, g). Transgene CXCL9 expression seems to be secure, as sign depth was constant in AAV6-CXCL9 transduced tumors at each the 1- and 2-week time factors in every tumor mannequin (Fig. 2f, g). Of be aware, a small improve in CXCL9 expression was noticed in AAV6-EGFP management transduced GL261 tumors and could possibly be indicative of a gentle inflammatory response to AAV6, nevertheless these values weren’t discovered to be statistically vital. Collectively these information display that AAV6 intratumor supply of CXCL9 ends in focal and sturdy expression of encoded transgene, the place tumor-reactive astrocytes are the goal of AAV6 transduction.
3D IHC displaying geospatial distribution of AAV6 encoded transgene (BFP) in (a) GL621 and (b) KR158 tumors collected 1 week following in vivo transduction (inexperienced), n = 2 per mannequin, scale bar 150 µm. DRAQ5 nuclear dye (pink) is used to determine tumor borders, as outlined by the white dashed line. c Intra-tumor AAV6 therapy schematic for protein detection of AAV6 encoded CXCL9. ELISA detection of CXCL9 protein in serum at one and a pair of weeks following AAV6-CXCL9 or AAV6-EGFP management intracranial injection in (d) GL261 and (e) KR158 fashions, n = 3 per time level, per group. Age-matched naïve controls used to determine baseline CXCL9 ranges indicated by dashed black line. ELISA detection of CXCL9 protein in mind tissues remoted at 1 and a pair of weeks following AAV6-CXCL9 or AAV6-EGFP management intracranial injection in (f) GL261 and (g) KR158 fashions. Left and proper hemispheres lysed individually to mirror tumor-bearing and contralateral (focal and distal) sign detection. Statistical analyses carried out utilizing two-way ANOVA evaluation with Tukey’s a number of comparisons take a look at. Age-matched naïve mind, and sham (saline) injected tumors included as detrimental management and tumor baseline management, with the latter represented by the dashed black line, n values proven. P-values ≤ 0.05 are thought-about statistically vital. Bar graphs depict group imply with error bars representing commonplace error of the imply. Supply information are offered as a Supply Knowledge File.
AAV6-CXCL9 enhances lymphocyte chemotaxis
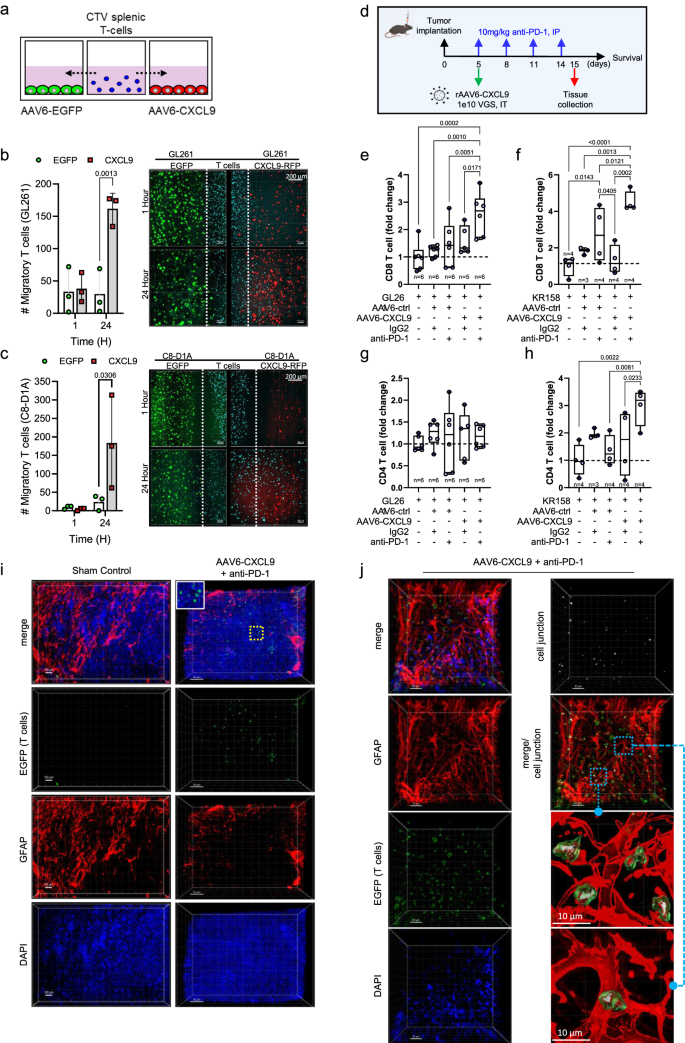
To judge the biologic exercise of AAV6-CXCL9 on lymphocyte recruitment, we carried out aggressive in vitro chemotaxis assays. Briefly, CTV-labeled splenic-derived T lymphocytes had been flanked by goal cells transduced with AAV6 encoding both EGFP or CXCL9, and migration was monitored through fluorescence microscopy at 1- and 24 h following co-culture as described within the strategies (Fig. 3a). Utilizing GL261 tumor cells because the goal inhabitants for AAV6 transduction, considerably extra T lymphocytes co-localized within the CXCL9 transduced tumor area as in comparison with EGFP at 24 h (Fig. 3b). On condition that astrocytes are the principal goal of AAV6 transduction in vivo, chemotaxis was reassessed through aggressive co-culture utilizing astrocytes (C8-D1A) in lieu of GL261 glioma cells. Astrocytes transduced with AAV6-CXCL9 equally confirmed enhanced recruitment of T lymphocytes (Fig. 3c), confirming that transgene encoding CXCL9 produces a biologically useful chemokine. We additionally in contrast lymphocyte chemotaxis within the context of AAV6-CXCL9 transduced tumor cells versus AAV6-CXCL9 transduced astrocytes, and located that lymphocytes had been evenly distributed in each the astrocyte area and tumor area (Supplementary Fig. S5a), although elevated ranges of CXCL9 secretion had been detected within the tumor area (Supplementary Fig. S5b). This we attribute partly in direction of the upper proliferative capability of tumor cells as in comparison with C8-D1A astrocytes (Supplementary Fig. S5c). Altogether, these information point out that CXCL9 capabilities in an autonomous method, successfully selling lymphocyte chemotaxis unbiased of the mobile supply. It additionally suggests that there’s a minimal organic threshold of CXCL9 sign wanted for lymphocyte chemotaxis, past which rising ranges of this chemokine could not impart a aggressive benefit.
a Diagrammatic overview of in vitro aggressive T lymphocyte chemotaxis assay. b Lymphocyte (CTV + , blue) chemotaxis in AAV6-EGFP (management, inexperienced) transduced or AAV6-CXCL9 (RFP + , pink) transduced GL261 area at 1- and 24 h following co-culture. Statistical analyses carried out by two-way ANOVA with Sidak’s a number of comparisons take a look at, n = 3 per time level, per group. Consultant photos proven. Dashed white line represents the lymphocyte-tumor border at assay begin. c Aggressive chemotaxis measured as described in (b) in C8-D1A astrocytes area at 1- and 24 h following co-culture. d Schematic outlining mixture AAV6 and PD-1 ICB therapy and tissue assortment and survival evaluation. Circulate cytometric detection of tumor-infiltrating CD8+ lymphocytes in management, single or mixture AAV6-CXCL9 plus anti-PD-1 ICB therapy in (e) GL261 and (f) KR158 fashions. Fold-change normalization based mostly on sham values (dashed black line). Statistical evaluation carried out utilizing extraordinary one-way ANOVA with Fisher’s least vital distinction (LSD) take a look at for a number of comparisons, n = 3–6 per therapy group, particular person values proven. Circulate cytometric detection of tumor-infiltrating CD4+ lymphocytes in management, single or mixture AAV6-CXCL9 plus anti-PD-1 ICB in (g) GL261 and (h) KR158 fashions. Fold-change normalization based mostly on sham values (dashed black line). Statistical evaluation carried out utilizing extraordinary one-way ANOVA with Fisher’s LSD take a look at for a number of comparisons, n = 3–6 per therapy group, particular person values proven. i 3D IHC of lymphocytes (EGFP + , inexperienced) in sham tumor management (scale bar 100 µm) and mixture AAV6-CXCL9 plus anti-PD-1 ICB handled GL261 tumors (scale bar 70 µm) remoted day 15. Tissues counterstained with DAPI (blue) and GFAP (astrocytes, pink). Photos consultant of n = 3 per therapy group. j Consultant 3D IHC of a area of lymphocyte infiltration (EGFP + , inexperienced) together handled GL261 tumors, n = 3, scale bar 30 µm. Tissues had been counterstained with DAPI nuclear dye (blue) and GFAP (astrocytes, pink). Voxel-based co-localization between EGFP and GFAP point out areas of convergence, ‘cell junctions’, between astrocytes and T cells, proven as a separate channel (white). 3D floor renderings of astrocytes and lymphocytes to visualise cell junctions in better element. P-values ≤ 0.05 are thought-about statistically vital. Bar graphs depict group imply with error bars representing commonplace deviation. Field-whisker plots show the field starting from the primary to the third quartile, the middle median worth, and the whiskers lengthen from every quartile to the minimal and most values. Supply information are offered as a Supply Knowledge File.
To find out the impact of AAV6-CXCL9 on T lymphocyte recruitment in vivo, multiparametric circulate cytometry was carried out to quantitate the variety of T cells current in dissociated tumors following intratumor supply. These research had been completed together with anti-PD-1 ICB, the place tissue was collected 1 day following the ultimate dose of ICB to seize occasions throughout the therapeutic response window as outlined in Fig. 3d. In each GL261 and KR158 tumor fashions AAV6-CXCL9 alone had minimal impression on enhancing T cell recruitment to the tumor, nevertheless vital will increase in T lymphocyte infiltration had been noticed within the context of mixture therapy. AAV6-CXCL9 plus ICB elevated CD8 T lymphocytes > 2.5-fold within the GL261 mannequin and >4.5-fold within the KR158 mannequin (Fig. 3e, f and Supplementary Fig. S4b). Whereas no vital adjustments in CD4 T lymphocyte recruitment in response to therapy was noticed within the GL261 mannequin (Fig. 3g), a >3-fold improve was detected within the KR158 mannequin (Fig. 3h). Anti-PD-1 ICB therapy together with management AAV6 (EGFP) modestly elevated CD8 T lymphocyte recruitment in GL261 by 1.4-fold and in KR158 by 2.7-fold, indicating that CXCL9 markedly improves tumor infiltration by these cells. These information spotlight a possible function for anti-PD-1 ICB in mobilizing T lymphocytes systemically, the place sequestration of T lymphocytes was not too long ago proposed as a mechanism of immune suppression in mind tumors7. To judge the geospatial distribution of T lymphocyte infiltration into handled tumors, we resected tumor tissue on day 15 following therapy with both sham management or mixture AAV6-CXCL9 with anti-PD-1 ICB, the place lymphocytes are identifiable by way of endogenous EGFP expression. Few, if any, lymphocytes could possibly be visualized in management handled tumors (Fig. 3i). A heterogenous distribution of lymphocytes could possibly be readily detected in each peritumor and intratumor areas together handled tissue (Fig. 3i), confirming that therapy will increase each lymphocyte tumor recruitment and penetration. Together handled tumors, T lymphocytes had been additionally noticed in areas of astrocytosis, the place they fashioned direct cell-cell contact factors, or ‘cell junctions (white),’ as detected by voxel co-localization between GFAP (astrocytes, pink) and EGFP (lymphocytes, inexperienced) (Fig. 3j). Whereas mobile communication between these two cell populations may be inferred given the bodily proximity and direct contact, the precise nature or respective affect imparted by these interactions stay an space of ongoing analysis.
AAV6-CXCL9 sensitizes preclinical GBM to anti-PD-1 ICB
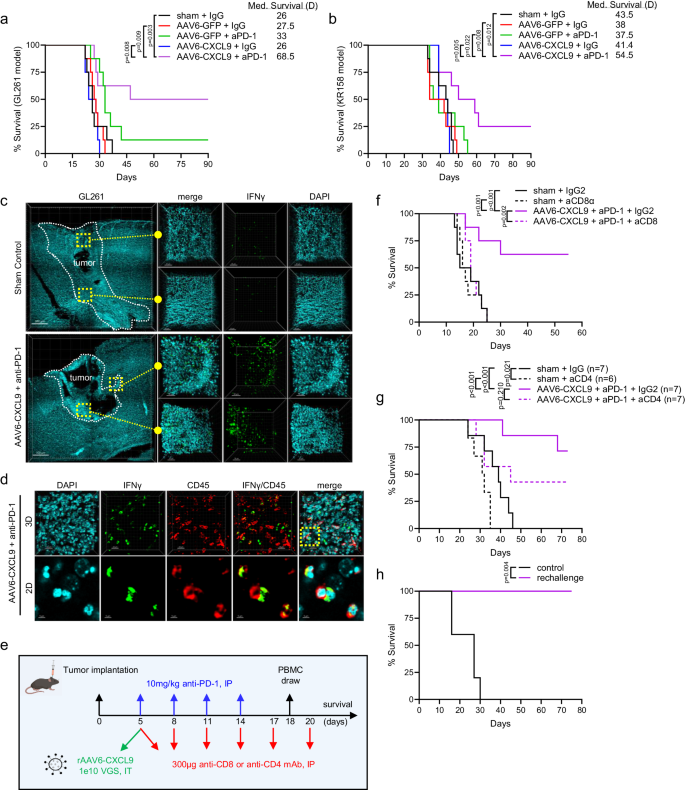
To evaluate if enhanced lymphocyte recruitment and immunological reprogramming by way of mixture therapy may produce anti-tumor responses in opposition to GBM, we carried out survival analyses in each the GL261 and KR158 syngeneic mannequin methods. 5 days following tumor implantation, AAV6 encoding CXCL9 or EGFP management transgene was injected intratumorally, with anti-PD-1 ICB (10 mg/kg) administered intraperitoneally for a complete of 4 doses given each 72 h (Fig. 3d). Within the GL261 mannequin we discovered that anti-PD-1 ICB produced a small, however non-significant improve in general survival as in comparison with sham handled management animals (p = 0.060), the place AAV6-CXCL9 therapy yielded no survival profit as a monotherapy (Fig. 4a). Mixture therapy considerably improved general survival, with 50% of animals exhibiting sturdy outcomes (Fig. 4a). We noticed comparable ends in the KR158 mannequin, which carries a low mutational burden and is recalcitrant to immunotherapy, with mixture therapy considerably bettering median survival, and long-term survival noticed in 25% of this cohort (Fig. 4b). As an extra metric to validate the flexibility of mixture AAV6-CXCL9 plus anti-PD-1 ICB to immunologically remodel GBM tumors, GL261 tumors had been implanted in GREAT transgenic mice to judge tumor-wide IFNγ expression following therapy as described in Fig. 3d. IFNγ was readily detected together handled tumors as in comparison with sham management handled tumors, evidenced by EYFP sign detection through 3D IHC (Fig. 4c). Immunolabeling of tissues for CD45 confirms that EYFP (IFNγ)+ cells are immune cells (Fig. 4d), indicative of pro-inflammatory immune activation.
Survival evaluation in (a) GL261 and (b) KR158 tumor-bearing mice handled with management, AAV6-CXCL9, and anti-PD-1 ICB alone and together. Median survival for every therapy group proven, n = 8 per group. Statistical evaluation was carried out utilizing Log-rank (Mantel-Cox) take a look at evaluating particular person therapy teams. c Tile-stitch 10x 3D IHC imaging of GL261 tumors resected from sham management and mixture AAV6-CXCL9 plus anti-PD-1 ICB handled GREAT mice, n = 3 per group, scale bar 300 µm. DAPI nuclear dye (blue) used to determine tumor space outlined by the dashed white line. Digital magnification of areas outlined within the far-left panel to point out larger picture decision for every therapy, scale bar 50 µm. EYFP (inexperienced) correlates with IFNγ expression. d 3D IF of tissue from GL261 tumor tissue as proven in (c) immunolabeled for CD45 expression (pink), scale bar 20 µm. Digital zoom of area outlined within the far-right panel reveals co-localization between CD45 and IFNγ, indicating these are immune cells, n = 3, scale bar 5 µm. e Diagrammatic abstract of mixture therapy technique with concomitant CD4 or CD8α antibody depletion. f Survival evaluation in GL261 tumor-bearing mice handled with mixture AAV6-CXCL9 plus anti-PD-1 monoclonal antibody, with or with out anti-CD8α depletion. Statistical evaluation carried out utilizing Log-rank (Mantel-Cox) take a look at evaluating particular person therapy teams, n = 8 per group. g Survival evaluation in GL261 tumor-bearing mice handled with mixture AAV6-CXCL9 plus anti-PD-1 monoclonal antibody, with or with out anti-CD4 depletion. Statistical evaluation carried out utilizing Log-rank (Mantel-Cox) take a look at evaluating particular person therapy teams, n = 7–8 per group. h Survival evaluation in long-term survivors from mixture handled animals re-challenged with tumor at day 55 of examine (n = 5). Age-matched naïve management mice had been orthotopically implanted with GL261 as survival management arm. Statistical evaluation was carried out utilizing Log-rank (Mantel-Cox) take a look at evaluating particular person therapy teams. P-values ≤ 0.05 are thought-about statistically vital. Supply information are offered as a Supply Knowledge File.
To find out if CD8 lymphocytes contribute to the therapeutic survival impact, we repeated combinatorial therapy with concomitant CD8 depletion (Fig. 4e) within the GL261 mannequin. We discovered that on examine day 18 all animals handled with CD8 depleting antibodies had no detectable ranges of circulating CD8 T lymphocytes, and no adjustments within the amount of circulating CD4 T lymphocytes (Fig. 4f, Supplementary Fig. S5d). CD8 depletion reversed the survival profit noticed with mixture AAV6-CXCL9 plus anti-PD-1 ICB, and this cohort progressed as rapidly as management handled topics (Fig. 4f). We equally evaluated the impression of CD4 lymphocyte depletion (Fig. 4e, Supplementary Fig. S5e), and located that whereas there was a pattern for lowered survival in animals handled with mixture remedy plus concomitant CD4 depletion, these outcomes weren’t thought-about to be statistically vital, and lots of animals within the cohort demonstrated prolonged survival as in comparison with management (Fig. 4g). Apparently, sham management animals with concomitant CD4 depletion demonstrated extra speedy illness development as in comparison with non-depleted controls, indicating an essential function for these lymphocytes in host tumor response (Fig. 4g). To find out if mixture therapy may confer long-term immune reminiscence formation, we carried out a GL261 tumor rechallenge in long-term survivors (>55 days) that had acquired AAV6-CXCL9 plus anti-PD-1 ICB. No observable residual tumor was current from the preliminary tumor implantation throughout the second implantation. A second cohort of age-matched naïve animals was intracranially injected with GL261 as a management. Management animals all succumbed to tumor burden inside 30 days of tumor implantation, whereas 100% of rechallenge animals remained illness free (Fig. 4h). These information verify that therapeutic response to mixture remedy relies on tumor infiltration by CD8 T lymphocytes as a part of the adaptive immune cascade, and mixture remedy can convey long-term immune reminiscence safety in opposition to recurrence.
To evaluate if intratumor supply of AAV6-CXCL9 is significant for therapeutic efficacy we in contrast general survival in GL261 tumor bearing mice the place AAV6-CXCL9 (1e10 VGS) was intratumorally (IT) injected, or injected into the contralateral (CL) hemisphere at coordinates symmetrical to intratumor supply. We discovered that intratumor supply of AAV6-CXCL9 produced a extra strong survival response as in comparison with contralateral supply (Supplementary Fig. S5f), though a subset of those animals did display prolonged survival as in comparison with management. We additionally examined therapeutic efficacy in additional established tumors, evaluating AAV6-CXCL9 intratumor supply plus mixture anti-PD-1 ICB given on day 5 or day 12 (delayed therapy, DT). Whereas delayed therapy yielded some survival profit as in comparison with management handled animals, is was considerably lowered as in comparison with animals receiving mixture therapy starting on day 5 (Supplementary Fig. S5g). Entire mind imaging of GL261 tumors excised 1 week following intratumor supply at day 12 with AAV6 encoding an mCherry fluorescent reporter transgene revealed intensive native and distant illness development, together with ventricular dissemination (Supplementary Fig. S5h). AAV6 transduction in these bigger tumors was confined largely to the positioning of intratumor injection (Supplementary Fig. S5h), indicating that in bigger tumors a multifocal supply methodology could also be required for optimum anti-tumor efficacy.
scRNAseq identifies treatment-related immune response to AAV6-CXCL9 and anti-PD-1 ICB
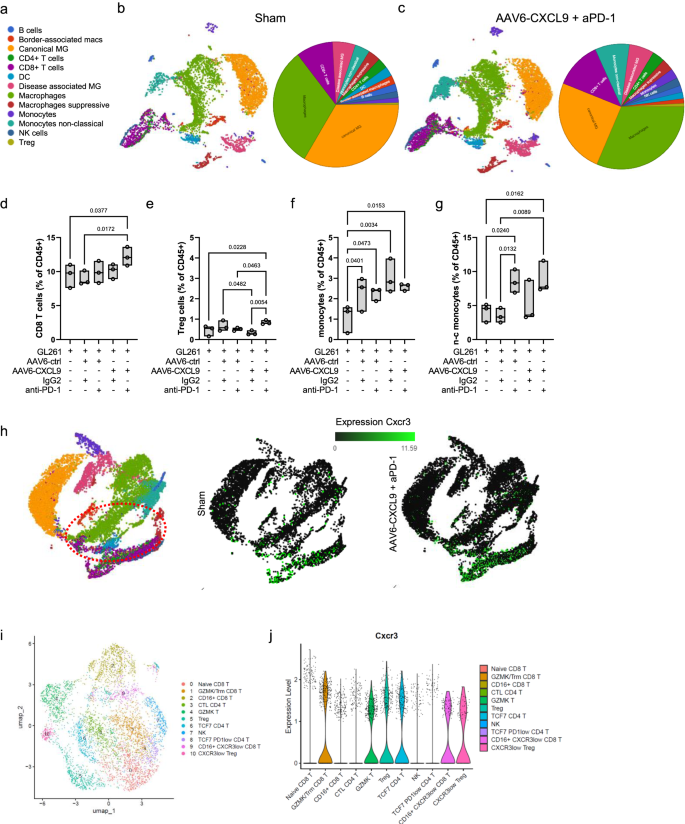
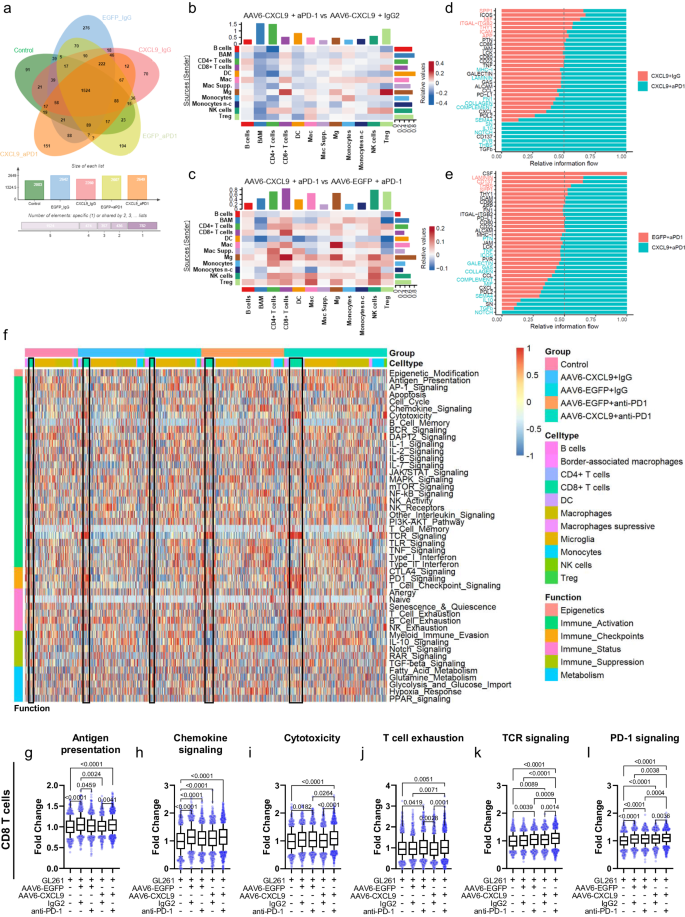
In an effort to outline the immunological panorama of AAV6-CXCL9 handled tumors with or with out concurrent anti-PD-1 ICB, we carried out single cell RNA sequencing (scRNAseq) on CD45-positive cells remoted from GL261 tumors collected on day 15 of therapy as outlined in Fig. 3d. High differentially expressed genes from every pooled inhabitants had been recognized, and cluster cell sorts had been outlined utilizing the expression of identified marker genes ensuing within the identification of 13 distinctive cell clusters34,35 (Fig. 5a, Supplementary Desk 1). Dimensionality discount utilizing uniform manifold approximation and projection (UMAP) was carried out on 52,344 cells collected throughout 5 therapy teams: sham (saline), AAV6-ctrl + IgG, AAV6-ctrl + aPD-1, AAV6-CXCL9 + IgG, and mixture AAV6-CXCL9 + aPD-1, 3 mice per group (Fig. 5b, c, Supplementary Fig. S7a–d). Analyses of lymphocyte tumor recruitment throughout therapy teams recapitulate our earlier statement, with mixture remedy yielding a big improve in whole infiltrating CD8 T lymphocytes (Fig. 5d), recognized utilizing the gene expression markers Cd3d, Cd8a, Cd8b1 as beforehand described36. T regulatory lymphocytes (Treg), outlined by Cd4, Foxp3, and Il2ra gene expression36, had been additionally elevated in response to mixture remedy, though collectively these signify <1% of the whole tumor-associated immune inhabitants (Fig. 5e). Elevated tumor infiltration by monocytes, labeled by excessive Ly6c1 expression, was noticed throughout all therapy teams as in comparison with sham management mice (Fig. 5f), with an enrichment of non-classical monocytes characterised by Spn, Cx3cr1, and Tnfrsf1b expression37 in teams receiving anti-PD-1 therapy (Fig. 5g). Graphical summaries for all remaining cells clusters in response to every therapy are proven in Supplementary Fig. S6a–h. On condition that CXCR3 is the concomitant receptor for CXCL9, we leveraged our scRNAseq information to evaluate which tumor-associated immune cells may work together with our AAV encoded transgene. We discovered that Cxcr3 was principally expressed by lymphocytes, as proven on the UMAP in Fig. 5h. Additional classification of lymphocyte subsets was completed to find out if particular populations can be roughly attentive to AAV-CXCL9 remedy by way of differential CXCR3 expression. We recognized 11 distinctive lymphocyte clusters (Fig. 5i, Supplementary Fig. S7e), with equal distribution of Cxcr3 transcript expression detected throughout all clusters, with cluster 9 and 10 exhibiting barely lowered general Cxcr3 transcript expression (Fig. 5i, Supplementary Fig. S7e). These findings counsel that CXCL9 broadly interacts with all lymphocyte subsets by way of CXCR3 expression.
a Abstract of UMAP cell clusters. UMAP of cell sorts clustered by scRNA transcriptional evaluation of 52,344 CD45+ cells remoted from GL261 tumor bearing mice handled with (b) sham (saline) or (c) mixture AAV6-CXCL9 + aPD-1 handled GL261 tumors, n = 3 mice per group. Abstract circle chart depicting cell cluster inhabitants frequency detected for every therapy included alongside every UMAP. Quantitative change in inhabitants frequency of (d) CD8 + T cells, (e) Treg cells, (f) monocytes, and (g) non-classical (n–c) monocytes throughout therapy teams. Statistical analyses carried out utilizing extraordinary one-way ANOVA with Fisher’s LSD take a look at for a number of comparisons, n = 3 per group, particular person values proven. h UMAP projection of Cxcr3 transcript expression (TPM) detected throughout all cell clusters. i UMAP of T cell clusters. j Abstract of Cxcr3 transcript expression throughout outlined T cell clusters. Field-whisker plots show the field starting from the primary to the third quartile, the middle median worth, and the whiskers lengthen from every quartile to the minimal and most values. Supply information are offered as a Supply Knowledge File.
Mixture AAV6-CXCL9 and anti-PD-1 ICB therapy will increase mobile crosstalk in lymphocytes
As proven in Fig. 6a, we recognized 2,260 differentially expressed genes (DEGs) related to AAV6-CXCL9 therapy, 2,607 DEGs related to anti-PD-1 therapy, and a pair of,649 DEGs related to these remedies mixed. Of those, 70, 194, and 151 DEGs look like distinctive to every given therapy technique, respectively, and should present distinctive perception towards therapy impression on immune cell useful states. By means of transcriptional expression of distinct ligands and receptors, cell-type-specific interactions had been inferred, offering extra perception in direction of the inflammatory profile of tumors and the way they modify in response to therapy38. Utilizing our predefined cell clusters, a simplified DEG set was established for every. DEGs had been then queried in opposition to public ligand-receptor databases (see Strategies). Abstract outcomes are proven in Chord Plots, the place line thickness represents the variety of predicted interactions between two outlined cell clusters (Supplementary Fig. S8a–e). Subsequent, we carried out direct comparisons of interactome exercise between therapy teams to elucidate heightened or decreased connectivity related to AAV6-CXCL9 and anti-PD-1 ICB, the place heatmap relative values in pink point out will increase and blue decreases in potential ligand-receptor interactions. In evaluating AAV6-CXCL9 together with both anti-PD-1 ICB or IgG2 management to resolve the contributions of ICB, notable will increase in alerts emanating from every macrophages (Mac), border-associated macrophages (BAM), microglia (Mg), and NK cells signaling to CD8+ and regulatory T cells had been noticed (Fig. 6b). Decreased incoming alerts had been famous in BAMs, CD4 + T cells, and dendritic cells (DCs) stemming from almost all cell clusters (Fig. 6b). Cell-cell interactions related to AAV6-CXCL9 proven in Fig. 6c reveal heightened communication directed towards each CD4+ and CD8 + T cell subsets, and NK cells prompted by all clusters excluding B cells and DCs. Signaling originating from all lymphocyte populations, and most innate immune cells together with Macs, Mgs, Monocytes, and NK cells was elevated, suggesting that AAV6-CXCL9 therapy broadly stimulates immune exercise.
a Venn Diagram representing differentially expressed genes affiliated with every therapy. b Heatmap depicting scRNA-seq-derived cell-cell communication networks enriched or decreased in response to mixture AAV6-CXCL9 + aPD-1 as in comparison with AAV6-CXCL9 + IgG2 therapy throughout recognized cell clusters. c Heatmap depicting scRNA-seq-derived cell-cell communication networks enriched or decreased in response to mixture AAV6-CXCL9 + aPD-1 as in comparison with AAV6-EGFP + aPD-1 therapy throughout recognized cell clusters. d Waterfall abstract plot of scRNA-seq-derived signaling pathways enriched in CD8 + T cells following mixture AAV6-CXCL9 + aPD-1 as in comparison with AAV6-CXCL9 + IgG2 therapy. e Waterfall abstract plot of scRNA-seq-derived signaling pathways enriched in CD8 + T cells following mixture AAV6-CXCL9 + aPD-1 as in comparison with AAV6-EGFP + aPD-1 therapy. f Heatmap illustration of gene expression evaluation derived from all cell clusters utilizing the nCounter® Immune Exhaustion Panel (nanoString) following AAV6-CXCL9 gene remedy with or with out PD-1 ICB. CD8 + T cell populations outlined in black for every therapy group. g–l Quantification of frequent pathways discovered to be differentially regulated in CD8 + T cells in response to therapy, derived from n = 669, 674, 1099, 912, and 1656 single cells pooled from n = 3 particular person samples per therapy group, graphically introduced from left to proper. Statistical analyses carried out utilizing Kruskal–Wallis take a look at adopted by Dunn’s a number of comparisons, with particular person values proven. P-values ≤ 0.05 are thought-about statistically vital. Field-whisker plots show the field starting from the primary to the third quartile, the middle median worth, and the whiskers lengthen from every quartile to the minimal and most values. Supply information are offered as a Supply Knowledge File.
On condition that mixture therapy promotes CD8 T cell tumor infiltration, which is required for anti-tumor efficacy, we sought to resolve how therapy may impression CD8 T cell effector perform through pathway evaluation of DEGs particularly inside these cells. Comparative pathway evaluation between CD8 T cell DEGs reveals selective enrichment of thrombospondin (Thbs1), poliovirus receptor (Pvr, Cd155), Cd137 (Tnfrsf9, 4-1BB), fibronectin-1 (Fn1), laminin (Lamc1), and several other genes related to main histocompatibility complicated class I signaling, amongst others, as uniquely affiliated with mixture remedy when in comparison with AAV6-CXCL9 plus IgG2 management (Fig. 6d). These information counsel that anti-PD-1 therapy prompts T cell activation through CD13739, but in addition reciprocal immune suppression through CD155 given its inhibitory perform as a ligand for T cell immunoreceptor with immunoglobulin and ITIM area (TIGIT)40. FN1, laminin, and THBS are main constituents of the extracellular matrix, and when produced by lymphocytes have been described to help cell-cell engagement, transendothelial migration, and lymphoproliferation41,42,43. DEG comparisons between mixture remedy and AAV6-EGFP management plus anti-PD-1 ICB therapy reveals selective pathway enrichment of macrophage migration inhibitory issue (Mif), progress arrest particular (Gas6), galectin-9 (Lgals9), inducible T cell co-stimulator (Icos1), tumor necrosis issue (Tnf), and pleiotrophin (Ptn) because of AAV6-CXCL9 therapy (Fig. 6e). These information infer that AAV6-CXCL9 straight promotes CD8 T cell activation by way of elevated ICOS and TNF expression44,45, T cell migration through PTN46, and reciprocally enhances innate immune stimulation of NK cells and myeloid cells through GAS6 and MIF secretion, respectively47,48. It additionally reveals galectin-9 as a potential mechanism for CD8 T cell acquired exhaustion49,50. Each anti-PD-1 ICB and AAV6-CXCL9 remedies stimulate NOTCH, TGFβ, IL-10, SEMA4, CXCL, Complement, and CCL pathway activation (Fig. 6d, e), every with various impression on T cell maturation, effector perform, homeostasis, survival, and migration51,52,53,54,55,56,57. Comparative pathway evaluation was additionally carried out for CD4 T cells throughout therapy teams (Supplementary Fig. S8f, g).
We subsequent leveraged the NanoString nCounter® Immune Exhaustion Panel to additional characterize immune standing and inflammatory signatures related to every respective therapy. A abstract of pathway activation throughout all cell subsets in response to particular person remedies is proven in Fig. 6f and Supplementary Fig. S8h, with CD8 T cell clusters outlined in black for every therapy group. From the NanoString evaluation, CD8 T cell particular differential pathway activation is graphically introduced in Fig. 6g–l, the place we determine treatment-associated adjustments in every antigen presentation (Fig. 6g), chemokine signaling (Fig. 6h), cytotoxicity (Fig. 6i), T cell exhaustion (Fig. 6j), TCR signaling (Fig. 6k), and PD-1 signaling (Fig. 6l). Particularly, mixture AAV6-CXCL9 plus anti-PD-1 ICB was related to the very best improve in cytotoxicity, TCR signaling, and PD-1 signaling. Elevated T cell exhaustion seems to be related to AAV6-CXCL9 therapy. On condition that monocyte tumor infiltration was moreover elevated in response to therapy (Fig. 5f, g), we evaluated pathway activation in these cells to raised perceive their useful standing. We discovered enhanced activation throughout 12 pathways, together with antigen presentation, chemokine signaling, cytotoxicity, IL-10 signaling, JAK/STAT signaling, different interleukin signaling, T cell checkpoint, TGFβ signaling, TNF signaling, Sort I interferon signaling, and Sort II interferon signaling (Supplementary Fig. S9). Of those, AAV6-CXCL9 therapy seems to be related to elevated antigen presentation, cytotoxicity, JAK/STAT signaling, and Sort I interferon signaling, the place anti-PD-1 ICB induces IL-10 signaling, TLR signaling, and TNF signaling. Collectively these information are suggestive that therapy could increase the pro-inflammatory perform of those cells.
Cytokine profiling of mixture AAV6-CXCL9 plus anti-PD-1 ICB
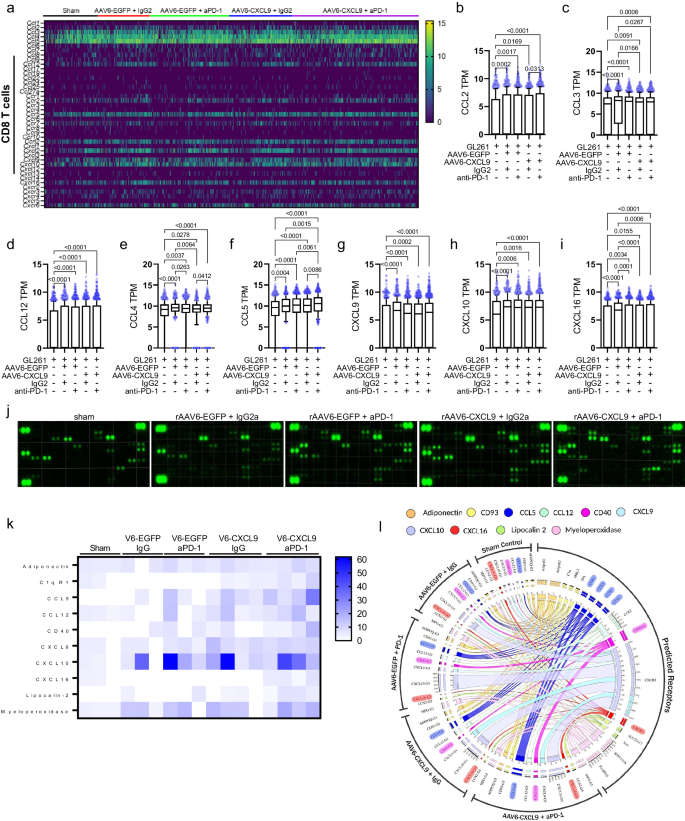
As mixture remedy will increase DEGs of each the CCL and CXC superfamily of secreted chemokines and cytokines, we sought to parse out transcriptional adjustments inside CD8 T cells as an extra means to judge the activation state of those cells given the central function of those ligands in directing migration and activation of immune cells throughout irritation58. A abstract of all CCL and CXC household ligand and receptor transcripts expressed by CD8 T cells is introduced within the heatmap in Fig. 7a. CD8 T cell mediated stimulation of monocytes/macrophages is evidenced by elevated transcription of Ccl2, Ccl3, and Ccl12 throughout all therapy teams as in comparison with sham management (Fig. 7b–d). Ccl4 transcription was additionally elevated (Fig. 7e), indicative of NK stimulation by CD8 T cells. Ccl5 was discovered to be probably the most differentially upregulated soluble ligand together handled CD8 T cells as in comparison with all different therapy teams (Fig. 7f), and is indicative of CD8 T cell effector perform59,60. Whereas we present that AAV6 delivered CXCL9 transgene expression predominantly emanates from tumor-reactive astrocytes, our scRNAseq information reveals that every anti-PD-1 and AAV gene remedy induces CXCL9 transcription inside CD8 T cells (Fig. 7g), moreover demonstrating immune activation because of therapy22,23,61. Cxcl10 was additionally discovered to be transcriptionally upregulated in response to anti-PD-1 and AAV gene remedy (Fig. 7h), which prompts additional CD4, CD8, and NKT lymphocyte recruitment61. Altogether, these information help that mixture AAV6-CXCL9 and anti-PD-1 ICB each will increase lymphocyte trafficking to intracranial GBM tumors and potently stimulates effector lymphocyte mobile communication and activation.
a Heatmap abstract of scRNA-seq-derived CCL-CXC expression in CD8 + T cells remoted from GL261 tumors in response to AAV6-CXCL9 and anti-PD-1 ICB therapy created utilizing GraphPad Prism. b–i Quantification of CCL-CXC genes discovered to be differentially expressed in CD8 + T cells in response to therapy derived from n = 669, 674, 1099, 912, and 1656 single cells pooled from n = 3 particular person samples per therapy group, graphically introduced from left to proper. Statistical analyses carried out utilizing Kruskal–Wallis take a look at adopted by Dunn’s a number of comparisons, with particular person values proven. j Consultant immunoblots depicting chemokine and cytokine protein expression detected in GL261 tumors resected following therapy with AAV6-CXCL9 with and with out PD-1 ICB (n = 3 for sham, rAAV6-EGFP + IgG2a, and rAAV6-EGFP + aPD-1; n = 4 for rAAV6-CXCL9 + IgG2a and rAAV6-CXCL9 + aPD-1). -4 per group. ok Heatmap abstract of CCL-CXC relative protein expression discovered to be differentially expressed in response to AAV6-CXCL9 with and with out PD-1 ICB, created utilizing GraphPad Prism. l Circos interactome evaluation of detected differentially expressed proteins and predicted receptors. P-values ≤ 0.05 are thought-about statistically vital. Field-whisker plots show the field starting from the primary to the third quartile, the middle median worth, and the whiskers lengthen from every quartile to the minimal and most values. Supply information are offered as a Supply Knowledge File.
As described above, secreted cytokines can affect the trajectory of tumors in a mess of ways- reprogramming tumor-associated cells and suppressing infiltrating inflammatory subsets which permits for tumor tolerance, development, metastasis, and even therapeutic resistance or, alternatively, creating an setting favorable for innate and adaptive immune activation to facilitate tumor rejection62. Furthermore, the cytokine profile of a tumor could function predictive and/or therapeutic biomarkers permitting for the detection of tumor presence, forecasting therapeutic response, and can be used to information therapeutic selections62. We carried out a large-scale cytokine proteomic evaluation of single agent and mixture handled tumors to determine candidate biomarkers of response to remedy. Tumors had been resected 10 days after the onset of therapy as proven in Fig. 3d. Of the 111 soluble murine proteins on the array, relative expression of 65 was detected in handled and/or management GL261 tumor samples as summarized Supplementary Fig. S10a, with consultant cytokine immunoblots proven in Fig. 7j. 10 secreted elements had been recognized as differentially expressed as in comparison with sham management tumors following both single or mixture therapy with AAV6-CXCL9 and anti-PD-1 ICB: ADIPOQ, C1QR1 (CD93), CCL5, CCL12, CD40, CXCL9, CXCL10, CXCL16, LCN2, and MPO (Fig. 7k, Supplementary Fig. S10b–ok). Of those, CCL5, CD40, and CXCL16 had been most potently induced by mixture therapy. These markers are indicative of lymphocyte presence and activation, the place CCL5 is a potent pro-inflammatory ligand manufactured principally by CD8 T lymphocytes, and CD40 is a co-stimulatory ligand that triggers lymphocyte proliferation and cytokine manufacturing59,60. Of be aware, elevated CCL5 ligand expression demonstrates concordance with scRNAseq information (Fig. 7f). As well as, CXCL9, CXCL10 and CXCL16 are robust chemotactic alerts for lymphocyte recruitment. Each CXCL10 and CXCL16 are induced by interferon gamma (IFNγ) and tumor necrosis alpha (TNFα), highly effective catalysts of innate and adaptive irritation63,64. A abstract of treatment-induced secreted ligands and identified receptor interactions are depicted through round interactome evaluation carried out utilizing Circos® visualization software program65, revealing perception in direction of immune reprogramming that happens in response to every respective therapy (Fig. 7l). These information mixed validate that AAV6 supply of CXCL9 to the tumor microenvironment in tandem with anti-PD-1 ICB not solely facilitate lymphocyte recruitment to GBM tumors, but in addition reprograms the immunological panorama in direction of a pro-inflammatory phenotype.
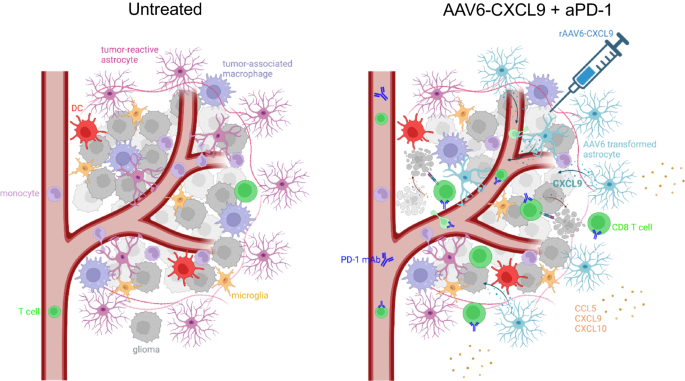
In summation (Fig. 8), intra-tumor supply of AAV6 encoded CXCL9 ends in the manufacturing of a pro-lymphocyte chemotactic gradient by transduced tumor-reactive astrocytes. This, in live performance with anti-PD-1 ICB, considerably will increase tumor infiltration by lymphocytes doubtless by way of CXCL9 engagement with its cognate receptor expressed by these cells- CXC motif chemokine receptor 3 (CXCR3). Particularly, CD8 T lymphocytes are the premier arbiters of anti-tumor response, the place depletion of this lymphocyte subset negates therapeutic efficacy. Furthermore, CD8 T cell effector activation and performance is evidenced by heightened expression of co-stimulatory molecules, resembling 4-1BB and ICOS, and manufacturing of pro-inflammatory chemokines and cytokines. Past CD8 T lymphocyte activation, AAV6-CXCL9 and anti-PD-1 ICB seem to contribute widespread immunological activation, demonstrated by heightened mobile cross-talk throughout quite a few immune clusters, and protein detection of pro-inflammatory molecules. Notably, CCL5, CXCL9, CXCL10, and CD40 are detected in response to mixture remedy inside resected tumors, and should function biomarkers of therapeutic response.
Intra-tumor supply of AAV6 encoding CXCL9 ends in strong transduction of tumor-reactive astrocytes, making a chemotactic gradient of secreted CXCL9. This improves lymphocyte trafficking together with anti-PD-1 ICB by way of chemokine-receptor engagement between CXCL9 within the TME and CXCR3 expression on lymphocytes. CD8 + T cells are required for sturdy survival response to therapy, indicating that tumor cell killing is mediated by the adaptive arm of immunity. Mixture therapy additionally transforms the inflammatory milieu of tumors, making a pro-inflammatory setting evidenced by the presence of cytokines and chemokines that additional promote innate and adaptive immune activation.