Liver cancers show a mess of distinct invasive behaviors
Liver most cancers structure was assessed by histological examination of patient-derived samples with background liver, HCC confined inside a fibrotic capsule, and HCC invading into the fibrotic stroma (Fig. 1a). Within the case of HCC encapsulated by a fibrotic capsule, liver most cancers cells expanded by collectively rising in opposition to the encircling setting (Fig. 1a, iv-v). HCC might additionally work together with the encircling stroma with discrete HCC cells invading into the fibrotic stroma (Fig. 1a, vii-viii). Furthermore, tumor cells confined by the encircling fibrotic capsule and people which invade into the stroma might each be discovered inside the identical tumor (Supplementary Fig. 1). We carried out E-cadherin IHC on our tissue samples as an epithelial cell indicator (Fig. 1a, iii, vi, ix). Liver most cancers cells confined inside the fibrotic capsule confirmed larger ranges of E-cadherin expression whereas the stromally invasive liver most cancers cells had much less E-cadherin expression. This additional indicated the presence of two distinct liver most cancers cell subtypes with the encapsulated liver most cancers being extra epithelial and the stromally invasive most cancers being much less epithelial. These two most cancers cell subpopulations have been reported in different research and should have the various useful functionality and subsequently therapeutic response25. We developed a easy hypothetical schematic to spotlight the distinct options of those two subtypes (Fig. 1b).
a Background liver exhibits properly ordered liver lobule and portal tract with gentle power irritation, portal and periportal fibrosis (i–iii), HCC encapsulated by a fibrotic capsule (iv–vi), and HCC tumor cells invading into the fibrotic stroma (vii–ix). Samples are hematoxylin and eosin stained. Decrease panels present IHC staining of E-cadherin in roughly corresponding areas. Scale bars: 200 μm. Yellow arrows point out stromal invasion of HCC in collagen whereas black arrows point out bile ductules. b Our working mannequin illustrates invasion modes of HCC. Whereas HCC tumor cells can proliferate in opposition to the fibrotic capsule, HCC tumor cells also can invade into the fibrotic stroma. HCC tumor cells can ship out dynamic protrusions and exert contractile forces on the collagen to permit for alignment and densification which can facilitate additional invasion. Made with Biorender.com.
In a single case, HCC may be confined by the matrix (Fig. 1b, left). As such, this subtype shows much less native dissemination and is surrounded by a fibrotic stroma. This fibrotic confinement has been noticed in a wide range of strong tumor varieties like HCC, neuroendocrine tumors, renal cell carcinoma, and adrenal cortical carcinoma2,26. Invasive cells in strong tumors also can migrate into the encircling stroma. Different research have discovered that invasive cells are sometimes extremely energetic and may ship out dynamic protrusions which exert contractile forces on collagen to permit alignment and densification which facilitates additional invasion (Fig. 1b, proper)9,15,27. This speculation of collagen reworking facilitating subsequent invasion is in line with different tumor varieties together with breast, lung, and colorectal most cancers28,29,30. These research have proven that invasive cells have distinct cytoskeletal signatures that allow invasion and cell-matrix interactions. By taking a look at cell strains which might be consultant of those subpopulations, we sought to grasp the mechanobiological variations between liver most cancers subpopulations and the way their distinct cytoskeletal options play a job in invasive conduct.
SNU-475 and HepG2 cells have distinct biochemical, bodily, and invasive signatures
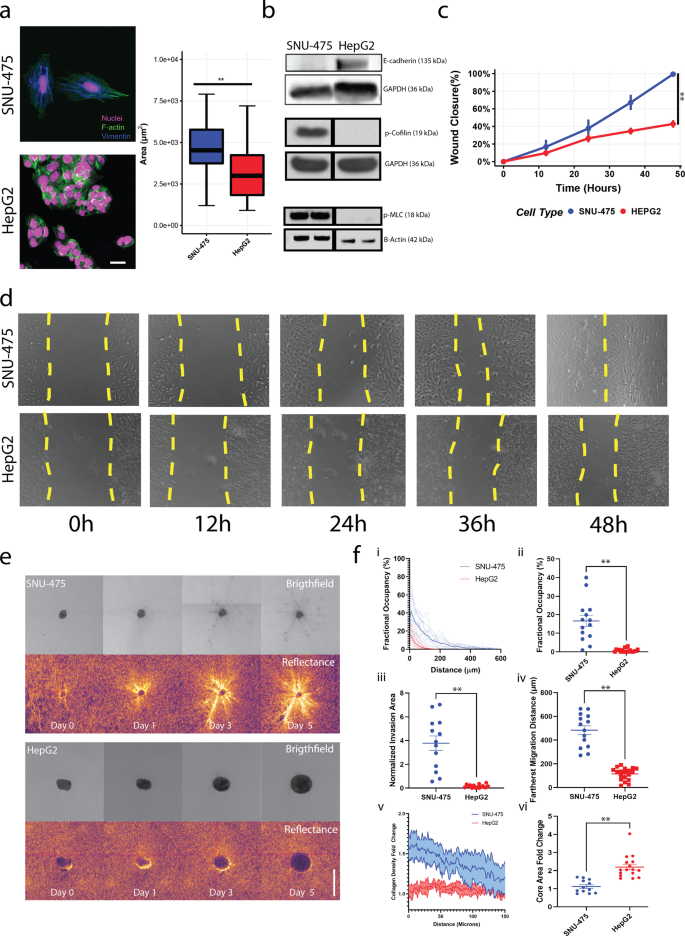
To review the differential responses of liver most cancers cells in 2D and 3D environments, we in contrast the SNU-475 cell line, which is extra invasive and mesenchymal, and the HepG2 cell line, which is much less invasive and extra epithelial19. We first assessed the variations between these two cell strains on 2D collagen-coated surfaces. Collagen was chosen as a result of it’s the major part of the ECM and is a serious ligand for integrin and subsequent cytoskeletal equipment in cell migration31. Fluorescent staining of each strains confirmed that SNU-475 cells had pronounced actin stress fibers and had an elongated mobile morphology, whereas HepG2 cells didn’t have these stress fibers and tended to be rounded and clump collectively (Fig. 2a). Such morphology led to decreased general cell space in HepG2 cells in comparison with SNU-475 cells. Immunoblotting indicated that HepG2 cells have larger ranges of E-cadherin than do SNU-475 cells (Fig. 2b, Supplementary Fig. 2). Moreover, HepG2 cells had considerably decrease ranges of phosphorylated cofilin (p-cofilin) and phosphorylated myosin II gentle chain (p-MLC) relative to SNU-475 cells (Fig. 2b), each of that are key signaling molecules in actin turnover and have been implicated in metastatic behaviors32. The excessive ranges of E-cadherin in HepG2 cells confirmed that these cells are extra epithelial-like and advised that the HepG2 cells have much less migratory functionality than SNU-475 cells33. We subsequent carried out scratch assays on collagen-coated surfaces to look at wound closure charge (Fig. 2c). In 48 hours, SNU-475 cells had been capable of totally shut the wound, however HepG2 cells had been solely capable of shut the wound 50% (Fig. 2nd). HepG2 cells require as much as ~144 hours to shut wounds after making the scratch. At the side of our histological evaluation, these outcomes demonstrated that our cell strains examined are consultant of distinct most cancers subtypes seen in vivo.
a Consultant immunofluorescent photos of SNU-475 cells and HepG2 cells. Inexperienced is F-actin, magenta is DAPI, and blue is Vimentin. Scale bar is 25 μm. b Immunoblotting of SNU-475 and HepG2 cells for E-cadherin, p-cofilin, and p-MLC. Black vertical separation strains for p-cofilin and p-MLC blot point out that measurements are separated lanes of the identical blot. c Scratch assay analyses and d corresponding consultant photos of scratch assays for SNU-475 and HepG2 wounds over 48 h. Yellow dotted strains point out wound space. Single dotted line signifies that the wound is closed. N = 11 replicates for every cell line. e SNU-475 (high panel) and HepG2 spheroids (backside panel) embedded in collagen matrices and imaged over 5 days. Photographs depict brightfield and reflectance microscopy of every spheroid. Scale bar: 500 μm. f After 5 days of tradition in collagen gel, the share occupancy of the disseminated cells at every of the distances from the spheroid periphery is calculated (i) and the share occupancy at 100 μm away from the spheroid periphery are in contrast between the 2 cell strains (ii). The full areas of the disseminated cells after normalization (iii) and the farthest cell migration distances (iv) are in contrast between the 2 cell strains. The collagen densification as a operate of distance from the spheroid (v) and core space fold change are in contrast throughout cell strains (vi). Plots present imply ± SEM. N = 14 spheroids for every cell line.Unpaired t-test with Welch’s correction is carried out. *P ≤ 0.05, **P ≤ 0.01.
To higher perceive liver most cancers cell motility and tumor aggressiveness in a extra in vivo–like setting, we grew SNU-475 and HepG2 cells into spheroids and encapsulated them in 3D collagen hydrogels. The escape of cells out of those spheroids was monitored over 5 days (Fig. 2e). SNU-475 spheroids tended to kind spherical shapes, whereas HepG2 spheroids tended to be flat and unfold out. As described beforehand34, HepG2 cells doubled extra rapidly than SNU-475 cells (48 h vs. 66 h), explaining variations in closing spheroid dimension. Qualitative evaluation of collagen matrix remodelling was noticed by brighter reflectance alerts which have been used beforehand to explain collagen reworking (Fig. 2e)35. SNU-475 cells tended to detach from the spheroid and migrated into the encircling gel greater than HepG2 cells (Fig. 2f, i-ii). After cells escaped from the spheroid, SNU-475 cells migrated a lot farther than HepG2 cells (Fig. 2f, iii-iv). SNU-475 spheroids brought on extra matrix reworking than HepG2 spheroids (Fig. 2f, v). HepG2 cells tended to not escape out of the spheroid, leading to an general enlargement of the HepG2 spheroid that was higher than SNU-475 spheroid enlargement (Fig. 2f, vi). Total, these comparisons spotlight the stark variations in SNU-475 and HepG2 cells molecularly and in motility in each 2D and 3D.
SNU-475 and HepG2 cell strains have distinct 2D responses to cytoskeletal inhibitors
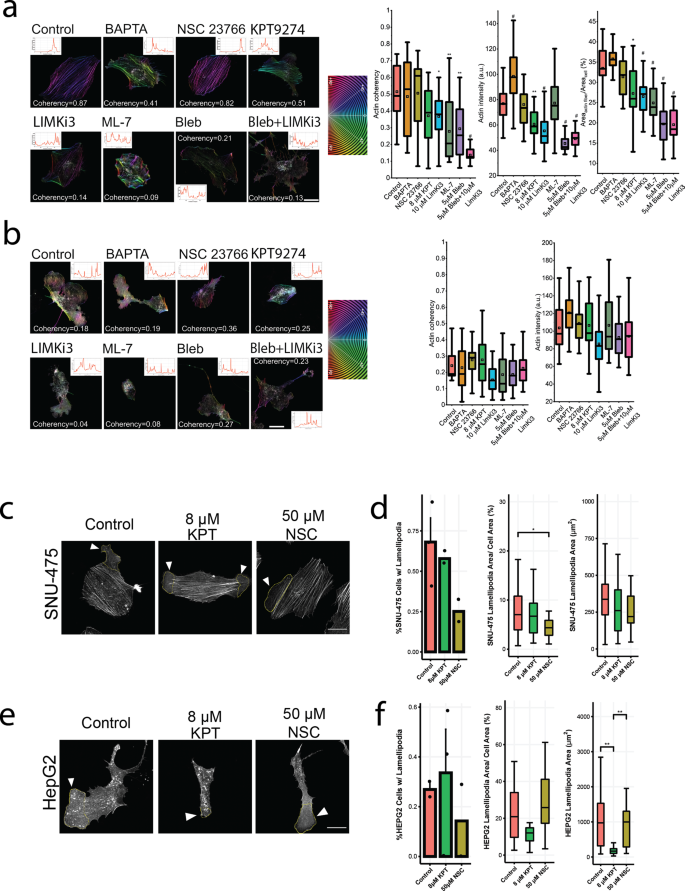
To establish pharmacological means to change 2D cell migration in liver most cancers, we focused key regulators of the Rho GTPases, together with calcium signaling, Rac1, PAK4, LIMK1, MLCK, and actomyosin contractility (Fig. 3a). Scratch assays over 48 h (SNU-475 cells) or 144 h (HepG2 cells) allowed evaluation of wound closure beneath drug therapy (Fig. 3b, Supplementary Fig. 3). For each cell strains, we additionally carried out scratch assays throughout Sorafenib therapy, which is probably the most generally used therapy for HCC36. HepG2 cells handled with Sorafenib had low viability and didn’t shut the wound. Along with therapy with single medication, we additionally used a double drug therapy to check for synergy amongst pathways. Specifically, we examined actomyosin contractility and LIMK1 inhibition co-treatment ((S)-4′-nitro blebbistatin (blebbistatin) + LIMKi3, respectively). This mixture allowed evaluation of the results of PAK4, which is ready to phosphorylate LIMK1 and, in flip, phosphorylate cofilin to its inactive kind in addition to management actomyosin contractility by way of the Rho-ROCK signaling axis37. For the SNU-475 cells, all medication apart from calcium inhibition by way of BAPTA had been capable of considerably inhibit wound closure. To interrogate cytoskeletal equipment that’s straight downstream of calcium signaling, we examined inhibition of the calcium-dependent myosin gentle chain kinase (MLCK). We discovered a major lower in wound closure for each cell strains after MLCK inhibition (Fig. 3b). The blebbistatin-LIMKi3-cotreated SNU-475 cells achieved wound closure to ranges corresponding to the blebbistatin therapy alone (Fig. 3b, left panel). For the HepG2 cells, nevertheless, solely inhibition of PAK4, LIMK1, and twin inhibition of LIMK1 and actomyosin contractility considerably inhibited wound closure (Fig. 3b, proper panel). Taken collectively, these knowledge recommend that SNU-475 cells are extra delicate to totally different cytoskeletal perturbations than HepG2 cells.
a Pathway diagram illustrating the impact of every drug used on cytoskeletal equipment. b Scratch assay bar plots for SNU-475 and HepG2 for all medication examined. Scratch assay bar plots at 48 h and 144 h for all medication examined for SNU-475 and HepG2 cells, respectively. N ≥ 2 replicates. Plots present imply ± SEM with circumstances the place N > 2. c Consultant immunofluorescence photos of SNU-475 spheroids (first row) and HepG2 spheroids (second row) handled with BAPTA (Ca2+ chelator), NSC23766 (Rac1 inhibitor), KPT9274 (PAK4 inhibitor), LIMKi3 (LIMK inhibitor), blebbistatin (myosin inhibitor inhibitor), each blebbistatin and LIMKi3, aor ML-7 (MLCK inhibitor), respectively. Inexperienced represents F-actin and magenta is DAPI. Inexperienced represents F-actin and magenta is DAPI. Scale bar: 30 μm. d SNU-475 (i) and HepG2 (ii) mobile space and solidity. n > 30 cells for every situation from N = 2 unbiased experiments. e p-MLC western blots for drug circumstances for SNU-475 and HepG2 cells. Plots present imply ± SD. One-way ANOVA with Tukey post-hoc testing was carried out. Important distinction (p < 0.05) was detected between any two of the circumstances. *P < 0.05, **P < 0.01.
To higher perceive how these medication inhibit migratory phenotypes, quantitative immunofluorescence was monitored to find out results on cell morphology. We quantified nuclear side ratio, nuclear circularity, cell side ratio, cell circularity, cell space, and mobile solidity (Fig. 3c, Supplementary Figs. 4–5). Each PAK4 and LIMK1 inhibition decreased cell spreading space in SNU-475 cells considerably (Fig. 3d, i). In distinction, PAK4 inhibition, however not LIMK1 inhibition, was capable of considerably enhance cell circularity and reduce HepG2 cell spreading (Fig. 3d, ii). Though LIMK1 appeared to inhibit HepG2 wound closure charge, it didn’t have an effect on HepG2 nuclear or mobile morphology. Curiously, PAK4 inhibition brought on HepG2 cells to have elevated solidity (Fig. 3d, ii). This was a results of the lower within the unfold space in addition to a lower in lamellipodial dimension. Blebbistatin therapy considerably decreased cell circularity, cell space, and cell solidity for SNU-475 cells. Co-treatment with LIMKi3 additional exacerbated this impact (Fig. 3d, i). There was no vital change in nuclear side ratio or circularity for blebbistatin or blebbistatin + LIMKi3 handled HepG2 cells (Supplementary Fig. 5e-f). There was a lower in HepG2 cell circularity with 5 or 10 μM blebbistatin, or 5 μM blebbistatin + 10 μM LIMKi3 therapy (Fig. 3d, ii). This lower in HepG2 cell solidity was noticed with both 10 μM blebbistatin therapy or 5 μM blebbistatin + 5 μM LIMKi3 therapy (Fig. 3d, ii). MLCK inhibition by way of ML-7 therapy decreased mobile solidity and circularity in SNU-475 cells (Fig. 3d, i, Supplementary Fig. 4d). Curiously, ML-7 therapy decreased HepG2 unfold space and subsequently brought on a rise in cell circularity (Fig. 3d, ii, Supplementary Fig. 5b). To confirm the impact of those medication, we carried out Western blotting after drug therapy (Fig. 3e, Supplementary Figs. 6–9). LIMKi3 therapy was capable of considerably lower p-MLC expression ranges in SNU-475 cells whereas KPT therapy confirmed a pattern in direction of reducing p-MLC ranges in SNU-475 cells. We additionally discovered reducing tendencies in p-cofilin after KPT or LIMKi3 therapy for HepG2 cells (Fig. 3e). Total, these knowledge recommend that inhibition of actomyosin contractility will lower 2D cell migration greater than cofilin-mediated actin turnover in SNU-475 cells. Conversely, our knowledge additionally recommend that HepG2 2D cell migration is extra depending on cofilin-mediated actin turnover than actomyosin contractility.
Actin and lamellipodial dynamics are regulated otherwise in SNU-475 vs. HepG2 cells
Based mostly on the discrete interactions of every drug on mobile form, we puzzled if these form adjustments had been a results of adjustments in mobile actin group or construction. We measured actin coherency (diploma of alignment of actin stress fibers), actin depth, and F-actin to cell space ratio. For the SNU-475 cells, actin coherency was decreased when cells had been handled with LIMKi3, blebbistatin, the co-treatment of the 2, or ML-7 (Fig. 4a). Except for Rac1 inhibition by way of NSC23766 therapy and MLCK inhibition by way of ML-7, all medication had been capable of lower actin depth. Except for BAPTA and NSC23766 therapy, all medication are capable of lower the F-actin space to cell space ratio. For the HepG2 cells, there have been only a few or no actin stress fibers, so there are not any measures of actin coherency out there. All the drug remedies examined considerably modified HepG2 actin depth and F-actin space to cell space ratio (Fig. 4b).
Coherency measurement on F-actin as an indicator for cytoskeleton group and cell morphology beneath drug remedies for a SNU-475 and b HepG2. Stress fiber-rich HCC cell line SNU-475 incorporates extra organized actin and well-defined morphology and is extra delicate to the cytoskeleton medication, in comparison with HepG2. Significance is in contrast between management and every drug situation. (From left to proper) Coherency of actin stress fiber (SF) depth and proportion of SF of complete F-actin. n > 12 cells for every situation from N = 2 unbiased experiments. a Rac1 inhibitor (NSC23766) doesn’t have an effect on stress fiber formation however reduces lamellipodia. BAPTA appears to induce stress fiber build up and induce skinny protrusions on the cell periphery. b The chosen inhibitors don’t considerably cut back complete F-actin depth for HepG2 cells. c Consultant photos denoting traced lamellipodia for SNU-475 cells. Yellow define signifies lamellipodia tracing. d Metrics denoting SNU-475 variety of cells with lamellipodia, lamellipodia space proportion of cell space, and lamellipodia space after no therapy, 8 μm KPT therapy, and 50 μm NSC therapy. e Consultant photos denoting traced lamellipodia for HepG2 cells. Yellow define signifies lamellipodia tracing. f Metrics denoting HepG2 variety of cells with lamellipodia, lamellipodia space proportion of cell space, and lamellipodia space after no therapy, 8 μm KPT therapy, and 50 μm NSC therapy. Plots present imply ± SEM. n > 12 cells for every situation from N = 2 unbiased experiments. One-way ANOVA with Tukey post-hoc testing was carried out. Important distinction (p < 0.05) was detected between any two of the circumstances. *P < 0.05, **P < 0.01, #P < 0.0001.
Because of the interaction of PAK4 and Rac1, we investigated the function of those two pathways in lamellipodial formation38. In line with earlier research, we discovered that Rac1 inhibition was adequate to indicate reducing lamellipodial quantity and to considerably lower lamellipodial dimension in SNU-475 cells (Fig. 4c-d). PAK4 inhibition didn’t considerably have an effect on lamellipodial dimension (Fig. 4d). Curiously, HepG2 lamellipodia formation was not affected by Rac1 inhibition (Fig. 4e-f). PAK4 inhibition in HepG2 didn’t trigger a lower in lamellipodia formation however did considerably lower lamellipodia space (Fig. 4f). PAK4 additionally decreased HepG2 cell space (Fig. 4f). Taken collectively, general HepG2 cell spreading and subsequent lamellipodial formation was extra depending on PAK4 than on Rac1.
3D invasion dynamics of liver most cancers spheroids is distinct from 2D invasion profiles
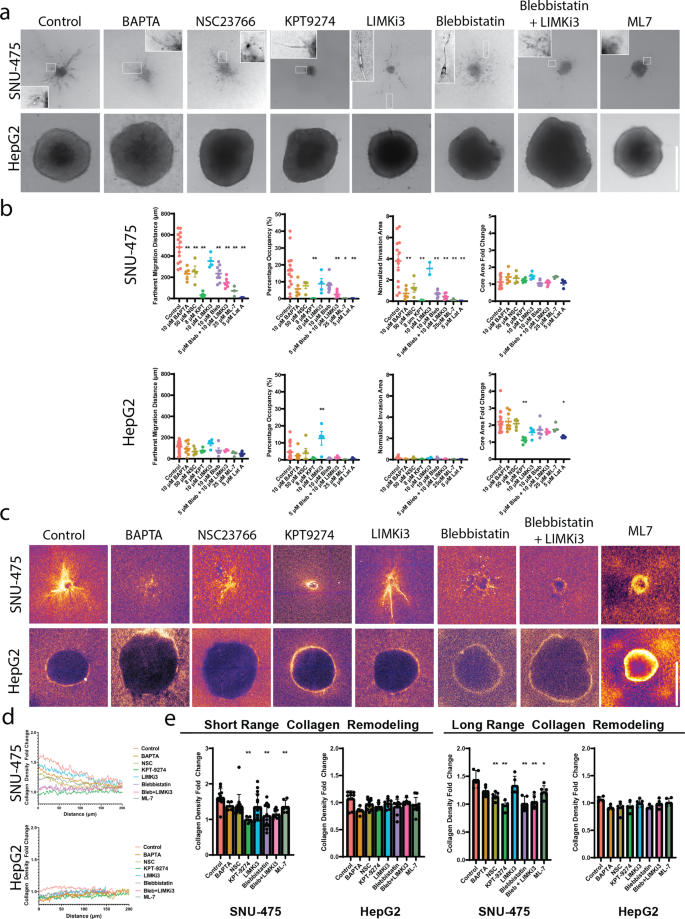
Given the placing results of cytoskeletal perturbation on 2D cell tradition molecularly and morphologically, we subsequent investigated how these perturbations would have an effect on 3D liver most cancers cell migration in collagen hydrogels. Earlier than embedding both SNU-475 or HepG2 spheroids in collagen gels, we pretreated spheroids for two h with every drug; through the course of the experiment, media with applicable drug focus was replenished each different day to make sure a drug response over your entire course of the experiment (Fig. 5a). All spheroids had been made with 1000 cells/ spheroid. Measures of the invasion space, farthest migration distance, proportion occupancy, and core space fold change had been measured after 5 days of development (Fig. 5b, Supplementary Fig. 10). We additionally measured adjustments in collagen densification because of drug therapy (Fig. 5d-e, Supplementary Fig. 11). For reference to well-studied remedies, we included SNU-475 spheroids handled with Sorafenib. Though excessive concentrations of Sorafenib (10 μM) had been capable of utterly inhibit invasion in 2D and 3D, intermediate concentrations of Sorafenib (3 μM) didn’t inhibit 2D or 3D invasion considerably (Supplementary Fig. 12). To find out if the long-term tradition of those spheroids with medication would have an effect on proliferation, we stained our spheroids with Ki67 after 5 days of tradition and didn’t see any vital adjustments in expression (Supplementary Fig. 13).
a Consultant photos of SNU-475 (high row) or HepG2 (backside row) spheroids 5 days after seeding in collagen gels. Arrows point out round morphology of invading SNU-475 cells. Scale bar, 500 μm. b We use fractional occupancy, normalized invasion space, and farthest migration distance to quantify the spheroid invasiveness, and calculate the core space fold change on day 5 normalized by that of day 0 to quantify the spheroid dimension development for SNU-475 and HepG2 spheroids. n > 4 spheroids for every situation from N = 2 unbiased experiments. c Consultant photos of reflectance microscopy to point collagen densification and alignment for NSC23766, KPT9274, Blebbistatin, and Blebbistatin + LIMKi3 therapy for each cell strains. Scale bar, 500 μm. d Common normalized collagen density profiles for SNU-475 SNU-475 (high row) or HepG2 (backside row) spheroids 5 days after seeding in collagen gels. Measurements are normalized to the final 30 knowledge factors of every plot profile. Plots present imply collagen density. e Quantification of short-range and long-range collagen reworking. The short-range collagen reworking metric is set because the collagen density fold change instantly exterior the periphery of the spheroid (30 μm). Lengthy-range collagen density is set because the collagen density fold change 80 μm away from the spheroid boundary. Plots present imply ± SD. n > 4 spheroids for every situation from N = 2 unbiased experiments. One-way ANOVA with Tukey post-hoc testing was carried out. Important distinction (p < 0.05) was detected between any two of the circumstances. *P < 0.05, **P < 0.01.
BAPTA therapy was used to check the results of calcium chelation on spheroid invasion. Though BAPTA therapy didn’t cut back 2D wound closure in SNU-475 cells, it was capable of lower SNU-475 spheroid invasion space and farthest migration distance (Fig. 5b). Moreover, BAPTA therapy inhibited collagen densification and alignment. BAPTA therapy didn’t have an effect on HepG2 spheroid invasion or collagen densification (Fig. 5c). Beforehand, BAPTA was reported to cut back collagen contraction39.
We subsequent examined the function of Rac1 inhibition on spheroid invasion. In settlement with our 2D knowledge, SNU-475 spheroids handled to inhibit Rac1 have considerably decreased cell migration distance (Fig. 5b). Strikingly, Rac1- inhibited SNU-475 cells which have migrated out of the spheroid are usually not spindle-shaped, however as a substitute undertake a round form (Fig. 5a, inset third column). As well as, they tended to have much less long-range collagen alignment and densification relative to regulate however had been nonetheless capable of generate short-range collagen alignment (Fig. 5d). Rac1 therapy neither impacts HepG2 spheroid invasion nor collagen densification (Fig. 5d). It was beforehand proven that Rac1 is ready to direct dynamic protrusion technology in addition to ECM alignment in metastatic breast most cancers cells17. Our outcomes recommend Rac1 performs an analogous function in HCC.
Because of the significance of PAK4 in 2D migration and morphology in each cell strains, we inhibited PAK4 with KPT9274 to check the function of PAK4 on spheroid invasion (Supplementary Fig. 14). PAK4 inhibition decreased the escape of SNU-475 cells out of the spheroid however didn’t have an effect on spheroid core development (Fig. 5b). Curiously, PAK4 inhibition decreased HepG2 spheroid core development however didn’t considerably have an effect on escape into the encircling matrix (Fig. 5b). When analyzing the encircling matrix, KPT9274 decreased short-range and long-range collagen densification by SNU-475 spheroids (Fig. 5c-e, Supplementary Fig. 11). This advised that PAK4 performs a job in SNU-475 matrix densification and alignment in addition to invasion equipment in 3D cell migration. As a result of HepG2 spheroids don’t usually trigger apparent collagen densification, PAK4 inhibition didn’t have an effect on collagen densification (Fig. 5d-e, Supplementary Fig. 15). As a result of collagen densification by HepG2 spheroids was seemingly attributable to enlargement in opposition to the encircling collagen, PAK4 seemingly additionally performs a job in regulating cell division and subsequent spheroid enlargement in 3D environments.
We subsequent sought to find out if PAK4’s major exercise is mediated by way of LIMK1 (by way of LIMKi3). Opposite to our 2D knowledge, LIMK1 inhibition didn’t considerably influence spheroid invasion nor qualitatively have an effect on collagen densification for both SNU-475 or HepG2 spheroids. LIMKi3 additionally didn’t have an effect on spheroid core dimension development (Fig. 5b). Though we hypothesized that LIMK1 inhibition would cease 3D migration for HepG2 spheroids as a result of it inhibited 2D cell migration, LIMK1 inhibition didn’t cease HepG2 cell invasion (Fig. 5a, inset fifth column) and resulted in larger proportion occupancy (Fig. 5b). We didn’t discover any adjustments in collagen densification for both cell line by LIMK1 inhibition (Fig. 5d-e, Supplementary Fig. 16–17). This consequence means that inhibition of LIMK1 alone just isn’t adequate to cut back 3D cell migration for SNU-475 spheroids, but for HepG2, LIMK1 inhibition is ready to enhance invasion.
We hypothesized that actomyosin contractility can be a key participant in figuring out SNU-475 3D invasion as a consequence of its dominant function in inhibiting 2D invasion. Therapy with blebbistatin brought on invading cells to have thinner protrusions (Fig. 5a, inset sixth column). As anticipated, blebbistatin therapy was capable of lower SNU-475 invasion space in addition to the furthest migration distance. Co-treatment with LIMKi3 additional exacerbated these results in addition to decreased proportion occupancy (Fig. 5b). Blebbistatin therapy and its co-treatment with LIMKi3 decreased brief and long-range collagen densification and alignment and brought on invading cells to undertake elongated shapes with thinner protrusions (Fig. 5a, inset on sixth and seventh columns, 5d). In distinction, HepG2 spheroids didn’t react considerably to blebbistatin therapy (Fig. 5b, Supplementary Fig. 18). These knowledge recommend that inhibition of actomyosin contractility was capable of impede SNU-475 cell migration, however doesn’t play a major function in HepG2 cell migration. Curiously, ML-7 therapy brought on SNU-475 invading cells to utterly lose their protrusions (Fig. 5a, eighth column). As well as, ML-7 therapy brought on a lower in invasion in addition to short-range and long-range collagen reworking (Fig. 5d-e). Conversely, HepG2 spheroids didn’t react considerably to ML-7 therapy. Total, these research confirmed that inhibiting the cells potential to generate motile and contractile forces results in abrogated 3D invasion for SNU-475 however not HepG2. Furthermore, these research confirmed how liver most cancers conduct and subsequently drug response in a 3D setting may be vastly totally different from their responses in conventional 2D techniques.
Liver most cancers stromal invasion is mediated by pathways concerned in mechanotransduction
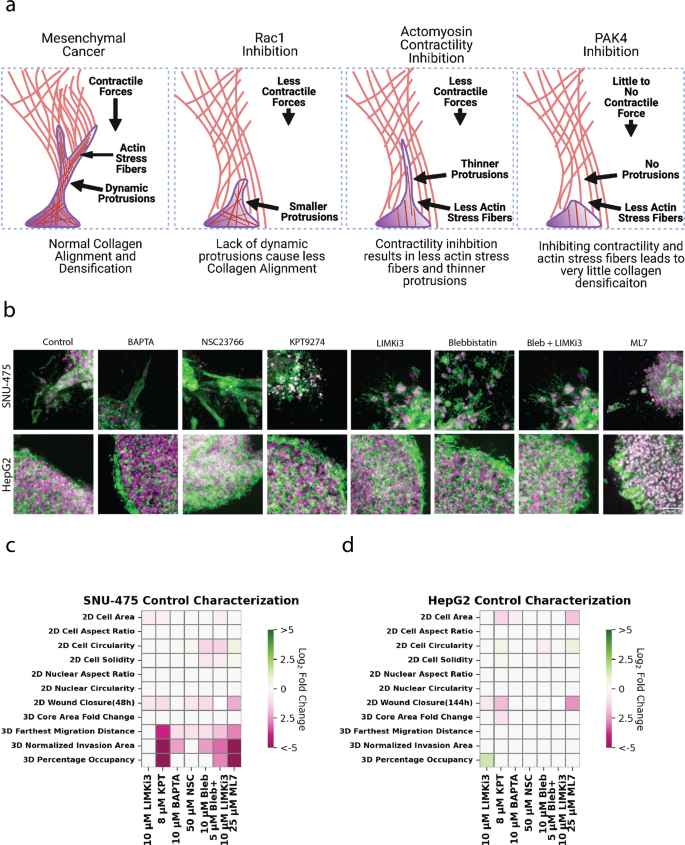
Our 2D and 3D research demonstrated the function of mechanotransduction in producing cell motile drive in addition to facilitating stromal invasion in liver most cancers. We offer a schematic that summarizes what every drug therapy will do to invasive liver most cancers cells (Fig. 6a). We discovered that inhibition of both dynamic protrusions or actomyosin contractility is adequate to inhibit collagen reworking in addition to subsequent liver most cancers invasion. Cross-sectional photos of spheroids confirmed distinct invasion profiles of SNU-475 cells based mostly on totally different cytoskeletal perturbations (Fig. 6b). Whereas Rac1 inhibition was capable of mitigate 3D invasion, invading cells had sturdy actin depth indicating the presence of tensional actin stress fibers. In distinction, cells handled with blebbistatin and blebbistatin + LIMKi3 had a lot thinner protrusions, which defined why these cells carried out much less collagen reworking. Within the excessive case, therapy with MLCK or PAK4 inhibitors brought on full lack of protrusions with invaded cells adopting a rounder phenotype. Our knowledge help this conclusion and have been summarized in heatmap format in SNU-475 (Fig. 6b, Supplementary Figs. 19–20) cells and HepG2 cells (Fig. 6c, Supplementary Figs. 21–22). A complete heatmap compares all circumstances to regulate for each cell strains (Supplementary Fig. 23). Our knowledge implicate PAK4, Rac1, and actomyosin contractility within the invasive properties of SNU-475 cells in 3D tradition. Though LIMK1 inhibition is ready to alter HepG2 2D invasion, it’s unable to vary HepG2 3D invasion. Total, our in vitro checks and subsequent quantification spotlight key pathways in HCC stromal invasion and establish molecular gamers appropriate for additional characterization.
a Schematic illustrating how altering cytoskeletal dynamics will have an effect on collagen densification and subsequent migration. b Consultant immunofluorescence photos of SNU-475 spheroids (first row) and HepG2 spheroids (second row) handled with BAPTA (Ca2+ chelator), NSC23766 (Rac1 inhibitor), KPT9274 (PAK4 inhibitor), LIMKi3 (LIMK inhibitor), blebbistatin (myosin inhibitor inhibitor), each blebbistatin and LIMKi3, or ML-7 (MLCK inhibitor), respectively. Inexperienced represents F-actin and magenta is DAPI. Scale bar: 50 μm. Heatmap illustrating 2D and 3D metrics characterised on this research for c SNU-475 and d HepG2 cells. Heatmap values are coloured if the distinction between two circumstances is taken into account considerably totally different (p < 0.05). White signifies no vital distinction between circumstances. Colour depth is set by log2 fold change of the group on the x-axis over management. Inexperienced signifies upregulation whereas pink signifies downregulation. One-way ANOVA with Tukey post-hoc testing was carried out to check for significance.
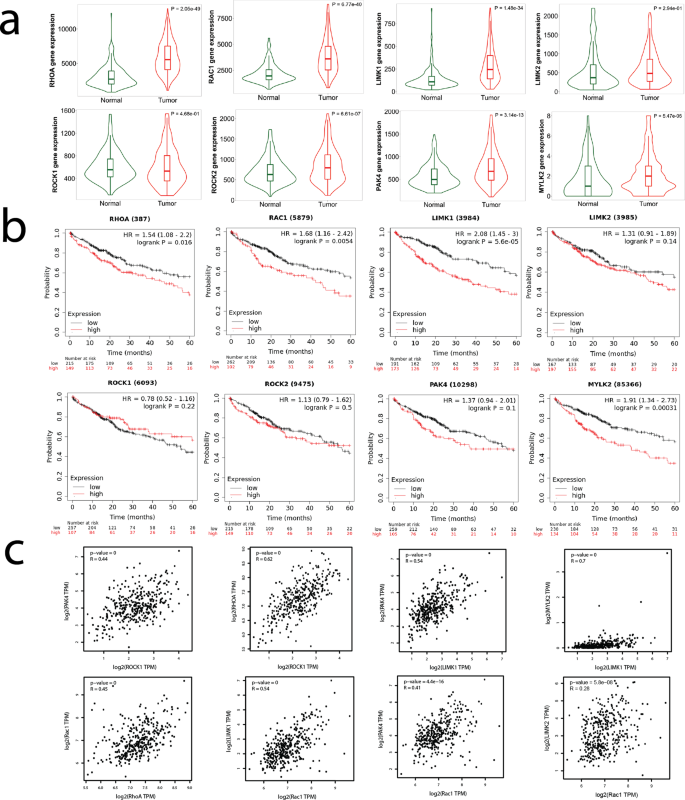
TCGA evaluation reveals Rho, Rac, LIM Kinase predict poor prognosis
We demonstrated the differential drug response between two distinctive liver most cancers cell strains in each 2D cultures and a 3D collagen-based mannequin. We then investigated whether or not the interrogated pathways play a job in affected person outcomes by analyzing liver most cancers cohorts from the TCGA database. Evaluating regular liver tissue samples (225 sufferers) and liver hepatocellular carcinoma (LIHC) samples (371 sufferers), the expression of the genes: RhoA, Rac1, LIMK1, LIMK2, ROCK1, ROCK2, MLCK2, and PAK4 is upregulated in liver most cancers (Fig. 7a). Excessive expression of those cytoskeletal regulators was correlated to a poorer survival charge amongst 364 sufferers (Fig. 7b). Our medical knowledge evaluation demonstrated a constructive correlation amongst RhoA, Rac1, LIMK1, and MLCK2 in LIHC sufferers (Fig. 7c, Supplementary Fig. 24). The TCGA evaluation confirmed that overexpression and subsequent dysregulation of this cytoskeletal equipment are related to liver most cancers. These datasets had been generated from bulk RNA sequencing and, subsequently, can’t differentiate between nor straight attribute these variations to tumor cells. Nevertheless, these knowledge do recommend that concentrating on these pathways within the liver most cancers microenvironment could also be a sound therapeutic technique.
a Gene expression ranges of RhoA, Rac1, LIMK1, LIMK2, ROCK1, ROCK2, PAK4, and MYLK2 are upregulated in HCC in comparison with regular liver tissue. b Kaplan–Meier evaluation (n = 364 sufferers) reveals that RhoA, Rac1, LIMK1, LIMK2, ROCK1, ROCK2, PAK4, and MYLK2 are linked to poor prognosis of HCC. c Correlation evaluation exhibits expressions of Rac1, RhoA, and LIMK1 are positively correlated, suggesting the signaling axes Rac1-LIMK and RhoA-LIMK could also be energetic in HCC. For every correlation plot, p < 10−16.