LIF performs a key position in cachexia improvement in mice
Earlier research have proven that mice bearing C26 tumors develop cachexia12,13, and it has been instructed that the elevated LIF ranges produced by C26 tumors contribute to cachexia within the tumor-bearing mice11. To verify that LIF contributes to cachexia on this mannequin, C26 cells with LIF knockout (KO) by the CRISPR/Cas9 system (C26-LIF KO) have been used to ascertain subcutaneous (s.c.) syngeneic xenograft tumors in Balb/c mice. LIF was extremely expressed in tumors fashioned by C26 cells, however undetectable in tumors fashioned by C26-LIF KO cells (Fig. S1A). Per earlier experiences11,12,13, Balb/c mice bearing C26 cell-formed tumors exhibited signs of cachexia shortly after tumor formation. Mice bearing tumors on the dimension of ~400 mm3 confirmed a major lower in physique weight (Fig. S1B), muscle losing, and white adipose tissue (WAT) loss as examined by H&E staining (Fig. S1C), that are foremost traits of cachexia, and these mice had a median survival of 15 days (Fig. S1D). Notably, LIF knockout in C26 cells drastically mitigated cachexia in tumor-bearing mice. In contrast with C26 tumors, the expansion of C26-LIF KO tumors was a lot slower (Fig. S1E). When tumors reached a comparable dimension, mice bearing C26-LIF KO tumors confirmed much less physique weight reduction, and decreased muscle and WAT losing in contrast with mice bearing C26 tumors (Fig. S1B, C). Consequently, mice bearing C26-LIF KO tumors had a for much longer survival than C26 tumor-bearing mice; the vast majority of mice bearing C26-LIF KO tumors reached the humane endpoint as a consequence of tumor dimension, whereas solely a small proportion of those mice reached the humane endpoint as a consequence of cachexia (Fig. S1D). These outcomes exhibit that prime LIF ranges produced from C26 tumor cells play a vital position in cachexia, validating LIF as a vital tumor-produced cachexia issue.
LIF performs a profound position in selling the proliferation, progress, survival, and metabolic reprogramming in lots of stable tumor cells9,10,14. At the moment, it stays unclear how LIF promotes cachexia. The C26 tumor-induced cachexia mannequin can’t differentiate the contribution of the direct impact of LIF secreted from tumor cells on cachexia from the secondary results of LIF in selling the expansion, proliferation and dietary wants of tumor cells on cachexia. To research whether or not systemic elevation of LIF induces cachexia in mice, we generated a transgenic LIF knock-in mouse mannequin (LIF-tgflox/+; known as TgL hereafter) by knocking within the mouse LIF gene, preceded by the CAG promoter and a transcriptional STOP cassette, into the Rosa26 locus (CAG-STOP-LIF-eGFP-Rosa26TV) utilizing the CRISPR/Cas9 system, as described beforehand15,16 (Fig. 1A). TgL mice have been then crossed with R26-CreERT2 mice to generate TgL/R26-CreERT2 (known as TgLC hereafter) mice (Fig. 1B). Eight-week-old TgLC mice have been injected (intraperitoneal, i.p.) with tamoxifen (TAM) to induce LIF expression. Age- and gender-matched TgL mice injected with TAM served as controls. TAM injection considerably elevated LIF expression ranges in TgLC mice, with serum LIF ranges at 330 ± 87 pg/mL as decided by ELISA assays (Fig. 1C). These ranges are comparable with the serum LIF ranges in some most cancers sufferers, together with these with pancreatic ductal adenocarcinoma (PDAC) or oesophageal adenocarcinoma (OAC), as reported beforehand17,18,19. Systemic elevation of the LIF ranges rapidly induced cachexia in TgLC mice. Mice began to shed some pounds at ~3 days after TAM injection in TgLC mice (Fig. 1D). After TAM injection, TgLC mice rapidly misplaced fats mass, and subsequently began to lose lean mass at ~3 days after TAM injection, as measured by EchoMRI (Fig. 1E). At 10 days after TAM injection, the common lack of fats and lean mass was 1.63 ± 0.51 g and 5.58 ± 1.35 g, respectively (Fig. 1E). The losing of the muscle and WAT was validated by H&E assays (Fig. 1F). Cachexia is usually related to anorexia2,3. A pattern of a slight lower in physique weight and lean mass, and a major lower in fats mass have been noticed within the pair-fed TgL mice with TAM injection in contrast with the regular-fed TgL mice with TAM injection (Fig. S2A, B). Nonetheless, the lower in physique weight, lean mass, and fats mass within the pair-fed TgL mice with TAM injection was considerably lower than their lower in TgLC mice with TAM injection (examine Fig. S2A, B vs. Fig. 1E).
A The technique to generate LIF-tgflox/+ (TgL) mice. The mouse LIF gene, preceded by the CAG promoter and a transcriptional STOP cassette, was knocked into the Rosa26 locus (CAG-STOP-LIF-eGFP-Rosa26TV) utilizing the CRISPR/Cas9 system. B The technology of TgL/R26-CreERT2 (TgLC) mice. Proper panel: the genotyping evaluation of TgL and TgLC mice by PCR. All mice with the identical genotype have comparable outcomes. C Serum LIF ranges in TgL (n = 6) and TgLC mice (n = 10) with TAM injection measured by the ELISA assay. D Mouse physique weight put up TAM injection in TgL mice (n = 6) and TgLC mice (n = 14). E Fats and lean mass loss put up TAM injection in TgL (n = 10) and TgLC (n = 8) mice. Physique composition was measured by EchoMRI. F Consultant H&E photos of muscle and WAT tissues from TgL and TgLC mice with TAM injection. No less than three impartial organic replicates have been carried out. G–M Mice have been housed in Promethion metabolic cages. Mice have been injected with TAM on the first gentle cycle (n = 4–10/group). Shaded areas symbolize the darkish cycle from 19:00 pm to 7:00 am. Values are hourly means. Vitality steadiness (G), meals consumption (H), complete vitality expenditure (TEE) (I), oxygen consumption (VO2) (J), carbon dioxide manufacturing (VCO2) (Okay), Respiratory alternate ratio (RER) (L) and locomotor exercise (M) of TgL and TgLC mice put up TAM injection have been measured. N The serum ranges of albumin, blood urea nitrogen (BUN), alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT) reflecting kidney and liver features in TgL and TgLC mice at 3 days after TAM injection for albumin and 9 days after TAM injection for different parameters (n = 3/group). O Kaplan-Meier survival curves of mice. The day of TAM injection was denoted as D0. Information are offered as imply ± SEM for (D, E), and as imply ± SD for (C, G–N). N.D. non-detectable. Every dot represents a person mouse. Each feminine and male mice have been used. For G–N: Two-tailed Scholar’s t-test; for D, E two-way ANOVA adopted by Sidak’s a number of comparability take a look at; and for O two-tailed Kaplan-Meier survival evaluation. Supply knowledge are offered as Supply Information file.
The decreased physique weight of TgLC mice after TAM injection represents a damaging vitality steadiness, indicating modifications in vitality consumption and/or vitality expenditure (EE). To characterize vitality consumption and EE in TgLC mice put up TAM injection, we employed the Promethion Metabolic Cage System. Vitality steadiness, calculated primarily based on vitality consumption and EE, was optimistic throughout the darkish cycle (19:00 pm to 7:00 am) and damaging throughout the gentle cycle (7:00 am to 19:00 pm) in these mice. TAM injection considerably decreased vitality steadiness in TgLC mice throughout the darkish cycle however not throughout the gentle cycle (Fig. 1G). Mice eat most of their meals throughout the darkish cycle20. TAM injection considerably decreased meals consumption in TgLC mice, normalized with or with out mouse physique weight, throughout the darkish cycle however not throughout the gentle cycle (Figs. 1H and S2C). The lower in meals consumption throughout the darkish cycle appeared from the primary darkish cycle put up TAM injection in TgLC mice and have become extra pronounced in later days (Figs. 1H and S2C).
Whole EE (TEE), which displays resting metabolism and bodily exercise, is calculated primarily based on the oxygen consumption (VO2) and carbon dioxide manufacturing (VCO2) by an oblique calorimeter21. TEE was considerably decreased in TgLC mice with TAM injection, normalized with or with out mouse physique weight, in each the darkish and lightweight cycles (Fig. 1I, S2D, E). In contrast with TgL mice, TAM injection decreased VO2 and VCO2 in TgLC mice, normalized with or with out mouse physique weight (Figs. 1J, Okay and S2F–I). This discount in each VO2 and VCO2 was noticed in each the darkish and lightweight cycles, but it surely was extra pronounced at nighttime cycle (Figs. 1J, Okay and S2F–I). The respiratory alternate ratio (RER), calculated because the ratio between VCO2 and VO2, displays the supply of metabolic gas for EE. RER was considerably decreased in TgLC mice put up TAM injection throughout the darkish cycle (Fig. 1L), indicating a shift in metabolic gas from carbohydrate to fats22. TAM injection additionally decreased spontaneous locomotor exercise in TgLC mice throughout each the darkish and lightweight cycles (Fig. 1M). These knowledge exhibit that the decreased meals consumption, decreased TEE, decreased bodily exercise, and decreased RER in TgLC mice with LIF overexpression throughout cachexia improvement collectively led to the decreased vitality steadiness.
Cachexia can result in multi-organ failure1. LIF overexpression in TgLC mice impaired renal and liver features, as mirrored by the decreased serum ranges of albumin and elevated serum ranges of blood urea nitrogen (BUN), which replicate renal operate, and the elevated serum ranges of alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT), which replicate liver operate, at 3 days after TAM injection for serum albumin ranges and 9 days after TAM injection for different parameters (Fig. 1N). TgLC mice with TAM injection had a median survival of 10 days (Fig. 1O). Taken collectively, these outcomes exhibit that the systemic elevation of LIF ranges induces cachexia in mice.
LIF overexpression disrupts lipid homeostasis and reduces hepatic de novo lipogenesis in mice
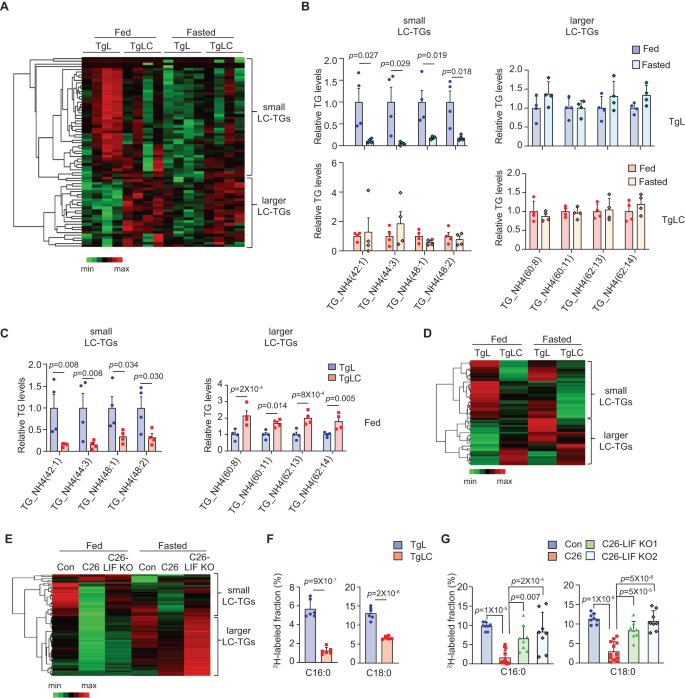
To evaluate the systemic metabolic modifications in TgLC mice with LIF overexpression, we carried out metabolomics analyses to measure polar and lipid metabolites within the serum from TgLC and TgL mice with TAM injection. Contemplating the anorexia induced by LIF overexpression in TgLC mice, we included mice below each fed and fasted circumstances for metabolomics evaluation. A Heatmap evaluation confirmed modifications in polar metabolite ranges within the serum between TgLC and TgL mice with TAM injection below each fed and fasted circumstances (Fig. S3A). Beneath the fed situation, there was a pattern indicating decreases in a number of metabolites concerned in glucose and amino acid metabolism, together with glucose, pyruvate, lactate, glycine, and glutamate in TgLC mice in contrast with TgL mice (Fig. S3B). An analogous change was noticed for some metabolites, together with glucose, pyruvate, and lactate, between TgLC mice and TgL mice below the fasted situation (Fig. S3B). It has now been acknowledged that lipid metabolism, together with triglyceride (TG) hydrolysis, is a serious metabolic pathway concerned within the initiation and/or development of most cancers cachexia4,7,23. Vital modifications in lipid metabolites, notably TGs, have been noticed in TgLC mice with TAM injection in contrast with TgL mice with TAM injection below each fed and fasted circumstances (Figs. 2A and S3C). These TGs are long-chain triglycerides (LC-TGs), composed of fatty acyl chains containing greater than 12 carbon atoms. In TgL mice with TAM injection, below the fed situation, the vast majority of TGs detected within the serum had a carbon chain equal to or lower than 54 (C ≤ 54) (known as small LC-TGs hereafter) (Fig. 2A). Fasting of TgL mice with TAM injection led to a major lower in TG ranges within the serum (Fig. 2A, B). Notably, below the fed situation, the degrees of small LC-TGs have been considerably decrease in TgLC mice with TAM injection in contrast with TgL mice with TAM injection, and the degrees of LC-TGs with a carbon chain bigger than 54 (C > 54) (known as bigger LC-TGs hereafter) have been considerably greater in TgLC mice with TAM injection in contrast with TgL mice with TAM injection (Fig. 2A, C). The modifications in these TGs in TgLC mice with TAM injection weren’t as a consequence of meals consumption, as fasting in TgLC mice didn’t trigger a major change within the majority of TGs (Fig. 2A, B). Additional identification of fatty acids (FAs) that kind TGs confirmed that almost all of small LC-TGs have been composed of saturated fatty acids (SFAs), together with C14:0, C15:0, C16:0 and C18:0, and the vast majority of bigger LC-TGs have been composed of long-chain polyunsaturated fatty acids (LC-PUFAs), together with C20:4, C20:5, C22:4, C22:5, C22:6, C24:6 (Supplementary Desk 1). Notably, these LC-PUFAs are important FAs that can not be synthesized de novo by mice, suggesting that these LC-PUFAs have been primarily produced from the lipolysis of different organs24,25. The liver is a crucial organ concerned in lipid homeostasis. Contemplating that TAM-induced LIF overexpression in TgLC mice led to impaired liver features (Fig. 1N), it’s doable that LIF overexpression disrupts correct lipid metabolism within the liver. Right here, we in contrast the degrees of lipid metabolites within the liver tissues from TgLC and TgL mice with TAM injection below each fed and fasted circumstances. Beneath the fed situation, LIF overexpression in TgLC mice with TAM injection led to an analogous change within the TG ranges within the liver tissues as that noticed within the serum; TAM injection in TgLC mice resulted in a major lower within the ranges of the vast majority of small LC-TGs, which indicated decreased lipogenesis, and a major improve within the ranges of bigger LC-TGs (Fig. 2D). The degrees of TGs have been largely comparable within the liver tissues of TgLC mice with TAM injection below each fed and fasted circumstances (Fig. 2D). Related observations have been made within the liver tissues from Balb/c mice bearing C26 or C26-LIF KO tumors (Fig. 2E). Beneath each fed and fasted circumstances, in contrast with management mice with out tumors, mice bearing C26 tumors however not C26-LIF KO tumors at an analogous dimension exhibited a major lower within the ranges of TGs, particularly small LC-TGs, within the liver (Fig. 2E).
A Heatmap of serum TG ranges below fed and fasted circumstances in TgL and TgLC mice with small LC-TGs (C ≤ 54) and bigger LC-TGs (C > 54) clustered collectively (n = 4/group). B Consultant serum TG ranges in TgL (higher panels) and TgLC (backside panels) mice injected with TAM below fed and fasted circumstances (n = 4/group). The TG ranges below the fed situation are designated as 1. C Consultant serum TG ranges below the fed situation (n = 4/group). The TG ranges in TgL mice below the fed situation are designated as 1. D Heatmap exhibiting the common TG ranges within the livers of TgL and TgLC mice injected with TAM below fed and fasted circumstances (n = 8/group). E Heatmap of the common TG ranges within the livers from Balb/c mice with or with out C26 or C26-LIF KO tumors (n = 6 for management (Con) mice with out tumors, n = 10 for C26 tumor-bearing mice and n = 6 for C26-LIF KO tumor-bearing mice). F, G Hepatic de novo lipogenesis in TgL and TgLC mice with TAM injection (F; n = 6/group) and in non-tumor bearing Balb/c mice (n = 8) and Balb/c mice bearing C26 (n = 12) or C26-LIF KO tumors (n = 8) (G). Mice drank water containing 20% D2O for 7 days earlier than tissue assortment. Ranges of C16:0 and C18:0 in every group have been proven. Information are offered as imply ± SD. Every dot represents a person organic repeat. Each feminine and male mice have been used. For B, C, F Two-tailed Scholar’s t-test was utilized for comparability between two teams; for G One-way ANOVA adopted by t-test with Tukey’s a number of comparability adjustment was utilized for comparability amongst a number of teams. Supply knowledge are offered as Supply Information file.
To straight look at the impact of LIF on hepatic de novo lipogenesis, we employed deuterated water (D2O) tracing experiments to label de novo synthesized FA in vivo26. TgLC and TgL mice with TAM injections have been supplied with consuming water containing 20% D2O for 7 days earlier than the liver tissues have been collected for lipid metabolomics evaluation. LIF overexpression in TgLC mice considerably decreased hepatic de novo lipogenesis as mirrored by the decreased ranges of de novo synthesized C16:0, and C18:0, that are the most typical and plentiful types of SFAs (Fig. 2F). A really comparable lower in hepatic de novo lipogenesis was noticed within the mice bearing C26 tumors however not C26-LIF KO tumors at comparable sizes, suggesting that tumor-secreted LIF inhibits hepatic de novo lipogenesis in mice (Fig. 2G). Taken collectively, these outcomes exhibit that LIF overexpression in mice inhibits hepatic de novo lipogenesis, which can contribute to the decreased ranges of small LC-TGs within the serum and liver tissues in TgLC mice with TAM injection and C26 tumor-bearing mice that develop cachexia.
Blocking the LIF signaling within the liver considerably attenuates cachexia
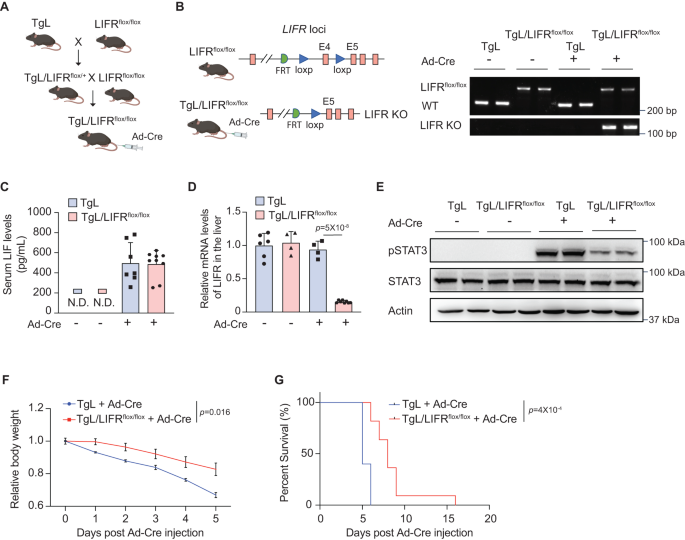
LIF features via binding to its receptor advanced, which consists of LIFR and gp1309. To research the contribution of LIF-induced purposeful and metabolic modifications within the liver to cachexia, we generated a mouse line with a conditional LIF knock-in allele and a conditional LIFR knockout allele (TgL/LIFRflox/flox) (Fig. 3A, B). Liver-specific LIF expression and LIFR knockout have been induced in TgL/LIFRflox/flox mice by hydrodynamic tail vein injection of Ad5CMVCre-eGFP (Advert-Cre) (Fig. 3A, B). TgL mice with Advert-Cre injection that induces liver-specific LIF expression with out LIFR knockout served as controls. Advert-Cre injection induced LIF expression to a comparable stage within the liver of TgL and TgL/LIFRflox/flox mice as decided by ELISA assays, and Advert-Cre injection considerably decreased LIFR mRNA ranges within the liver of TgL/LIFRflox/flox however not TgL mice as decided by quantitative real-time PCR (qPCR) assays (Fig. 3C, D). STAT3 is a serious downstream signaling pathway of LIF, and the degrees of STAT3 phosphorylation at Tyr705 (pSTAT3) can replicate STAT3 exercise11,12,14,18. Advert-Cre injection in TgL mice drastically enhanced the exercise of STAT3 within the liver tissues as mirrored by the elevated ranges of pSTAT3 measured by Western-blot assays, whereas Advert-Cre injection in TgL/LIFRflox/flox mice led to solely a really restricted improve of pSTAT3 ranges within the liver tissue (Figs. 3E and S4). Advert-Cre injection rapidly induced cachexia in TgL mice, which exhibited physique weight reduction and brief survival (median survival of 5 days) (Fig. 3F, G). Notably, TgL/LIFRflox/flox mice with Advert-Cre injection which have liver-specific LIFR knockout to dam the LIF signaling exhibited a much less pronounced physique weight reduction and extended survival in contrast with TgL mice with Advert-Cre injection (Fig. 3F, G). These outcomes exhibit that blocking the LIF signaling within the liver by liver-specific LIFR knockout considerably attenuates LIF-induced cachexia, suggesting that LIF-induced purposeful modifications within the liver, together with its metabolic modifications, contribute to LIF-induced cachexia.
A The technology of TgL/LIFRflox/flox mice. Liver-specific LIF expression and LIFR knockout was induced in TgL/LIFRflox/flox mice by hydrodynamic tail vein injection of Ad5CMVCre-eGFP (Advert-Cre). B The genotyping evaluation of TgL and TgL/LIFRflox/flox mice with or with out Advert-Cre injection by PCR. All mice with the identical genotype have comparable outcomes. C Serum LIF ranges in TgL and TgL/LIFRflox/flox mice with or with out Advert-Cre injection (n = 7–9/group). D Relative LIFR mRNA ranges in TgL and TgL/LIFRflox/flox mice with or with out Advert-Cre injection (n = 4–8/group). E The degrees of Tyr 705 phosphorylated STAT3 (pSTAT3) and complete STAT3 protein within the liver of TgL and TgL/LIFRflox/flox mice with or with out Advert-Cre injection decided by Western-blot assays. No less than three impartial organic replicates have been carried out. F Relative physique weight of TgL (n = 5) and TgL/LIFRflox/flox (n = 10) mice put up Advert-Cre injection. G Kaplan-Meier survival curves of TgL and TgL/LIFRflox/flox mice put up Advert-Cre injection. The day of Advert-Cre injection was denoted as D0. Information are offered as imply ± SD for (C, D), and as imply ± SEM for (F). N.D. non-detectable. Every dot represents a person organic repeat. Each feminine and male mice have been used. For D: one-way ANOVA adopted by t-test with Tukey’s a number of comparability adjustment; for F: two-way ANOVA adopted by Sidak’s a number of comparability take a look at; for G: two-tailed Kaplan-Meier survival evaluation. Supply knowledge are offered as Supply Information file.
LIF overexpression reduces the expression of lipogenesis genes in TgLC mice
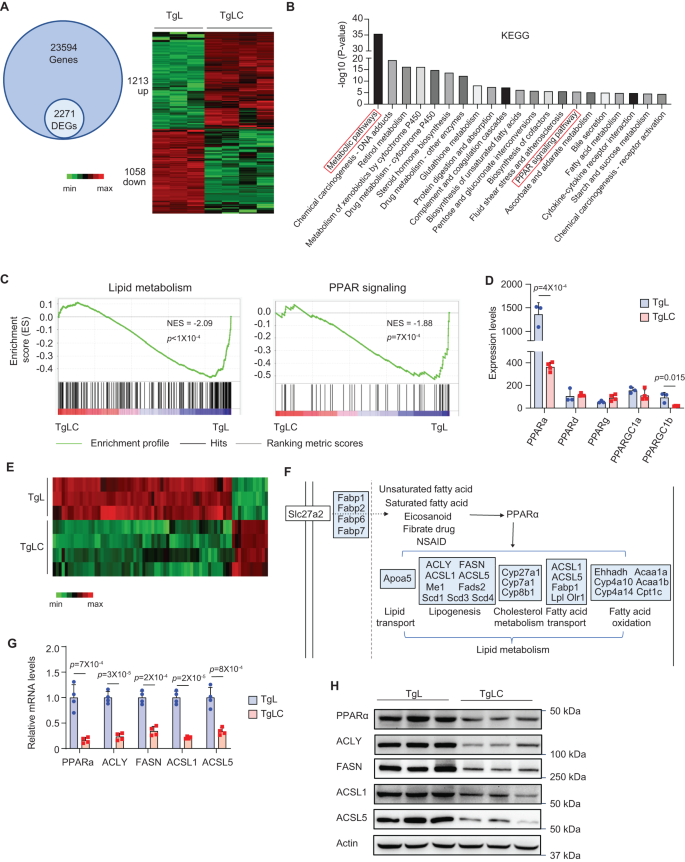
To research the underlying mechanism by which LIF overexpression impairs hepatic de novo lipogenesis, we carried out transcriptome evaluation utilizing RNA-seq assays to match the gene expression profiles of the liver tissues of TgLC and TgL mice with TAM injection. Amongst a complete of 23,594 genes examined, there have been 2271 differentially expressed genes (DEGs) between the liver tissues of TgLC and TgL mice with TAM injection, with 1213 genes being upregulated, and 1058 genes being downregulated (Fig. 4A). Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment evaluation of the DEGs revealed that the top-ranked pathway was metabolic pathways (Fig. 4B), which aligns with the outcomes from the metabolomics evaluation exhibiting modifications within the ranges of many metabolites within the liver tissues of TgLC mice with TAM injection. PPARs are nuclear receptors that operate as transcription elements regulating the expression of genes concerned in lipid metabolism27,28. KEGG evaluation confirmed that most of the DEGs have been enriched within the PPAR signaling pathway (Fig. 4B). Gene-set enrichment evaluation (GSEA) revealed that each lipid metabolism and PPAR signatures have been downregulated within the livers of TgLC mice with TAM injection (Fig. 4C). The outcomes of the RNA-seq assays confirmed that among the many 5 PPAR member of the family genes (PPARa, PPARd, PPARg, PPARGC1a and PPARGC1b), the mRNA ranges of PPARa and PPARGC1b have been considerably decreased within the liver tissues of TgLC mice with TAM injection in contrast with TgL mice with TAM injection (Fig. 4D). PPARa is probably the most plentiful of the PPAR household that’s expressed within the liver; its basal expression ranges in liver tissues have been a lot greater than these of different PPAR relations (Fig. 4D). The qPCR outcomes confirmed that the expression of PPARa is most plentiful in mouse liver tissues in contrast with its expression in different tissues, together with the guts, muscle, and spleen (Fig. S5A). PPARa encodes for PPARα, a grasp transcription issue for a number of genes concerned in lipogenesis, together with ACLY, FASN, ACSL1, and ACSL529,30. The RNA-seq knowledge confirmed differential expression of 72 PPARα targets within the liver tissues of TgLC mice with TAM injection in contrast with TgL mice with TAM injection (Fig. 4E). Amongst them, 25 DEGs mapped to the PPARα signaling pathway are concerned in lipid metabolism, together with 9 genes (ACLY, FASN, ACSL1, ACSL5, ME1, FADS2, SCD1, SCD3 and SCD4) concerned in lipogenesis (Fig. 4F). The expression modifications of PPARα and 4 goal genes (ACLY, FASN, ACSL1, ACSL5) have been validated at each mRNA and protein ranges by qPCR and Western-blot assays, respectively (Fig. 4G, H). The lower in PPARα and ACLY ranges was predominantly noticed in hepatocytes within the livers of TgLC mice with TAM injection as examined utilizing immunohistochemistry (IHC) assays (Fig. S5B). No apparent modifications within the PPARa mRNA ranges have been noticed within the livers of the pair-fed TgL mice with TAM injection, indicating that the lower of PPARα shouldn’t be as a consequence of decreased meals consumption (Fig. S5C). Persistently, a major lower within the mRNA ranges of PPARa, ACLY, FASN, ACSL1 and ACSL5 was noticed within the livers of C26 tumor-bearing mice in contrast with management mice (Fig. S5D). Notably, little to no change within the mRNA ranges of those genes was noticed within the livers of C26-LIF KO tumor-bearing mice (Fig. S5D). These outcomes reveal that LIF overexpression decreases the expression ranges of PPARa and its goal genes, particularly these concerned in lipogenesis in liver tissue, which can result in decreased hepatic lipogenesis in TgLC mice with TAM injection and C26 tumor-bearing mice.
A RNA-seq outcomes exhibiting gene expression ranges within the liver from TAM-injected TgLC (n = 4) and TAM-injected TgL mice (n = 3). The variety of recognized genes and DEGs have been proven (left). The DEGs have been proven within the Heatmap (proper). B KEGG evaluation of DEGs by the DAVID database. C GSEA enrichment plots for lipid metabolism (left) and PPAR signaling pathway (proper). D RNA-seq outcomes exhibiting the expression ranges of genes (PPARa, PPARd, PPARg, PPARGC1a and PPARGC1b) encoding for 5 PPAR relations (n = 3 for TgL group, n = 4 for TgLC group). E Heatmap of PPARα goal genes among the many DEGs within the liver from TgLC and TgL mice with TAM injection. F KEGG map of PPARα signaling pathway. DEGs have been mapped to the “PPARα signaling pathway”, in keeping with the “PPARs signaling pathway” map in KEGG with some modifications. DEGs are coloured in blue. G, H Validation of expression ranges of PPARa and a few of its goal genes after LIF overexpression by qPCR assays (G; n = 4/group) and Western-blot assays (H). No less than three impartial organic replicates have been carried out. All knowledge are offered as imply ± SD. Every dot represents a person mouse. Each feminine and male mice have been used. For D, G: two-tailed Scholar’s t-test. Supply knowledge are offered as Supply Information file.
LIF overexpression downregulates the expression of PPARa by way of the activation of STAT3 signaling in hepatic cells
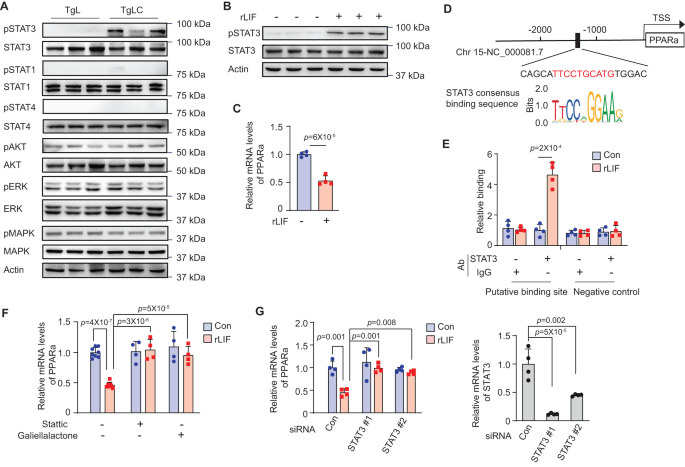
LIF exerts its features via the regulation of assorted downstream signaling pathways in a extremely tissue-, development- and context-specific method9. To research the mechanism underlying the downregulation of PPARa expression in liver tissue by LIF overexpression, we examined a panel of LIF-regulated downstream pathways, together with the STATs, AKT, ERK, and MAPK signaling pathways, within the liver tissues of TgLC and TgL mice with TAM injection. LIF overexpression induced by TAM in TgLC mice clearly enhanced the exercise of STAT3 in liver tissues, as mirrored by elevated ranges of pSTAT3, with out affecting the entire STAT3 protein ranges measured by Western-blot and IHC assays (Figs. 5A and S6A). No main modifications within the pSTAT3 ranges have been noticed within the liver tissues of the pair-fed TgL mice with TAM injection in comparison with the liver tissues of the regular-fed TgL mice with TAM injection (Fig. S6B). Except for STAT3 signaling, there was no clear activation of different signaling pathways within the livers of TgLC mice with TAM injection (Fig. 5A). Equally, recombinant mouse LIF protein (rLIF) therapy elevated pSTAT3 however not complete STAT3 protein ranges in main cultured mouse hepatic cells remoted from wild-type C57BL6/J mice (Fig. 5B). rLIF therapy additionally markedly decreased the expression of PPARa in main mouse hepatic cells (Fig. 5C). A putative STAT3 binding website was recognized within the promoter of the PPARa gene (Fig. 5D). Using chromatin immunoprecipitation (ChIP) assays, we discovered that the anti-STAT3 antibody can immunoprecipitate chromatin fragments similar to the potential STAT3 binding website within the PPARa promoter in hepatic cells handled with rLIF, however not in cells with out rLIF therapy, indicating that LIF prompts STAT3 to advertise its binding to the PPARa promoter (Fig. 5E). To additional examine whether or not LIF-induced STAT3 activation mediates the downregulation of PPARa by LIF in hepatic cells, we examined the impact of rLIF on PPARa expression within the main hepatic cells handled with small molecule inhibitors particular for STAT3 and siRNAs focusing on STAT3, respectively, to dam STAT3 signaling. The downregulation of PPARa expression by rLIF in main hepatic cells was largely abolished by two small molecule STAT3 inhibitors, Stattic and Galiellalactone, in addition to by two siRNA oligos focusing on STAT3 (Fig. 5F, G). Collectively, these outcomes point out that LIF overexpression downregulates PPARa expression in hepatic cells, primarily via the activation of STAT3 signaling.
A LIF overexpression elevated the degrees of pSTAT3 however not complete STAT3 protein or different LIF downstream pathways, together with STAT1, STAT4, AKT, ERK, and MAPK within the liver of TgLC mice put up TAM injection as decided by Western-blot assays. No less than three impartial organic replicates have been carried out. B rLIF therapy (100 ng/ml for 30 min) of main mouse hepatic cells elevated the degrees of pSTAT3 however not complete STAT3 protein as decided by Western-blot assays. Three replicates have been offered in every group. No less than three impartial organic replicates have been carried out. C rLIF therapy decreased the mRNA ranges of PPARa in main cultured hepatic cells (n = 4/group). D The sequence and placement of a putative STAT3 binding website within the mouse PPARa promoter area. TSS: transcription begin website. E rLIF elevated the binding of STAT3 to a putative STAT3 binding website within the promoter of PPARa as decided by ChIP assays in main mouse hepatic cells. A chromatin area with out STAT3 binding website was included as a damaging management (n = 4/group). F Blocking STAT3 by STAT3 inhibitors, Stattic (2 μM) or Galiellalactone (5 μM), largely abolished the inhibitory impact of rLIF on the expression of PPARa in main mouse hepatic cells. The mRNA ranges of PPARa have been decided by qPCR assays and normalized to β-actin (n = 4–8/group). G STAT3 siRNAs largely abolished the inhibitory impact of rLIF on PPARa expression in main mouse hepatic cells. Left: relative PPARa mRNA ranges; proper: relative STAT3 mRNA ranges in main cultured hepatic cells (n = 4/group). All knowledge are offered as imply ± SD. Each feminine and male mice have been used. For C, E: two-tailed Scholar’s t take a look at; for F, G: one-way ANOVA adopted by t-test with Tukey’s a number of comparability adjustment. Supply knowledge are offered as Supply Information file.
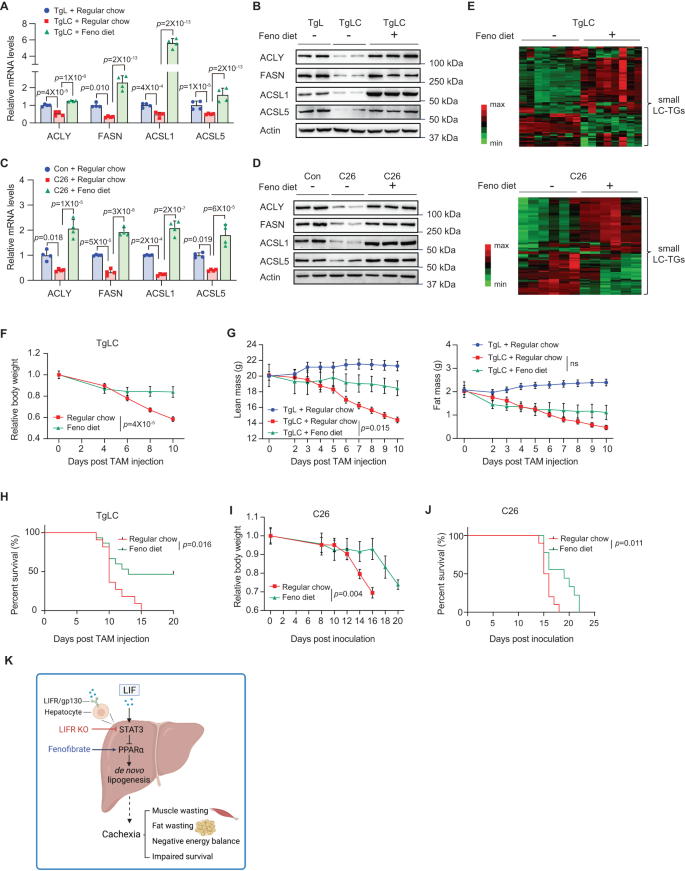
Activating PPARα by fenofibrate restores lipid homeostasis within the liver and inhibits cachexia
Our outcomes exhibit that LIF overexpression downregulates PPARα expression and its goal genes concerned in lipogenesis, which recommend that LIF overexpression could lower lipogenesis and disrupt lipid homeostasis within the liver, thereby inducing cachexia. To check this speculation, we employed fenofibrate, a fibric acid spinoff extensively used as a PPARα agonist31, to research whether or not PPARα activation ameliorates the disrupted lipid metabolism induced by LIF overexpression. Whereas LIF overexpression decreased the expression of ACLY, FASN, ACSL1, and ACSL5 within the liver tissues of TgLC mice injected with TAM, their mRNA and protein ranges have been considerably greater in TgLC mice fed with a fenofibrate-containing eating regimen beginning 3 days earlier than TAM injection (Fig. 6A, B). In distinction, in TgL mice with TAM injection, fenofibrate eating regimen didn’t considerably influence the expression of those genes within the liver tissues the place PPARa ranges have been excessive (Fig. S7A). Equally, whereas the expression ranges of ACLY, FASN, ACSL1, and ACSL5 within the liver tissues have been decreased in C26 tumor-bearing mice in contrast with management mice with out tumors, their expression ranges have been considerably elevated within the liver tissues of the mice fed with a fenofibrate eating regimen (Fig. 6C, D). These outcomes clearly present that LIF overexpression downregulates the expression of PPARα and its downstream targets within the liver, and that the downregulation of PPARα goal genes by LIF overexpression could be blocked by fenofibrate.
A, B mRNA ranges (A; n = 4/group) and protein ranges (B) of PPARα goal genes within the livers of TgLC mice fed with common chow or fenofibrate (Feno) eating regimen (0.2% w/w). C, D mRNA ranges (C; n = 4/group) and protein ranges (D) within the liver of Balb/c mice bearing with or with out C26 tumors fed with common chow or fenofibrate eating regimen. E Fenofibrate eating regimen elevated the degrees of majority of small LC-TGs (C ≤ 54) within the liver of TgLC mice and C26 tumor-bearing mice. Heatmap exhibiting the TG ranges within the liver of TgLC mice (n = 8/group) and C26 tumor-bearing mice (n = 6/group) fed with common chow or fenofibrate eating regimen. F Physique weight of TgLC mice fed with common chow or fenofibrate eating regimen (n = 5/group). G Lean mass (left) and fats mass (proper) of TgLC mice fed with common chow or fenofibrate eating regimen put up TAM injection. n = 6–8/group. H Kaplan-Meier survival curves of TgLC mice fed with common chow or fenofibrate eating regimen. I Physique weight of C26 tumor-bearing mice fed with common chow or fenofibrate eating regimen. The day of C26 cells inoculation was denoted as D0. n = 5/group. J Kaplan-Meier survival curves of C26 tumor-bearing mice. Okay The diagram depicting the mechanism by which LIF induces cachexia. The diagram was created with BioRender.com. Information are offered as imply ± SD for (A, C), and as imply ± SEM for (F, G, I). Every dot represents a person mouse. Each feminine and male mice have been used. ns: non-significant. For A, C: one-way ANOVA adopted by t-test with Tukey’s a number of comparability adjustment; for F, G, I: two-way ANOVA adopted by Sidak’s a number of comparability take a look at; for H, J: two-tailed Kaplan-Meier survival evaluation. Supply knowledge are offered as Supply Information file.
We then investigated whether or not fenofibrate can restore hepatic lipid homeostasis in TgLC mice with LIF overexpression. Whereas TAM injection in TgLC mice led to decrease ranges of small LC-TGs and better ranges of bigger LC-TGs within the liver, fenofibrate considerably elevated the degrees of the vast majority of small LC-TGs, and decreased the degrees of many bigger LC-TGs within the liver of TAM-injected TgLC mice (Figs. 6E and S7B). An analogous impact of fenofibrate on lipid metabolites was noticed in C26 tumor-bearing mice; in contrast with the livers of C26 tumor-bearing mice below the fed situation, which had a majority of small LC-TGs at decrease ranges than management mice, fenofibrate considerably elevated the degrees of many small LC-TGs within the livers of C26 tumor-bearing mice (Fig. 6E).
To additional examine the contribution of impaired lipid metabolism within the liver to cachexia induced by LIF overexpression, we examined whether or not fenofibrate inhibits cachexia improvement in each TgLC and C26 tumor-bearing mice. Notably, fenofibrate considerably decreased the physique weight lack of TgLC mice put up TAM injection (Fig. 6F). TgLC mice with TAM injection fed with fenofibrate eating regimen confirmed a pattern of improved meals consumption in contrast with TgLC mice with TAM injection fed with common chow (Fig. S7C). Moreover, fenofibrate largely blocked lean mass loss and likewise exhibited a pattern of much less in depth fats mass loss, though the distinction in fats mass loss was not vital (Fig. 6G). Importantly, fenofibrate extended the survival of TgLC mice with TAM injection (Fig. 6H). Related outcomes have been obtained in C26 tumor-bearing mice; fenofibrate considerably delayed physique weight reduction and extended the survival of C26 tumor-bearing mice (Fig. 6I, J). Collectively, these outcomes exhibit that fenofibrate prompts PPARα and its downstream targets concerned in lipogenesis within the liver to ameliorate the impaired lipid metabolism induced by LIF overexpression in TgLC mice and C26 tumor-bearing mice, which in flip inhibits cachexia improvement (Fig. 6K).