1 Introduction
Glioblastoma is essentially the most aggressive kind of mind tumor, with a median survival of 15 months. Its therapy efficacy stays low regardless of the mixture of full surgical resection, radiotherapy, and temozolomide (TMZ) chemotherapy, in addition to important advances in glioma prognosis, together with the event of deep learning-based approaches (Rui et al., 2023; Redlich et al., 2024). Problems of glioblastoma therapy embrace tumor infiltration into the mind tissue, excessive recurrence charges, and resistance to irradiation and TMZ (Rodríguez-Camacho et al., 2022). Subsequently, new medication are below growing to extend the susceptibility of glioblastoma to standard remedy (Ali et al., 2020; Tomar et al., 2021).
Glioblastoma displays intratumoral heterogeneity, which is a vital consideration for drug growth. The transcriptomic subtypes of glioblastoma, particularly, classical, proneural, and mesenchymal, can coexist inside a single tumor (Becker et al., 2021). Because the illness progresses, there’s a shift towards a mesenchymal phenotype, leading to elevated tumor invasive potential. This course of is much like the epithelial-mesenchymal transition (EMT) in carcinomas, nevertheless it differs in regulation and manifestation, and is due to this fact termed glial-mesenchymal transition (GMT) (Lai et al., 2024). Glioblastoma recurrence is related to a subset of glioblastoma stem cells (GSCs) that divide asymmetrically, thereby restoring the tumor cell inhabitants suppressed by remedy (Xie et al., 2022). As well as, each GMT and GSCs have been discovered to boost glioblastoma resistance to radiotherapy and TMZ therapy (Wang et al., 2021; Lai et al., 2024).
Latest critiques recommend that a number of compounds with anti-GMT and anti-GSC properties have been developed as adjuncts to glioblastoma remedy (Hersh et al., 2022; Lai et al., 2024). Though these compounds have exhibited promising antitumor exercise in vitro and in vivo, their affect on affected person survival in scientific trials has been restricted (Hersh et al., 2022; Lai et al., 2024). Subsequently, additional researches are required to totally notice the potential of such therapeutic brokers. One of many fundamental challenges on this area is crossing the blood-brain barrier, which limits drug supply as a consequence of tight junctions and multidrug efflux transporters (Sprowls et al., 2019). Attainable options embrace disrupting the blood-brain barrier or growing compounds able to each crossing into mind parenchyma and exhibiting anti-GMT and anti-GSC actions.
Our analysis group has beforehand developed soloxolone methyl (SM), a semisynthetic triterpenoid bearing a cyanoenone pharmacophore group that determines its excessive antitumor exercise (Logashenko et al., 2011) (Determine 1). It was proven that SM induced cervical carcinoma cell dying by way of endoplasmic reticulum stress, in all probability as a consequence of its direct interplay with SERCA2 and GRP94 (Markov et al., 2019; Alper et al., 2021). As well as, SM successfully inhibited the metastatic efficiency of lung adenocarcinoma and melanoma cells by suppressing EMT, hypothetically via its interactions with MMP-2/9 and JNK1, as decided by community pharmacology evaluation (Markov et al., 2020).
Determine 1. Chemical buildings of the soloxolone amides: N-(20-hydroxyethyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (1), N-(5′-hydroxypentyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (2), N-(2′-(dimethylamino)ethyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (3), N-(3′-(dimethylamino)propyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (4), N-(4′-bromophenyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (5), N-(pyridin-3-yl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (6), N-p-tolyl-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (7), N-(3′-(3″,5″-di-tert-butyl-4″-hydroxyphenyl)propyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30- amide (8), N-(2′-(1H-indol-2-yl)-ethyl)-2-cyano-3,12-dioxo-18βH-olean-9(11),1(2)-dien-30-amide (9). The cyanoenone pharmacophore group, inherited by soloxolone amides from their predecessor soloxolone methyl (SM), is highlighted in crimson. The blue coloration signifies the place the place the depicted amide moieties bind to the soloxolone scaffold.
To enhance the pharmacological properties of SM, together with poor blood-brain barrier permeability, a collection of its derivatives containing totally different amide moieties on the C-30 place have been developed (Determine 1). Soloxolone amides had been discovered not solely to cross the blood-brain barrier, but in addition to inhibit glioblastoma progress by inducing ROS-dependent apoptosis (Markov et al., 2022b). Our latest research present that soloxolone tryptamide (compound 9) inhibits the motility and spheroid progress of glioblastoma cells, lowering their invasive potential and stemness, respectively (Markov et al., 2022b; Markov et al., 2023). As well as, it acts synergistically with TMZ to scale back glioblastoma cell viability (Markov et al., 2023). Based mostly on these findings, it’s value exploring the soloxolone amide library to determine potential GMT and GSC inhibitors that might be used as a compound of mixture regimens to enhance outcomes of standard remedy.
Right here we report the GMT-modulating exercise of soloxolone amides in human and murine glioblastoma cells. The results of hit compound 7 on cell migration, adhesion, GMT marker expression, stemness, and TMZ cytotoxicity had been investigated utilizing three glioblastoma cell strains. Reactive oxygen species (ROS), TGF-β1 signaling, and aldehyde dehydrogenase (ALDH) exercise had been examined to achieve mechanistic perception into the exercise of compound 7. As well as, the efficacy of 7 as an anti-glioblastoma agent was verified in each mono- and TMZ-combined regimens in a mouse xenograft mannequin. Our outcomes assist the rationale for additional investigation of soloxolone amides as potential drug candidates that have an effect on glioblastoma invasion and stemness.
2 Supplies and strategies
2.1 Reagents and antibodies
Soloxolone amides had been synthesized as beforehand described (Markov et al., 2022b) and had been dissolved in DMSO at a focus of 10 mM and saved at −20 °C earlier than use. Human recombinant TGF-β1 (CYT-716) was bought from ProSpec-Tany TechnoGene Ltd. (Ness-Ziona, Israel). The rabbit anti-Smad2/Smad3 antibody (ab202445) and the goat anti-rabbit IgG antibody conjugated with Alexa Fluor 488 (ab150077) had been obtained from Abcam (Cambridge, MA, USA). Diamidino-2-phenylindole (DAPI) was bought from Thermo Fisher Scientific (Rockford, IL, United States).
2.2 Cell cultures
Human glioblastoma U87 and U118 and mouse glioblastoma EPNT-5 cell strains had been obtained from the Russian Tradition Assortment (Institute of Cytology of the Russian Academy of Sciences (RAS), St. Petersburg, Russia). Human non-transformed hFF3 foreskin fibroblasts had been kindly supplied by Dr. Olga A. Koval (Institute of Chemical Biology and Basic Drugs of the Siberian Department of RAS, Novosibirsk, Russia). All cells had been maintained in DMEM (U87, U118, EPNT-5) or IMDM (hFF3) medium supplemented with 10% fetal bovine serum (FBS; Dia-M, Moscow, Russia) and 1% antibiotic-antimycotic answer (100 U/mL penicillin, 100 μg/mL streptomycin, 0.25 μg/mL amphotericin B; Central Drug Home Pvt. Ltd., New Delhi, India) at 37 °C and 5% CO2 in a humidified environment.
2.3 Mice
Six-to-eight-week-old feminine athymic nude mice with common weight of twenty-two–24 g had been bought from the Middle for Genetic Sources of Laboratory Animals on the Institute of Cytology and Genetics of the Siberian Department of the Russian Academy of Sciences (SB RAS) (Novosibirsk, Russia). Animals had been saved in plastic cages (5-6 animals per cage) below a traditional daylight schedule in temperature-controlled, particular pathogen-free circumstances. Water and meals had been supplied advert libitum. All animal procedures had been carried out in strict accordance with the European Communities Council Directive 86/609/CEE. The experimental protocol was permitted by the Committee on the Ethics of Animal Experiments on the Institute of Cytology and Genetics SB RAS (protocol no. 56 of 10 August 2019). Subcutaneous and intraperitoneal injections had been carried out utilizing a needle with a diameter of not more than 0.25 mm. The hectic impact lasted now not than the time interval required for subcutaneous or intraperitoneal injection. Mice had been euthanized on the finish of the experiment below isoflurane anesthesia utilizing a fuel combination containing 3% isoflurane +97% air at a circulate price of two L/min.
2.4 Cell viability assay
Cells had been seeded in quadruplicate in 96-well plates at 10,000 cells/properly and allowed to stick in a single day. The medium was then changed with serum-free DMEM containing rising concentrations of soloxolone amides for 48 h. After incubation, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT dye) was added at a ultimate focus of 0.5 mg/mL and left for two h. The ensuing formazan crystals had been dissolved in DMSO and the optical density was measured at 570 nm utilizing a Multiscan RC plate reader (Thermo LabSystems, Helsinki, Finland).
For synergy evaluation, cell viability was assessed after 72-h incubation with every dose mixture of compound 7 (0–400 nM) and TMZ (0–500 μM) utilizing the MTT assay. The drug synergy rating was calculated for the dose-response matrix in keeping with the best single-agent (HSA) mannequin utilizing the SynergyFinder + internet utility (https://www.synergyfinder.org/).
2.5 Cell adhesion assay
Adhesiveness of glioblastoma cells was assessed by two impartial strategies. First, cell adhesion to tradition plastic was examined utilizing a trypsin therapy assay, as beforehand described (Sava et al., 2004; Mazuryk et al., 2015; van Dijk et al., 2017). Briefly, U87 and U118 cells (10,000 cells per properly of a 96-well plate, n = 4) had been cultured with TGF-β1 (50 ng/mL) and non-toxic concentrations of soloxolone amides for 48 h after which handled with a 1:20 dilution of TrypLE Categorical (Gibco, USA) in PBS for 3 min at 37°C. Non-adherent cells had been then eliminated by a triple PBS wash, and the remaining adherent cells had been mounted with 4% formaldehyde and stained with crystal violet dye (0.1% w/v). Photographs had been captured utilizing the iBright 1,500 Imaging System (Invitrogen, USA), and the cell-occupied space was calculated utilizing ImageJ. Alternatively, the variety of adherent cells was quantified utilizing the MTT assay.
Second, to evaluate cell adhesion to the extracellular matrix (ECM), an ECM adhesion assay was carried out in keeping with beforehand printed methodology (Guan et al., 2015; Hu et al., 2019). Briefly, U87 cells had been pretreated with TGF-β1 (50 ng/mL) and compound 7 (50 nM) for 48 h, after which seeded in 96-well plates coated with Matrigel (BD Biosciences, Bedford, MA, USA) at a focus of 30,000 cells for 1 h (n = 3). After that, the wells had been washed to take away non-adherent cells, adherent cells had been mounted with formaldehyde, stained with crystal violet dye, and analyzed as described above.
2.6 Evaluation of cell morphology
After seeding at a focus of 5,000 cells/properly in triplicate in 96-well plates and incubating in a single day, U87 and U118 cells had been uncovered to TGF-β1 (50 ng/mL) and non-toxic concentrations of soloxolone amides for 48 h. An EVOS XL Core microscope with an built-in CMOS digital camera (Thermo Fisher Scientific, USA) was then used to look at cell morphology at ×20 magnification. Side ratio (AR) and space had been quantified for at the very least 150 cells in every experimental group utilizing ImageJ software program (NIH, USA).
2.7 Evaluation of cell migration
To carry out transwell migration, U87 cells had been pretreated with TGF-β1 (50 ng/mL) and compound 7 (50 nM) for 48 h. Subsequently, 250,000 cells had been positioned within the higher chamber of the CIM-16 xCELLigence plate in serum-free DMEM supplemented with compound 7 and TGF-β1 (n = 4), whereas the decrease chamber was stuffed with DMEM containing 10% FBS. Cell migration was monitored for 72 h utilizing the xCELLigence RTCA DP system (ACEA Biosciences Inc., San Diego, CA, USA). The electrodes positioned below the porous membrane detected modifications in resistance, which had been then transformed into cell indices proportional to the variety of migrating cells.
For the scratch assay, U118 cells had been seeded in triplicate in 24-well plates at a focus of 300,000 cells/properly and allowed to stick in a single day. The cells had been then scratched with a ten μL plastic pipette tip, washed with PBS, and lined with serum-free DMEM containing TGF-β1 (50 ng/mL) and compound 7 (50 nM). Scratches had been photographed at 0, 24, and 48 h utilizing an EVOS XL Core microscope. Wound closure was decided by estimating the scratch space at every time level utilizing ImageJ software program and normalizing to the scratch space at 0 h.
2.8 Quantitative real-time polymerase chain response (qRT-PCR)
Whole RNA was extracted utilizing TRIzol Reagent (Ambion, Austin, TX, USA) in keeping with the producer’s protocol. Subsequently, the primary strand cDNA was synthesized from 4 μg of complete RNA utilizing an oligo (dT)18 primers and M-MuLV-RH revertase (Biolabmix, Novosibirsk, Russia). Lastly, RT-qPCR was carried out utilizing particular primers (Desk 1) and BioMaster SYBR Blue reagent equipment (Biolabmix, Novosibirsk, Russia). Relative expression was decided by normalization to the housekeeping gene HPRT utilizing the two−ΔΔCT methodology (Livak and Schmittgen 2001).
2.9 Colony formation assay
U87, U118 and EPNT-5 cells had been seeded at a density of 200 cells/properly in 96-well plates and handled with rising concentrations of compound 7 (0–1,000 nM) dissolved in 10% FBS-containing DMEM (n = 5). On day 10, colonies had been mounted with 4% formaldehyde and stained with crystal violet dye (0.1% w/v). Photographs had been captured utilizing the iBright 1,500 Imaging System (Invitrogen, USA) and the world of colonies with a minimal diameter of 100 µm was calculated utilizing ImageJ. Within the case of overlapping colonies, their complete space was calculated. Lastly, the world of the fashioned cell colonies was summed and normalized to the whole colony space within the management.
2.10 Tumorsphere formation assay
For analysis of spheroid progress, the underside of the 96-well plate was lined with 1% agarose for non-adherent circumstances. U87 cells (5,000 cells/properly) had been then seeded and allowed to kind spheroids for twenty-four h, after which rising concentrations of compound 7 (0–1,000 nM) had been added (n = 8). Each different day of the 10-day experiment, recent medium supplemented with compound 7 was added to half of the medium, and the spheroids had been photographed utilizing a camera-equipped ZEISS Primo Vert microscope (Carl Zeiss Microscopy GmbH, Jena, Germany). To evaluate the formation of secondary tumorspheres, cells had been dissociated from major tumorspheres after 4 days of compound 7 stimulation utilizing TrypLE Categorical and had been then seeded into agarose-coated 96-well plates (n = 8). On day 3, the ensuing secondary spheroids had been examined microscopically. Main and secondary spheroid areas had been quantified utilizing ImageJ.
2.11 Stream cytometry
To evaluate ALDH exercise, U87 cells had been dissociated from spheroids utilizing TrypLE Categorical after 4 days of induction with compound 7 (0–100 nM) (n = 3). Cells had been then stained with the AldeRed ALDH Detection Assay (SCR150; EMD Millipore, Burlington, MA, USA) in keeping with the producer’s protocol. Experimental controls included U87 cells grown in 2D tradition and cells handled with the ALDH inhibitor diethylaminobenzaldehyde (DEAB). Gates of ALDH-positive cells had been set in keeping with the inhibitory impact of DEAB on management cells.
To watch apoptosis and necrosis, U87 and U118 cells had been stained with Annexin V-FITC and propidium iodide (ab14085; Abcam, Inc., Cambridge, MA, USA) after 48 h of incubation with compound 7 (0–2000 nM) in keeping with the producer’s protocol (n = 3). To quantify the whole proportion of apoptotic cells in management and experimental teams, the occasions accrued within the decrease proper quadrant (early apoptosis) and higher proper quadrant (late apoptosis) had been summarized.
Intracellular ROS ranges had been measured in keeping with beforehand printed research (Han et al., 2008; Samandari-Bahraseman et al., 2023; Kopsida et al., 2024), utilizing the DCFDA/H2DCFDA Mobile ROS Assay Package (Abcam, Cambridge, United Kingdom). U87 cells had been incubated with 7 and TMZ with or with out NAC for 72 h (n = 3). Cells had been then washed with PBS and incubated with 20 µM H2DCFDA for 30 min below commonplace circumstances, adopted by the removing of H2DCFDA-containing medium and double washing with PBS.
After staining, 10,000 cells per pattern had been analyzed on a NovoCyte circulate cytometer utilizing NovoExpress software program (ACEA Biosciences, Inc., USA).
2.12 Immunofluorescence
U87 cells (150,000 cells/properly) had been allowed to connect to glass coverslips in 24-well plates previous to 48-h incubation with compound 7 (50 nM) below serum-free circumstances, adopted by stimulation with TGF-β1 (50 ng/mL) for 1 h. After fixation in 4% formaldehyde for 20 min, cells had been incubated with major antibodies towards Smad2/3 (1:100, ab202445) for 1 h below permeabilizing circumstances (0.1% Triton X100, 5 mg/mL BSA, 37°C) and Alexa Fluor 488-conjugated secondary antibodies (1:500, ab150077) for 1 h at nighttime. Lastly, nuclei had been stained with DAPI (1 μg/mL) for 10 min and cells had been mounted on glass slides with Fluoromount-G (Thermo Fisher Scientific, Rockford, IL, USA). Photographs had been captured utilizing a LSM710 confocal microscope (Zeiss, Oberkochen, Germany) at ×40 magnification. Nuclear translocation was quantified by measuring the ratio of nuclear to perinuclear depth of Alexa Fluor 488 in CellProfiler software program (Broad Institute, Cambridge, MA, USA) for 50 cells per experimental group (n = 3).
2.13 Molecular docking
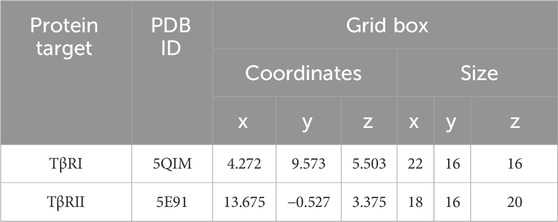
The crystal buildings of TGF-β receptors kind I and II (TβRI and TβRII) had been obtained from the Protein Information Financial institution (PDB IDs 5QIM and 5E91, respectively). Protein preparation included removing of co-crystallized ligands and solvent molecules, addition of polar hydrogen atoms, and calculation of Gasteiger prices in AutoDockTools v.1.5.7. The construction of compound 7 was created utilizing MarvinSketch v.22.1, optimized with the MMFF94 pressure area in Avogadro v.1.2.0, and let out rotation utilizing AutoDockTools. Lastly, molecular docking was carried out utilizing AutoDock Vina v.1.2.5 with the parameters described in Desk 2. The ensuing docking fashions of compound 7 had been analyzed utilizing UCSF ChimeraX v.1.7.1. The inhibitors initially co-crystallized with TβRI and TβRII (PubChem CIDs 121411739 and 118988613, respectively) underwent the identical processing as compound 7 and served as optimistic controls.
2.14 Tumor transplantation and design of animal experiment
A xenograft mannequin of tumor development was induced by subcutaneous (s.c.) implantation of U87 glioblastoma cells (2×107 cells/mL) suspended in 0.1 mL of saline buffer into the left flank of athymic nude mice. On day 4 after tumor transplantation, the mice had been divided into 4 teams: (1) mice obtained intraperitoneal (i.p.) injections of 10% Tween-80 (car) (n = 6); (2) mice obtained i.p. injections of compound 7 in 10% Tween-80 at a dose of 20 mg/kg (n = 5); (3) mice obtained i.p. injections of TMZ in saline buffer at a dose of 10 mg/kg (n = 5); (4) mice obtained mixture remedy of compound 7 and TMZ on the similar dosage and administration route (n = 6). Compound 7 was administered thrice per week; in complete seven injections had been carried out. TMZ was administered each day besides weekends; in complete eleven injections had been carried out. In the course of the experiment, the tumor volumes had been decided thrice per week utilizing caliper measurements and had been calculated as V = (D × d2)/2, the place D is the longest diameter of the tumor node and d is the shortest diameter of the tumor node perpendicular to D. Mice had been sacrificed on day 19 after tumor transplantation and materials (tumor nodes, livers, and kidneys) was collected for subsequent evaluation.
2.15 Toxicity evaluation
In the course of the experiment, the overall standing and physique weight of the animals had been monitored. On the finish of the experiment, livers and kidneys had been collected and organ indices had been calculated as (organ weight/physique weight) × 100%, adopted by the normalization of organ indices of experimental mice to organ indices of management mice.
2.16 Histology and immunohistochemistry
For the histological research, the tumor specimens had been mounted in 10% neutral-buffered formalin (BioVitrum, Moscow, Russia), dehydrated in ascending ethanols and xylols and embedded in HISTOMIX paraffin (BioVitrum, Russia). The paraffin sections (3–4 µm) had been sliced on a Microm HM 355S microtome (Thermo Fisher Scientific, Waltham, MA, USA) and stained with hematoxylin and eosin.
For the immunohistochemical research, the tumor sections had been deparaffinized and rehydrated. Antigen retrieval was carried out after publicity in a microwave oven at 700 W. The samples had been incubated with anti-Ki-67 (ab16667, Abcam, Boston, MA, USA) (n = 3), anti-GFAP (ab194325, Abcam, USA) (n = 3), and anti-N-cadherin (ab76011, Abcam, USA) (n = 3) major antibodies in keeping with the producer’s protocol. Then, the sections had been incubated with secondary horseradish peroxidase (HPR)-conjugated antibodies, uncovered to the three,30-diaminobenzidine (DAB) substrate (Rabbit Particular HRP/DAB (ABC) Detection IHC Package, ab64261, Abcam, USA) and stained with Mayer’s hematoxylin.
Morphometric evaluation of tumor sections included analysis of the numerical density (Nv) of mitoses (n = 5–6) and Ki-67-positive cells (n = 3) indicating the variety of particles studied within the sq. unit, 3.2 × 106 μm2 on this case. 5 to 10 random fields had been examined from the tumor specimens of three mice in every group, for a complete of 15–30 check fields.
All the pictures had been examined and scanned utilizing an Axiostar Plus microscope outfitted with an Axiocam MRc5 digital digital camera (ZEISS, Germany) at magnification of ×200 and ×400.
2.17 The affiliation of CDH2 and GFAP expression with pathogenesis of glioma/glioblastoma in sufferers
The evaluation of the affiliation of CDH2 and GFAP expression ranges with the survival charges of sufferers with recurrent glioma and mesenchymal-versus proneural-type of glioblastoma was carried out based mostly on the Chinese language Glioma Genome Atlas (CCGA) and the Repository for Molecular BRAin Neoplasia DaTa (REMBRANDT), respectively, utilizing GlioVis device (Bowman et al., 2017).
2.18 Statistical evaluation
Statistical evaluation was carried out utilizing R Studio (2023.09.1 + 494) for R model 4.3.2 (in vitro experiments) and GraphPad Prism model 8.0.1 (GraphPad Software program, San Diego, CA, USA) (in vivo experiment). Normality of the datasets was assessed utilizing the Shapiro-Wilk criterion, and the suitable statistical checks had been carried out utilizing the “rstatix” package deal. Information from cell adhesion, cell morphology, transwell migration and immunofluorescence experiments had been analyzed by Kruskal–Wallis check adopted by Dunn’s post-hoc comparisons. Information from scratch assay, colony formation, tumorsphere formation and RT-qPCR experiments had been analyzed by one-way ANOVA adopted by Tukey’s post-hoc comparisons. Information from in vivo experiment had been analyzed by unpaired Pupil’s t-test.
3 Outcomes
3.1 Screening for inhibitory exercise of soloxolone amides towards glial-mesenchymal transition (GMT) and identification of a success compound
To determine essentially the most promising soloxolone amide drug candidates, we screened their impact on glial-mesenchymal transition (GMT), a course of related to elevated invasiveness of glioblastoma cells (Lai et al., 2024). First, the MTT assay was carried out to find out low poisonous concentrations of the compounds towards U87 glioblastoma cells (Determine 2A). The soloxolone amides had been categorized into three teams based mostly on their relative cytotoxicity after 48 h of incubation, particularly, group 1 (1, 3, and 4), group 2 (2, 6, and 9), and group 3 (5, 7, and 8) exhibited low, average, and excessive cytotoxicity, respectively. Based mostly on the obtained cytotoxic profiles, non-toxic concentrations of compounds that decreased U87 cell viability by lower than 10%, which had been 300 nM, 100 nM, and 50 nM for compounds from group 1, 2, and three, respectively, had been chosen for additional screening experiments (Determine 2A). Apparently, essentially the most energetic soloxolone amides include a benzene ring with lipophilic substituents, particularly, methyl- (5), bromo- (7) and tert-butyl (8) teams, whereas compounds with average cytotoxicity bear moieties able to forming H-bonds (2, 6, 9), being both H-donors because of the acidity of the O-H (2) and N-H (9) bonds or H-acceptor because of the basicity of the nitrogen atom of the pyridine ring (6). The least cytotoxic had been compounds 1, 3 and 4, which have substituents that may additionally act as H-acceptors as a consequence of lone pairs of heteroatoms, however their skill to kind H-bonds is far much less pronounced in comparison with compound 6 of group 2.
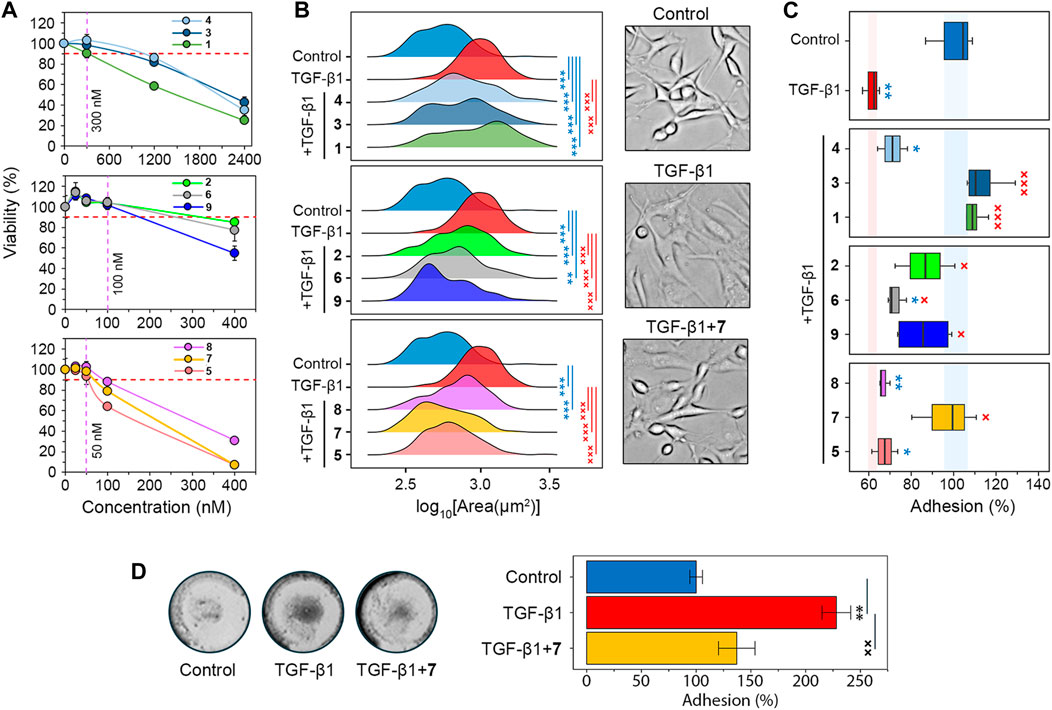
Determine 2. Screening of the soloxolone amide library for GMT inhibitory exercise. (A) The cytotoxicity of soloxolone amides towards U87 cells assessed by MTT assay at 48 h (n = 4). The purple dotted line reveals non-toxic concentrations inflicting lower than 10% cell dying (crimson dotted line). (B) Measurement distribution of U87 cells after therapy with TGF-β1 (50 ng/mL) and soloxolone amides (non-toxic concentrations) for 48 h (n = 3). (C) Adhesion of U87 cells to tradition plastic after therapy with TGF-β1 (50 ng/mL) and soloxolone amides (non-toxic concentrations) for 48 h (n = 4). (D) Adhesion of U87 cells to Matrigel after therapy with TGF-β1 (50 ng/mL) and compound 7 (50 nM) for 48 h (n = 3). Information in line graph are represented as imply ± commonplace deviation (SD). Statistical significance was calculated by comparability with the management (marked by *) or TGF-β1-treated group (marked by ×). */×, **/××, ***/××× point out that p-values had been lower than 0.05, 0.01, and 0.001, respectively.
Morphological modifications, the weakening of cell-cell adhesion, and strengthening of cell-ECM adhesion are attribute options of GMT in glioblastoma cells (Joseph et al., 2014; Iser et al., 2016; Jin et al., 2020; Zhitnyak et al., 2020). Microscopic examination confirmed that TGF-β1-induced U87 cells had elongated protrusions typical for mesenchymal cells (Joseph et al., 2014), which elevated their measurement by 76% in comparison with management cells. All soloxolone amides shifted the distribution of cell measurement in the direction of smaller values, and three compounds, 5, 7, and 9, restored their morphological traits to the management cells (Determine 2B).
To guage adhesion of U87 cells to tradition plastic, cells had been handled with TrypLE (a recombinant analog of trypsin) and the variety of cells remaining connected to the tradition plates after washing was quantified utilizing crystal violet assay (Supplementary Determine S1A). In line with earlier reviews (Liu et al., 2015; Kang et al., 2019; Xu et al., 2022), incubation with the GMT-inducing cytokine TGF-β1 for 48 h decreased U87 adhesion by 38.5% (Determine 2C). The vast majority of examined soloxolone amides, apart from 4, 5 and 8, markedly elevated the resistance of TGF-β1-treated U87 cells to TrypLE-mediated cell dislodgement, with 1, 3 and 7 restoring it to regulate ranges (Determine 2C). To double-check the outcomes obtained, the adhesion assay was repeated utilizing the MTT assay to evaluate the quantity of adherent cells by spectroscopy. The outcomes obtained (Supplementary Determine S1B) had been much like these proven in Determine 2B, with 7 demonstrating one of the crucial pronounced pro-adhesive results.
To additional characterize the power of 7 to modulate glioblastoma cell adhesion, its impact on U87 cell adhesion to Matrigel, which mimics the ECM, was examined. Surprisingly, in distinction to earlier assays, TGF-β1 was discovered to considerably enhance cell adhesion to Matrigel, whereas 7 successfully suppressed this induction to virtually management ranges (Determine 2D). We hypothesize that the alternative impact of compound 7 on the adhesion of U87 cells to tradition plastic (Determine 2C) and Matrigel (Determine 2D) could also be decided by variations in the important thing kinds of adhesion contacts within the adhesion assays used. Within the case of the Matrigel adhesion assay, the principle contribution to cell adhesion is made by cell-matrix contacts, whereas within the case of TrypLE-mediated cell detachment, a big function in cell adhesion is performed by cell-cell interactions, the energy of which decreases at GMT (Zhitnyak et al., 2020). In any case, our outcomes clearly point out the power of compound 7 to dam the impact of TGF-β1 on the adhesiveness of glioblastoma cells. Based mostly on these findings, soloxolone para-methylanilide (compound 7) was revealed as a success compound that inhibited each morphological modifications and tumor cell adhesiveness throughout GMT.
3.2 Compound 7 inhibited GMT in TGF-β1-induced glioblastoma cells
The following section of the research aimed to research the affect of compound 7 on glioblastoma aggressiveness, particularly mesenchymal and stem cell traits. To make sure the reliability of the outcomes obtained in U87 cells (Figures 2B, C), experiments had been additionally carried out in human glioblastoma U118 and mouse glioblastoma EPNT-5 cells. The MTT assay confirmed comparable ranges of cytotoxicity of 7 in U118 and EPNT-5 cells, which had been decrease than that in U87 cells (IC50U118 = 309.9 nM, IC50EPNT-5 = 257.4 nM, IC50U87 = 160.6 nM) (Figures 2A, 3A). Observe that the cytotoxicity of 7 towards non-malignant human hFF3 fibroblasts was considerably decrease than the above values: 7 at 400 nM resulted within the dying of solely about 21% of the cells (Determine 3A).
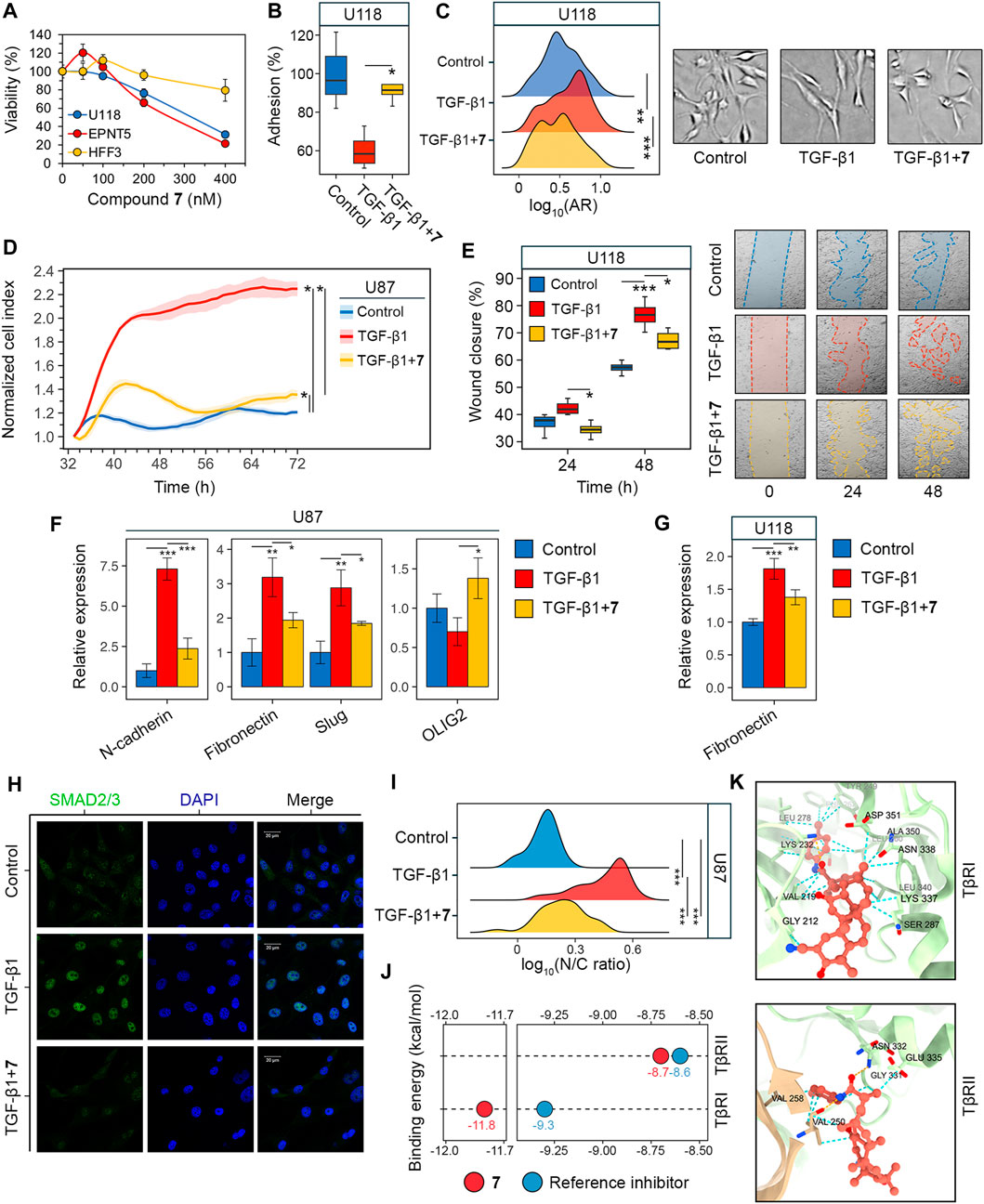
Determine 3. Analysis of GMT inhibition by compound 7. (A) Cytotoxicity of 7 towards U118, EPNT-5 and hFF3 cells assessed by MTT assay at 48 h (n = 4). (B–G) The impact of 7 on the phenotypical manifestations of GMT was assessed by incubating U87 and U118 cells with TGF-β1 (50 ng/mL) and 7 (50 nM) for 48 h. (B) Adhesion of U118 cells to tradition plastic measured by MTT assay (n = 4). (C) Form distribution of U118 cells (n = 3). (D) Transwell migration of U87 cells (n = 4). (E) Wound closure on a monolayer of U118 cells (n = 3). (F, G) Expression of GMT markers in U87 (F) and U118 (G) cells assessed by RT-qPCR (n = 3). (H) Immunofluorescence staining of SMAD2/3 in U87 cells after pre-incubation with 7 for 48 h and activation with TGF-β1 for 1 h (I) SMAD2/3 distribution in U87 cells expressed because the ratio of nuclear (N) to cytoplasmic (C) depth (n = 3, 100 cells/pattern). (J) Binding energies of 7 to the kinase domains of TGF-β receptors kind I and II (TβRI and TβRII) estimated utilizing AutoDock Vina. For comparability, the affinities of recognized receptor inhibitors are given (PubChem CIDs 121411739 and 118988613 for TβRI and TβRII, respectively). (Okay) Docking construction of 7 with TβRI and TβRII visualized in ChimeraX. The orange and cyan dotted strains point out hydrogen bonds and van der Waals interactions, respectively. Information in bar graphs are represented as imply ± commonplace deviation (SD). Statistical significance was calculated by comparability with the management or TGF-β1-treated group. *, **, *** point out that p-values had been lower than 0.05, 0.01, and 0.001, respectively.
Much like U87 cells, U118 cells had been additionally delicate to 7: it was discovered that 7 at a non-toxic focus of fifty nM successfully reversed TGF-β1-induced 40% lower in U118 cell adhesion to tradition plastic (Determine 3B). Apparently, in distinction to U87 cells, TGF-β1 didn’t alter the dimensions of U118 cells (Determine 2C), however brought on them to accumulate a extra elongated spindle-like form, as evidenced by a 20% enhance within the ratio of the foremost to minor axes (facet ratio, AR) of TGF-β1-treated cells in comparison with management (Determine 3C). 7 restored the form of TGF-β1-induced cells to that of the management cells (Determine 3C).
To substantiate the inhibition of GMT by 7, we examined modifications in cell motility and expression of GMT-related genes in glioblastoma cells (Zhang et al., 2017; Ouanouki et al., 2018). To evaluate the impact of 7 on GMT-associated motility, the transwell migration of U87 cells was monitored in an actual time routine utilizing the xCELLigence analyzer. Determine 3D reveals that the migration of U87 cells elevated considerably in response to TGF-β1 as much as 66 h of incubation, reaching 186% of that of management cells. The addition of 7 resulted in a drastic lower within the motility of TGF-β1-treated U87 cells near the extent within the management (Determine 3D). Regardless of a distinction from the management at 72 h, compound 7 suppressed transwell migration of TGF-β1-treated glioblastoma cells by 1.7-fold in comparison with cells stimulated with TGF-β1 alone (Determine 3D). The impact of 7 on TGF-β1-induced motility of glioblastoma cells was additional independently verified utilizing the scratch assay. As proven in Determine 3E, TGF-β1 considerably enhanced the motility of U118 cells, whereas the examined compound utterly reversed or decreased the impact of TGF-β1 by 34% at 24 and 48 h, respectively. Analysis of the degrees of key GMT-associated markers in glioblastoma cells confirmed that TGF-β1 elevated the expression of mesenchymal genes encoding N-cadherin, fibronectin, and Slug in U87 cells by 7.3-, 3.2-, and a couple of.9-fold, respectively, in comparison with untreated management, whereas the addition of 7 decreased these results by 67.6%, 39.2%, and 35.8%, respectively, in comparison with TGF-β1-stimulated cells (Determine 3F). An identical inhibitory impact of 7 on fibronectin expression was noticed in U118 cells (Determine 3G). As well as, qRT-PCR evaluation revealed a low sensitivity of the proneural gene OLIG2 to TGF-β1 and its 2-fold upregulation in TGF-β1-induced U87 cells handled with 7 (Determine 3F), which can additionally point out the anti-GMT exercise of 7.
3.3 Compound 7 inhibited TGF-β1/SMAD2/3 signaling axis in all probability as a consequence of direct interactions with TβRI/II
Within the subsequent step of the research, the potential molecular mechanism behind the anti-GMT impact of 7 was investigated. The TGF-β1 signaling includes interplay of TGF-β1 with TGF-β receptor kind II (TβRII) adopted by the activation of TGF-β receptor kind I (TβRI) and phosphorylation of SMAD2 and SMAD3. The latter proteins, upon binding to SMAD4, translocate to the nucleus and act as transcription elements to activate GMT-associated genes (Katsuno and Derynck, 2021). The impact of compound 7 on SMAD2/3 translocation was examined in U87 cells by oblique immunofluorescence microscopy. As proven in Determine 3H, SMAD2/3 had been dispersed within the cytoplasm of untreated U87 cells, exhibiting dim fluorescence. Stimulation with TGF-β1 for 1 h resulted within the nuclear translocation of SMAD2/3 (Determine 3H), as evidenced by a 3.3-fold enhance within the nuclear-cytoplasmic ratio of their fluorescence depth (Determine 3I). Pretreatment with 7 for 48 h previous to TGF-β1 induction almost restored the dispersed sample of SMAD2/3 localization in U87 cells (Determine 3H). Though the nuclear-cytoplasmic ratio of SMAD2/3 was decreased by 52% below hit compound therapy, it remained considerably totally different from the management group (Determine 3I). This discovering is per a statistically important, however not full, inhibition of downstream targets of the TGF-β1 signaling pathway, particularly, N-cadherin, fibronectin, and Slug, in TGF-β1-activated U87 cells handled with 7 (Determine 3F). Compound 7 was additional assumed to inhibit SMAD2/3 signaling by blocking TGF-β receptors. Molecular docking simulations revealed that 7 can bind to the kinase domains of TβRI and TβRII with equal or decrease binding energies (∆G) than beforehand reported inhibitors of those receptors (Determine 3J). Compound 7 penetrated deeply into the hydrophobic pocket of the TβRI kinase area, whereas remaining near the doorway of the TβRII kinase area (Determine 3K). In consequence, it exhibited the next affinity for TβRI than TβRII (∆GTβRI = −11.8 kcal/mol vs. ∆GTβRII = −8.7 kcal/mol), which was supported by better stabilization with van der Waals forces (24 vs. 8 interactions in TβRI and TβRII, respectively) (Determine 3K). As well as, 7 fashioned one hydrogen bond with TβRI (Lys232; performs a important function in TβRI kinase exercise (Chaikuad and Bullock, 2016) and two hydrogen bonds with TβRII (Val250, Asn332; recognized amino acid residues positioning TβRII inhibitors (Verdura et al., 2022; Ye et al., 2023) (Determine 3K). Taken collectively, these information recommend that compound 7 inhibits GMT in glioblastoma cells via the TGF-β1/SMAD2/3 pathway.
3.4 Compound 7 decreased stemness of glioblastoma cells
GSCs are one other hallmark of glioblastoma aggressiveness as a consequence of their excessive capability for self-renewal and tumor initiation (Biserova et al., 2021). Colony formation assays had been used as first step to evaluate the impact of 7 on the stemness of glioblastoma cells. The outcomes present that rising the focus of 7 from 50 to 1,000 nM resulted in a gradual lower in colony space from 51.3% to three.9% in U87 cells after 10 days of incubation (Determine 4A). An identical pattern was noticed in U118 cells, though they confirmed a weaker response to decrease concentrations of 7 (Determine 4B). The inhibition of colony formation in U118 cells was of 26.8% and 42.8% at 50 nM and 100 nM, respectively, in comparison with 48.7% and 74.2% inhibition noticed on the similar concentrations in U87 cells (Figures 4A, B). In distinction, EPNT-5 cells had been extra delicate to 7 therapy, exhibiting 56.1% and 69% inhibition at 50 nM and 100 nM, respectively, and full abrogation of colony progress at 450 nM and 1,000 nM (Determine 4C).
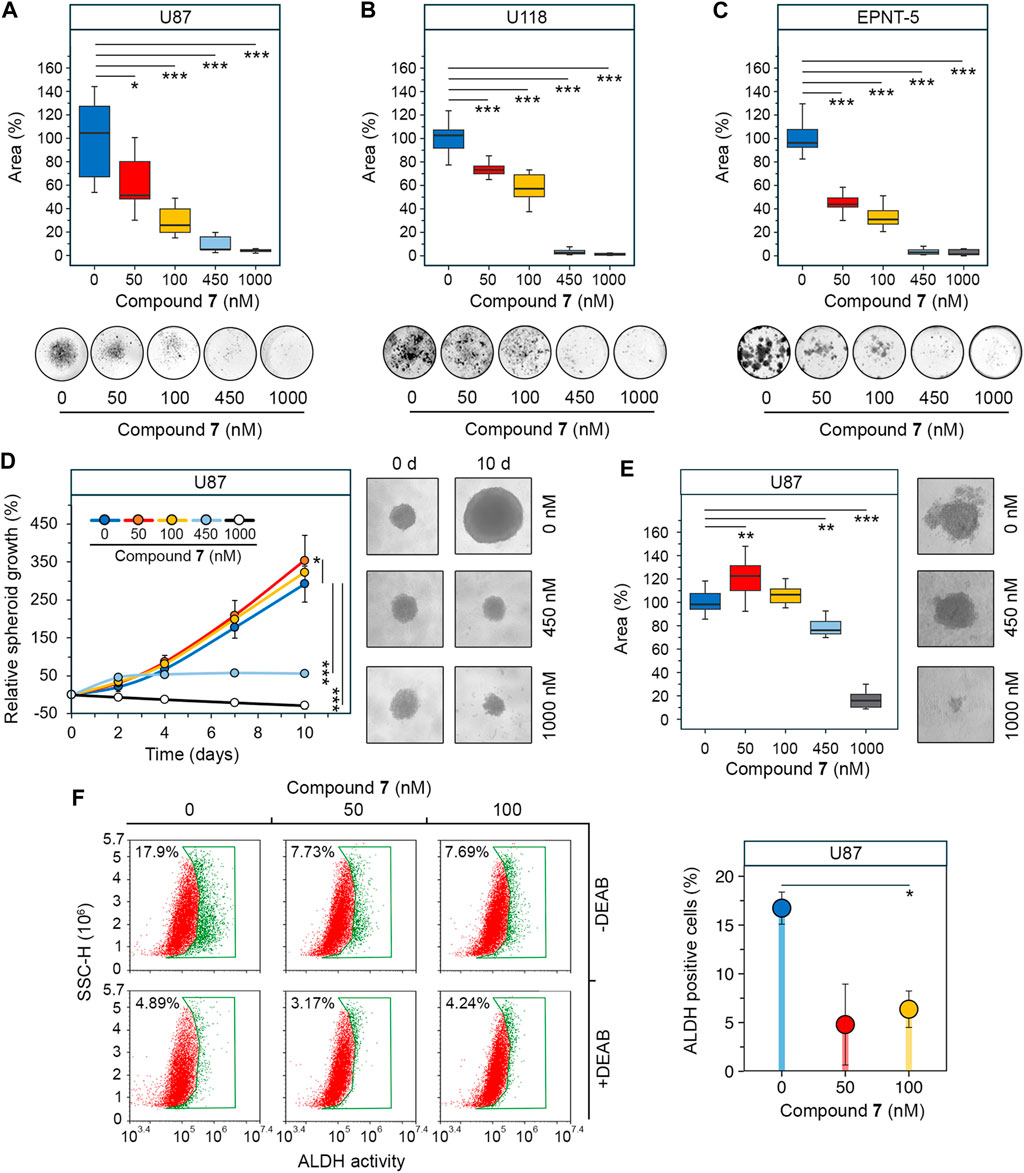
Determine 4. Impact of 7 on stemness of glioblastoma cells. (A–C) Colony forming exercise of U87 (A), U118 (B) and EPNT-5 (C) cells assessed after 10 days of incubation with 7 (0–1,000 nM) (n = 5). (D) Development of U87 major tumorspheres over 10 days of 7 therapy (0–1,000 nM) (n = 8). (E) Measurement of U87 secondary tumorspheres that had been fashioned from cells of major tumorspheres after therapy with 7 (0–1,000 nM) for 4 days (n = 8). (F) ALDH exercise in U87 major tumorspheres after 4 days of incubation with 7 (0–100 nM) for 4 days assessed utilizing AldeRed staining and circulate cytometry (n = 3, 104 occasions/pattern). Line graph information are represented as imply ± commonplace deviation (SD). Statistical significance was calculated by comparability with the untreated group. *, **, *** point out that p-values had been lower than 0.05, 0.01, and 0.001, respectively.
Since earlier research have proven that the stemness of glioblastoma cells is elevated when they’re grown in tumorspheres (Lee et al., 2016), we investigated the anti-stem cell exercise of 7 in a three-dimensional cell mannequin. U87 cells had been allowed to kind spheres below non-adherent circumstances and their progress was monitored within the presence of accelerating concentrations of 7. Determine 4D reveals that untreated tumorspheres grew progressively, reaching 2.9 occasions their authentic measurement by day 10. The impact of 7 on tumorspheres was concentration-dependent. Surprisingly, low concentrations of 7 stimulated spheroid progress, rising it by 21% at 50 nM (p-value <0.05) and 10.4% at 100 nM (not statistically important) in comparison with management (Determine 4D). In distinction, excessive concentrations of 7 inhibited spheroid progress, stopping it after 4 days at 450 nM or utterly blocking it at 1,000 nM. Notably, 7 at 1,000 nM brought on tumorsphere disruption, lowering their measurement by 28.6% in comparison with management by day 10 (Determine 4D). To evaluate the self-renewal capability, U87 tumorspheres had been uncovered to rising concentrations of compound 7 for 4 days. Afterward, the cells had been dissociated by trypsinization and left in non-adherent circumstances for 3 days to kind secondary tumorspheres. Much like major tumorspheres, 7 elevated the world of secondary tumorspheres by 22.6% at 50 nM (p-value <0.01) and 6.6% at 100 nM (not statistically important) (Determine 4E). In distinction, greater concentrations of 7 considerably decreased the self-renewal capability of U87 cells. Particularly, induction with 450 nM and 1,000 nM of 7 decreased secondary tumorsphere space by 23.9% and 84.1%, respectively (Determine 4E).
Compound 7 was hypothesized to have an effect on tumorsphere progress not solely via cytotoxicity but in addition by interfering with stem cell habits. Aldehyde dehydrogenases (ALDHs) induce stem cell traits by assuaging oxidative stress and activating the retinoic acid signaling pathway, making them a marker of GSCs (Xia et al., 2023), and ALDH exercise was discovered to be considerably elevated in tumorspheres in comparison with U87 cells cultured in monolayer (Supplementary Determine S2). ALDH exercise was analyzed in U87 tumorspheres after 4 days of induction with low-toxic concentrations of 7 utilizing AldeRed staining and circulate cytometry. The outcomes point out that untreated tumorspheres had excessive ALDH exercise, with 17.9% of cells being ALDH+ (Determine 4F). Therapy with 7 at 50 and 100 nM decreased the variety of ALDH + cells by 2.3-fold in comparison with the management, whatever the dose. The addition of DEAB, a selective inhibitor of ALDH, decreased the fluorescence in all samples to the basal stage, demonstrating that the noticed variation was as a consequence of ALDH exercise (Determine 4F). Thus, the outcomes obtained clearly indicated that the noticed progress of tumorspheres at low-toxic doses of 7 was not mediated by a rise within the GSC inhabitants. The poisonous profile of 7 revealed in U87 tumorspheres (Determine 4D) suggests the necessity for additional detailed research of the bioactivity of 7 at low concentrations and confirms its eligibility for glioblastoma therapy.
3.5 Compound 7 elevated the efficacy of temozolomide in glioblastoma cells by inducing ROS-dependent apoptosis
Since soloxolone tryptamide (compound 9) has been proven to induce apoptosis in glioblastoma cells (Markov et al., 2022b), we investigated whether or not this type of cell dying contributed to the cytotoxic results of soloxolone para-methylanilide 7 in U87 and U118 cells. Annexin V-FITC and propidium iodide double staining outcomes present that 7 induced apoptotic dying in each cell strains, with U118 cells being extra delicate. The share of U118 cells in the correct quadrants was 82.5%, 95.6%, and 96.4% at concentrations of 450 nM, 1,000 nM, and 2000 nM, respectively, in comparison with 3.7%, 40%, and 83.3% for U87 cells on the similar concentrations (Figures 5A, B). The vast majority of cells present process apoptosis had been at a late stage (higher proper quadrant), probably because of the late time level of 48 h (which corresponds to our MTT evaluations). Notably, 1,000 and 2000 nM of 7 induced necrosis in 23.4% and 14.8% of U87 cells, respectively (Determine 5A). In distinction, the proportion of necrotic U118 cells didn’t exceed 3.5% at any focus (Determine 5B). Thus, 7 induced the dying of glioblastoma cells primarily via apoptosis.
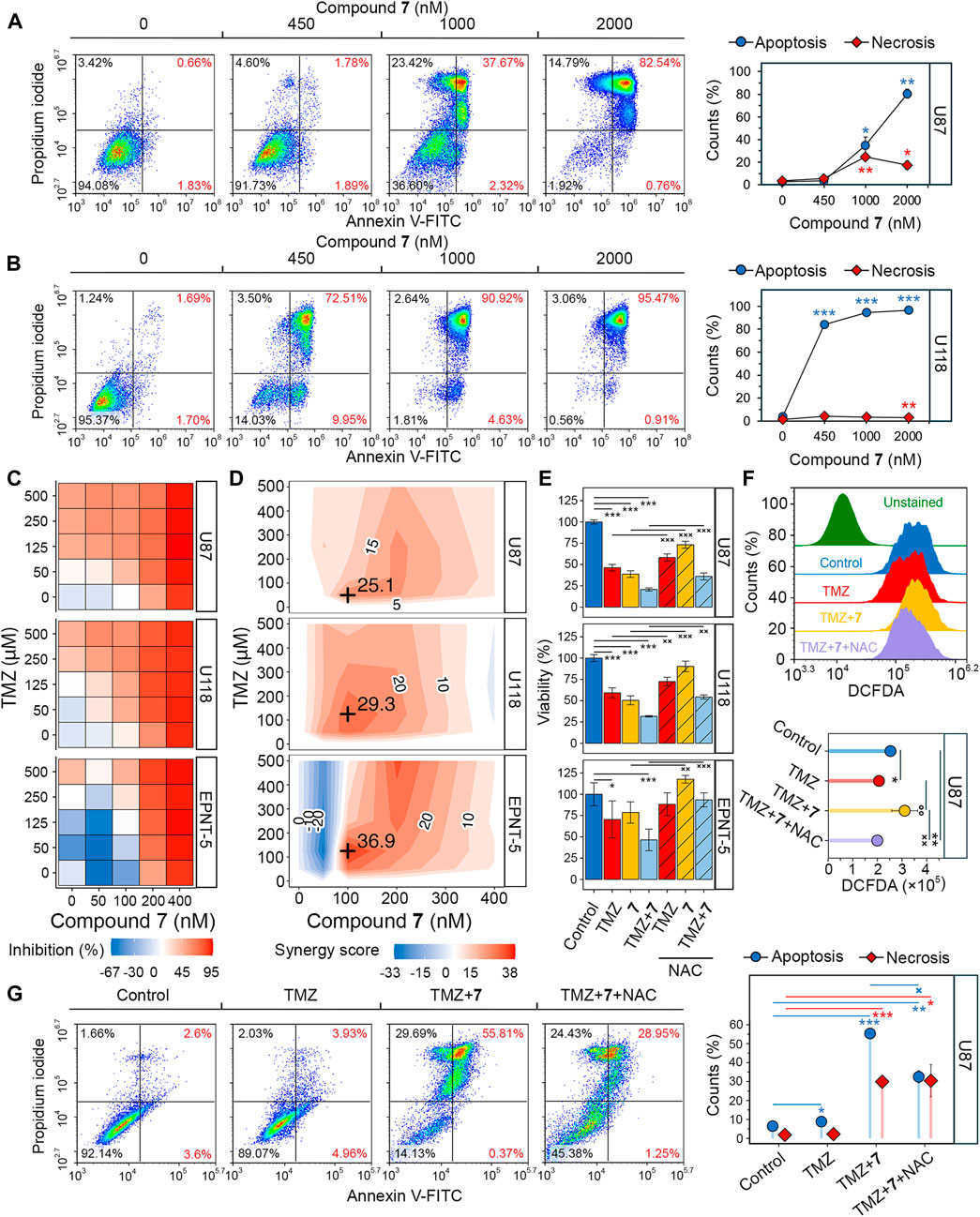
Determine 5. Analysis of the synergistic impact of 7 and TMZ towards glioblastoma cells. (A, B) Apoptosis and necrosis assessed in U87 (A) and U118 (B) cells after 48 h of incubation with 7 (0–2000 nM) utilizing Annexin V-FITC and propidium iodide double staining and circulate cytometry (n = 3, 104 occasions/pattern). (C) Cytotoxicity of mixtures of 7 (0–400 nM) and TMZ (0–500 μM) towards U87, U118 and EPNT-5 cells evaluated by MTT assay (n = 4). (D) Synergy rating (δ) throughout totally different concentrations of 7 and TMZ in U87, U118 and EPNT-5 cells (n = 4). δ was calculated utilizing the HSA mannequin within the SynergyFinder + platform. Black crosses point out most δ values. (E) The affect of ROS scavenging by NAC (2 mM) on the mixed cytotoxicity of TMZ (125 μM) and 7 (100 nM) in U87, U118, and EPNT-5 cells (n = 4). Incubation time: 72 h (F) ROS manufacturing in U87 cells after 72 h of incubation with TMZ (125 μM) and 7 (100 nM) assessed by DCFDA staining and circulate cytometry (n = 3, 104 occasions/pattern). (G) Apoptosis and necrosis assessed in U87 cells after 72 h of incubation with TMZ (500 μM), 7 (600 nM), and NAC (2 mM) utilizing Annexin V-FITC and propidium iodide double staining and circulate cytometry (n = 3, 104 occasions/pattern). Information in line graphs, bar graphs, and lollipop chart are represented as imply ± commonplace deviation (SD). Statistical significance was calculated by comparability with management (indicated by *) or the corresponding NAC-treated group (indicated by ×). */×, **/××, ***/××× point out that p-values had been lower than 0.05, 0.01, and 0.001, respectively.
Poor tumor cell response to TMZ is a serious concern within the therapy of glioblastoma, and mixture regimens with different chemotherapeutics have been developed to enhance survival outcomes (Tomar et al., 2021). To guage whether or not compound 7 might improve the efficacy of TMZ, we assessed the viability of glioblastoma cells after incubation with numerous dose mixtures of TMZ and 7 for 72 h. TMZ exhibited low cytotoxicity at micromolar concentrations (IC50U87 = 212.7 μM, IC50U118, EPNT-5 > 500 μM), whereas 400 nM 7 brought on virtually full dying of glioblastoma cells (IC50U87 = 197.2 nM, IC50U118 = 168.6 nM, IC50EPNT-5 = 199.3 nM) (Determine 5C). The mixed utility of TMZ and 7 resulted in elevated cytotoxicity in U87 and U118 cells in comparison with both compound alone. Notably, in EPNT-5 cells, a dose-dependent peculiarity within the cytotoxicity profile of 7 was revealed: the examined compound decreased TMZ-induced toxicity when used on the lowest focus of fifty nM and elevated it at greater concentrations (Determine 5C). Synergy scores (δ) had been calculated for every dose mixture in keeping with the HSA mannequin utilizing the SynergyFinder + platform. Analysis of drug interplay landscapes revealed a synergistic interaction between 7 and TMZ in U87 and U118 cells (Determine 5D). U118 cells confirmed a bigger space of excessive synergy in comparison with U87 cells, leading to the next δ worth (imply δU118 = 15 vs. imply δU87 = 11.6) (Figures 4C, D). Essentially the most important synergistic impact was noticed when 100 nM of 7 was mixed with 50 μM of TMZ in U87 cells (δU87 = 29.3) and 100 nM of 7 with 125 μM of TMZ in U118 cells (δU118 = 25.1). Nonetheless, no statistically important synergy of 7 with TMZ was present in EPNT-5 cells (imply δEPNT-5 = 6) because of the antagonistic impact of 7 at 50 nM on TMZ cytotoxicity (Determine 5D).
Dico et al. confirmed that TMZ cytotoxicity is restricted in drug-resistant glioblastoma cells as a result of it fails to advertise the manufacturing of reactive oxygen species (ROS) (Dico et al., 2019). On condition that compound 9 causes ROS-dependent dying of glioblastoma cells (Markov et al., 2022b), we investigated whether or not the synergistic impact of compound 7 with TMZ was as a consequence of ROS induction. Determine 5E reveals that co-incubation of U87 and U118 cells with the ROS scavenger N-acetyl-L-cysteine (NAC) for 72 h decreased the cytotoxic impact of 125 μM TMZ by 22.3% and 32.5%, respectively. The affect of NAC on the toxicity of 100 nM 7 was much more pronounced, leading to a 56.2% and 80.3% discount in U87 and U118 cells, respectively. Comparable traits had been noticed for co-treatment of EPNT-5 cells with NAC and TMZ or 7, though these information had been statistically insignificant. As anticipated, NAC decreased the cytotoxic impact of the mixture of TMZ and 7 by 19.3%, 33.1%, and 87.6% in U87, U118, and EPNT-5 cells, respectively (Determine 5E). DCFDA staining confirmed that this impact was as a consequence of ROS depletion. Co-treatment of U87 cells with TMZ and 7 for 72 h resulted in a 1.35-fold enhance in ROS in U87 cells, which was considerably suppressed by NAC (Determine 5F). Apparently, TMZ itself didn’t enhance ROS accumulation in U87 cells, which may be defined by the excessive basal ROS stage in U87 cells that’s attribute of TMZ-insensitive glioblastoma cells (Figures 5C, F) (Dico et al., 2019).
The mechanism of cell dying induced by the mixture of TMZ and 7 was additional investigated utilizing Annexin V-FITC/propidium iodide double staining. After incubation of U87 cells with 500 μM TMZ for 72 h, the apoptosis price was corresponding to that of the management group, with 9% of glioblastoma cells in the correct quadrants (Determine 5G). The mixture therapy of TMZ (500 μM) and 7 (600 nM) produced 6.2-fold greater exercise in comparison with TMZ alone, leading to 56.2% of U87 cells present process apoptosis. The addition of NAC decreased the noticed impact by 41%, which is according to the MTT outcomes (Determine 5E). Notably, NAC exhibited no impact on the 30% inhabitants of necrotic cells noticed with mixture therapy (Determine 5G). These outcomes display that 7 can improve the response of glioblastoma to TMZ via a mechanism involving ROS-dependent apoptosis.
3.6 Compound 7 successfully suppressed U87 glioblastoma progress in a mouse xenograft mannequin, each as a single agent and together with TMZ
To confirm the pronounced anti-glioblastoma efficiency of 7 and its potentiating impact on TMZ noticed in vitro, athymic nude mice bearing subcutaneously implanted U87 glioblastoma had been handled intraperitoneally with 7 (20 mg/kg), TMZ (10 mg/kg) or their mixture on the similar doses (Determine 6A). Compound 7 was administered thrice per week for a complete of seven injections, whereas TMZ was injected each day besides weekends for a complete of 11 occasions. Beginning on day 11, 7 was discovered to clearly suppress tumor progress, leading to 2.1-, 3.2-, 6.6-, and seven.2-fold discount in tumor quantity in comparison with the car group on days 11, 13, 15, and 18, respectively (Determine 6B), demonstrating comparable efficacy to TMZ. Notably, on day 13, the mixture scheme exceeded the results of 7 and TMZ administered alone by 3.9- and a couple of.2-fold, respectively (Determine 6B), which is per the power of 7 to boost cytotoxicity of TMZ noticed in vitro (Figures 5C, D). Nonetheless, with subsequent injections, the antitumor efficacy of the mixture of 7 with TMZ turns into corresponding to that of TMZ injected alone, attaining 17.9- and 25.8-fold reductions in tumor quantity in comparison with the car group on days 15 and 18, respectively (Determine 6B). On the finish of the experiment, along with the impact on tumor quantity, 7, TMZ and their mixture resulted in an 8-, 21.9- and 22.5-fold discount in tumor weight, respectively, in comparison with the car (Determine 6C). Evaluation of physique weight and organ indices confirmed no poisonous results of 7 and TMZ injected alone, whereas their mixture resulted in a slight discount in physique weight and a statistically important enhance in liver index, however since these modifications didn’t exceed 10% in comparison with the car group, the therapy routine used was thought of properly tolerated (Figures 6E, F).
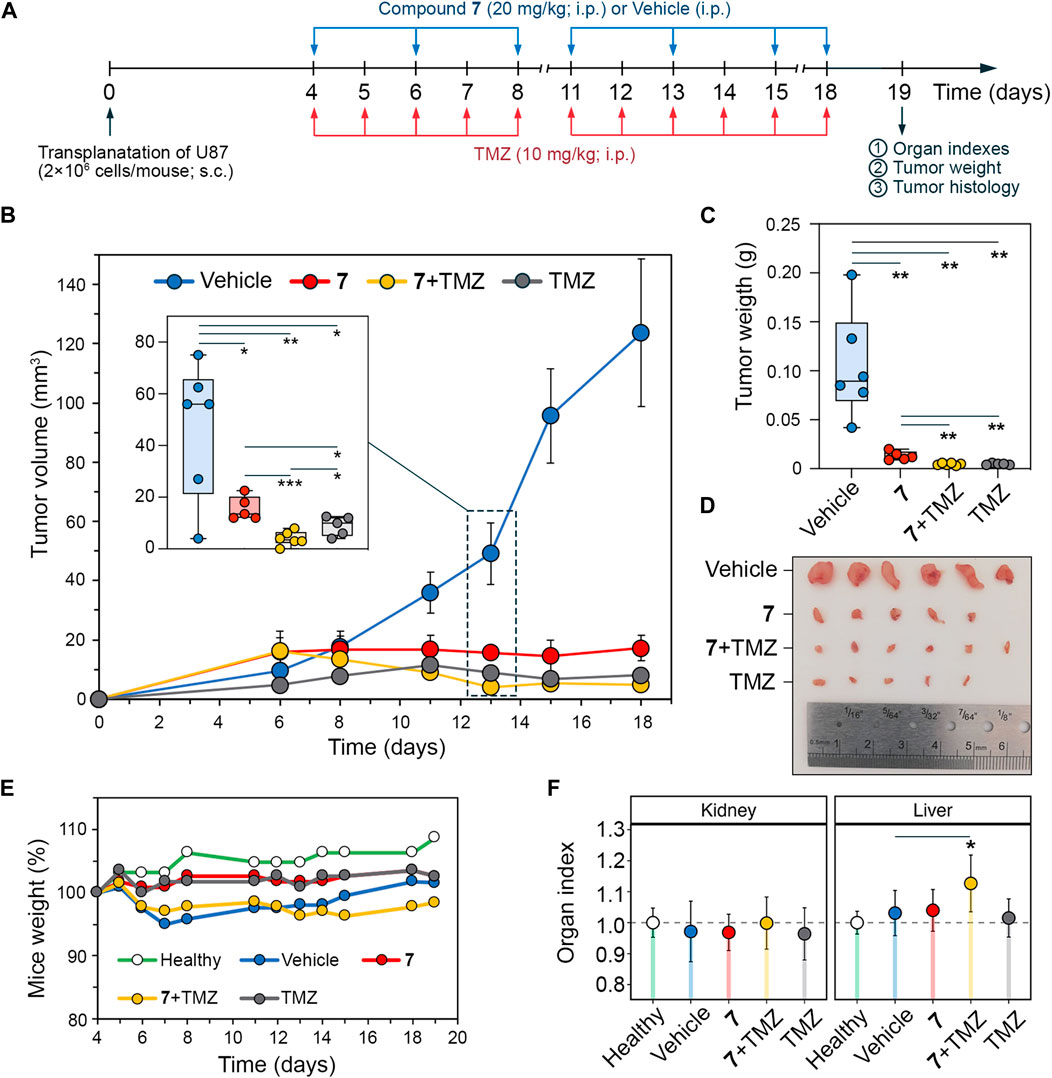
Determine 6. Antitumor impact of compound 7 and its mixture with TMZ on U87 glioblastoma xenograft mannequin. (A) Experimental setup. U87 glioblastoma cells had been subcutaneously (s.c.) implanted into athymic nude mice. On day 4 after tumor transplantation mice had been handled with compound 7 (20 mg/kg, i.p., n = 5), TMZ (10 mg/kg, i.p., n = 5) or their mixture on the similar doses and administration route (n = 6). Compound 7 was administered thrice per week for a complete of seven injections. TMZ was administered each day, besides weekends, for a complete of 11 injections. Car-treated mice had been used as management (n = 6). Mice had been sacrificed on day 19 after tumor transplantation, and materials (tumor nodes, livers, and kidneys) was collected for additional evaluation. (B–D) Compound 7 successfully suppresses the expansion of U87 glioblastoma. Tumor volumes (B), tumor weights (C), and first tumor nodes (D) of U87 glioblastoma with out therapy and after administration of 7, TMZ or their mixture. The insert reveals tumor volumes on day 13 of tumor progress. (E, D) Mice weights (E) and relative organ indices (F) of U87 glioblastoma-bearing mice with out therapy and after administration of 7, TMZ or their mixture.
Histologically, tumor nodes of U87 glioblastoma within the vehicle-treated group had been represented by sheets and bundles of undifferentiated polygonal to spindle-shaped cells with variable quantities of fibrovascular stroma (Determine 7A). The cells had been characterised by centrally situated spherical to irregular nuclei with punctate chromatin and 1-2 nucleoli and average nuclear to cytoplasmic ratio. Some cells, amounting to 4.3 ± 0.8 per area of view, contained mitotic occasions. Alongside the periphery of the tumor node, tumor cells turned extremely elongated forming a demarcating pseudocapsule. Immunohistochemical staining of U87 glioblastoma with major anti-Ki-67 antibody signifies that tumor tissue of the vehicle-treated group is characterised by the pronounced proliferative exercise, expressing in a considerable amount of Ki-67-positive cells within the tumor construction with a numerical density of 62.3 ± 2.9 within the sq. unit of tumor part (Figures 7B, C). A major 13- to 17-fold discount within the variety of mitoses was noticed in tumor nodes handled with any of the regimens used in comparison with car (Determine 7D). In line with this, tumors following compound 7 and mixture remedy had been characterised by a big 4-fold discount within the numerical density of Ki-67-positive cells in comparison with the vehicle-treated group (Determine 7B). TMZ was discovered to end in an virtually full disappearance of Ki-67 optimistic cells within the tumor tissue, expressed by a 9-fold lower of their numerical density in comparison with car. Surprisingly, regardless of an identical impact on the variety of mitoses within the tumor, the impact of TMZ on Ki-67 optimistic cells was extra pronounced than that of 7 administered alone or together with TMZ (Determine 7B).
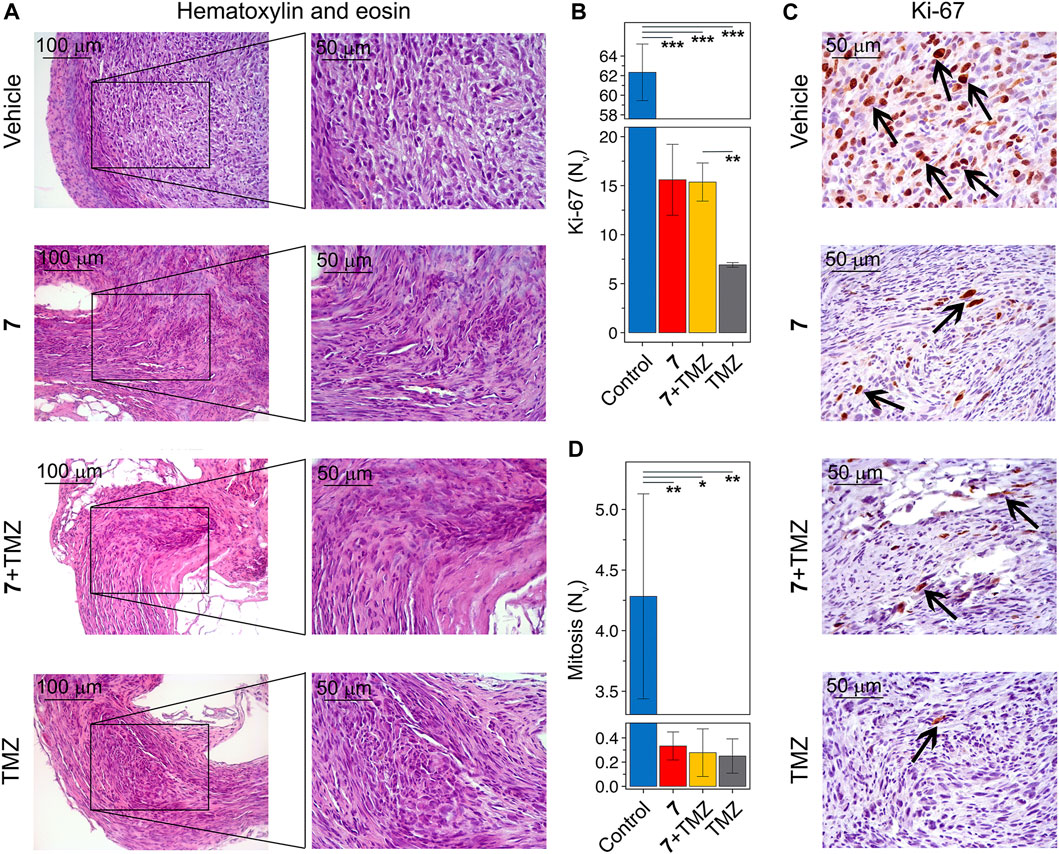
Determine 7. Histological construction of U87 glioblastoma with out therapy and after administration of 7, TMZ or their mixture. (A) Consultant histological photos of U87 glioblastoma in management and experimental teams. The black bins present areas that had been examined additional at the next magnification. Hematoxylin and eosin staining. Unique magnification ×200 (left panel) and ×400 (proper panel). (B) The numerical density (Nv) of tumor cells within the state of mitosis (n = 5–6). (C) Immunohistochemical staining of U87 glioblastoma with major anti-Ki-67 antibodies. The black arrows point out Ki-67 optimistic cells. Unique magnification ×400. (D) The numerical density (Nv) of Ki-67 optimistic cells (n = 3).
Lastly, the impact of 7 and its mixture with TMZ on the expression of markers related to glioblastoma aggressiveness was evaluated. We chosen N-cadherin, a mesenchymal-type marker that’s delicate to 7 in U87 cells in vitro (Determine 3F), and glial fibrillary acidic protein (GFAP), which performs an necessary regulatory function in glioma cell motility (Uceda-Castro et al., 2022), as markers of curiosity. Further evaluation of the Chinese language Glioma Genome Atlas (CCGA) confirmed that top expression ranges of CDH2 and GFAP genes encoding these proteins had been considerably related to poor survival in sufferers with recurrent glioma (Determine 8A), and in keeping with the Repository for Molecular BRAin Neoplasia DaTa (REMBRANDT), their overexpression is attribute of mesenchymal-type glioblastoma cells in comparison with much less aggressive proneural-type glioblastoma cells (Determine 8B). Immunohistochemical staining of vehicle-treated U87 glioblastoma sections with anti-N-cadherin and anti-GFAP antibodies confirmed excessive expression of each markers, whereas administration of 7, its mixture with TMZ, and TMZ alone considerably decreased their ranges, with essentially the most pronounced impact noticed in mice handled with TMZ or mixed remedy (Determine 8C). These outcomes independently verify the power of 7 to inhibit N-cadherin expression in glioblastoma cells noticed in vitro (Determine 3F) and are per not too long ago printed information on the power of TMZ to inhibit GFAP expression in U87 glioblastoma in a murine xenograft mannequin (Bakhtiyari-Ramezani et al., 2024).
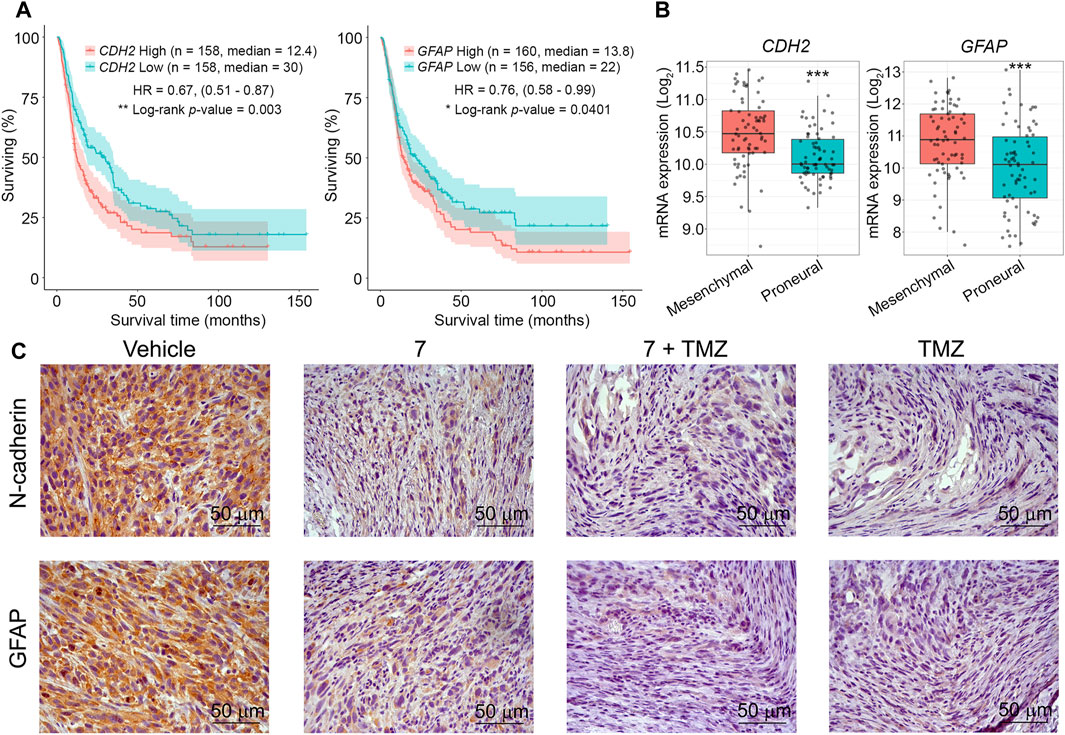
Determine 8. The impact of 7, TMZ or their mixture on the expression of N-cadherin and GFAP in U87 glioblastoma. (A) Survival of sufferers with recurrent glioma relying on the expression ranges of CDH2 and GFAP (CCGA database). (B) Expression ranges of CDH2 and GFAP in mesenchymal- and proneural-type glioblastoma (REMBRANDT database). *** signifies that p-values had been lower than 0.001. (C) Consultant photos of U87 glioblastoma immunohistochemically stained with major anti-N-cadherin (higher panel) and anti-GFAP (decrease panel) antibodies. Unique magnification ×400.
Thus, the outcomes of the in vivo experiment confirmed the pronounced anti-glioblastoma potential of 7 and its skill to boost the antitumor impact of TMZ, however this enhancement within the mouse mannequin was short-term, which might be associated to the peculiarities of pharmacokinetics and biodistribution of 7 and requires additional cautious research.
4 Dialogue
Glioblastoma is the commonest major mind tumor, with an annual incidence of three–4 instances per 100,000 person-years (Grochans et al., 2022). The present commonplace of care is maximal protected resection adopted by radiotherapy and TMZ chemotherapy (Rodríguez-Camacho et al., 2022). Nonetheless, its efficacy is restricted and recurrence is virtually inevitable as a consequence of two fundamental elements. First, glioblastoma infiltrates the mind tissue, making full resection inconceivable (Seker-Polat et al., 2022). Second, therapeutic stress promotes the survival of radio- and chemoresistant tumor cells, resulting in difficult-to-treat relapses (Wu W. et al., 2021). Subsequently, novel therapeutic approaches are wanted to fight glioblastoma invasion and drug resistance.
Pure metabolites with multi-target results are thought of a promising supply of latest drug candidates for glioblastoma remedy. Thus far, numerous pure compounds, together with sesquiterpene lactones (Hsu et al., 2024), endocannabinoids (Duzan et al., 2023), quinones (Liu et al., 2024), flavonoids (Majchrzak-Celińska and Studzińska-Sroka, 2024), terpenoids (Wang and Hu, 2023) and others, have been reported to have pronounced anti-glioblastoma potential. A number of pentacyclic triterpenoids (PTs) have proven promising anti-glioblastoma exercise in preclinical research, together with ursolic acid (Wang et al., 2012; Lu et al., 2014; Bergamin et al., 2017), asiatic acid (Kavitha et al., 2015; Thakor et al., 2017), oleanolic acid (Fujiwara et al., 2011), and celastrol (Boridy et al., 2014; Cha et al., 2019), which raises the prospect of additional PTs derivatization for the event of efficient anti-glioblastoma medication (Bache et al., 2014; Ciftci et al., 2021; Tsai et al., 2021; Markov et al., 2022b; Markov et al., 2023; Tsai et al., 2023). The addition of a cyanoenone group to the A-ring of oleanolic and 18βH-glycyrrhetinic acids led to the event of bardoxolone (CDDO) and soloxolone methyl (SM), semisynthetic triterpenoids with pronounced antitumor potential (Logashenko et al., 2011; Borella et al., 2019). To boost their applicability for mind dysfunction remedy, amide derivatives of CDDO and SM with improved blood-brain barrier permeability and excessive anti-glioblastoma efficiency had been synthesized (Stack et al., 2010; Markov et al., 2022b). The trifluoroethylamide spinoff of CDDO (CDDO-TFEA) was proven to scale back the viability of GBM8401 and U87 cells by way of AKT-mediated transcriptomic regulation of key genes related to cell cycle, apoptosis, and autophagy (Tsai et al., 2021; Tsai et al., 2023). The soloxolone tryptamide (compound 9) inhibited the expansion of U87 and U118 cells each in vitro and in vivo by inducing intrinsic apoptosis and inhibiting angiogenesis (Markov et al., 2022b; Markov et al., 2023). Subsequently, PTs symbolize a promising platform for the event of novel anti-glioblastoma medication.
The invasion of glioblastoma cells into mind tissue is facilitated by glial-mesenchymal transition (GMT). This course of represents a change of glioblastoma cells from a proneural to a mesenchymal subtype that features morphological modifications, lack of cell-cell contacts and enhancement of cell-ECM adhesion, elevated motility and transcriptomic alterations (Joseph et al., 2014; Ouanouki et al., 2018; Xu et al., 2022). Though concentrating on GMT-associated pathways has proven promise, it has not yielded important ends in the scientific setting, indicating the necessity for additional optimization of this technique (Lai et al., 2024). Though PTs have been proven to inhibit glioblastoma motility (Guimarães et al., 2017; Thakor et al., 2017; Li et al., 2018; Solar et al., 2021), their results on the molecular traits of GMT haven’t been characterised. To our data, solely two PT saponins, raddeanin A and tubeimoside-1, have been reported to downregulate the basal stage of GMT markers together with their suppressive impact on glioblastoma cell migration and invasion within the absence of GMT inducers (Cao et al., 2019; Wu B. et al., 2021). On condition that mesenchymal-like options in glioblastoma cells may be considerably enhanced by numerous tumor-related processes, corresponding to hypoxia, necrosis and irritation (Kim et al., 2021), we determined to research the impact of soloxolone amides on GMT induced by reworking progress issue beta (TGF-β), a recognized GMT activator produced by microglia, tumor-associated macrophages, and tumor cells themselves within the glioblastoma microenvironment (Golán-Cancela and Caja, 2024). In line with earlier information, soloxolone amides inhibited GMT-associated processes in U87 cells, together with modifications in mobile morphology and adhesiveness (Figures 2B–D). Moreover, hit compound 7 bearing the p-tolylamide group, decreased motility and the expression of the mesenchymal markers N-cadherin, fibronectin, and Slug in U87 and U118 cells induced with TGF-β1 (Figures 3D–G). The demonstrated skill of 7 to upregulate OLIG2 in TGF-β1-stimulated U87 cells (Determine 3F) might, on the one hand, additionally attest to its anti-GMT efficiency, as OLIG2 performs an necessary pro-neural operate within the growth of the central neural system and is a marker for the proneural glioblastoma subtype (Popova et al., 2014), however alternatively, the noticed, albeit weak, enhance in OLIG2 expression below compound 7 therapy can also display the induction of a compensatory response of glioblastoma cells to the investigated triterpenoid. Based on Szu et al., upregulation of OLIG2 is attribute of glioma cells and is related to their excessive proliferative potential, and pharmacological inhibition of OLIG2 considerably improved the survival of mice with high-grade glioma and medulloblastoma (Szu et al., 2023). Contemplating that the upregulation of OLIG2 by compound 7 was nonetheless weakly expressed (Determine 3F), this impact of the investigated triterpenoid was not accompanied with enhanced cell proliferation.
Upon binding to TGF-β, the sort II receptor (TβRII) on the floor of glioblastoma cells recruits and phosphorylates the sort I receptor (TβRI). This activation results in phosphorylation of SMAD2 and SMAD3, which then complicated with SMAD4 and translocate to the nucleus to induce transcription of genes regulating GMT (Golán-Cancela and Caja, 2024). Compound 7 was proven to dam SMAD2/3 nuclear translocation in U87 cells upon TGF-β induction (Figures 3H, I). This exercise is much like that beforehand reported by Wang et al. for ursolic acid, which successfully inhibited SMAD2/3 phosphorylation in U251 cells (Wang et al., 2012). Molecular docking simulations revealed that compound 7 can kind steady complexes with TβRI and TβRII (Figures 3J, Okay), and these interactions might underlie the noticed modulatory impact of 7 on the nuclear-to-cytoplasmic ratio of SMAD2/3. Notably, the limonoid isotoosendanin, which is structurally much like PTs, binds to the kinase area of TβRI in a way much like 7 by interacting with Lys 232, thereby lowering the affiliation of TβRI with SMAD2/3 (Zhang et al., 2023). As well as, oleanolic, ursolic, and asiatic acids, in addition to the triterpene saponin 20(S)-ginsenoside Rg3, have been proven to bind on to TβRI, however the exact determinants governing these interactions stay to be elucidated (Yoshimura et al., 2003; Zhang et al., 2022; Xu et al., 2023). Whereas a pc simulation means that 7 might work together with TβRI, additional experimental verification is required to substantiate this exercise.
Glioblastoma stem cells (GSCs) play a important function in therapeutic resistance as a consequence of their skill to face up to the results of radiation and chemotherapy in a quiescent state and subsequently resume tumor progress (Xie et al., 2022). Varied GSC-targeting therapeutics are below growth, and a few have entered scientific trials (Sahoo et al., 2024). Within the area of PT-based drug candidates, β-escin and 7β-22 dihydroxyhopane have been proven to selectively goal proliferation and induce apoptosis in GSCs however not in differentiated glioblastoma cells. As well as, they block GSC stemness, as evidenced by the inhibition of tumorsphere formation, self-renewal capability, and expression of stem cell markers (Harford-Wright et al., 2016; Kim et al., 2022). In line with these information, compound 7 exhibited anti-stem cell exercise by inhibiting colony formation (Figures 4A–C), tumorsphere progress and renewal (Figures 4D, E), and the exercise of the stem cell marker aldehyde dehydrogenase (ALDH) (Determine 4F). Nonetheless, in distinction to β-escin and 7β-22 dihydroxyhopane, 7 not solely affected GSCs but in addition confirmed cytotoxicity towards differentiated glioblastoma cells (Figures 2A, 3A). The impact of 7 on the expansion of U87 tumorspheres, each major and secondary, was, surprisingly, dose-dependent. At a low poisonous focus of fifty nM, 7 elevated the tumorsphere measurement in comparison with the management, whereas its greater, extra poisonous concentrations of 450 and 1,000 nM resulted in a big discount of spheroid progress as much as its full blockage (Figures 4D, E). Given the noticed lower in stem cell-related ALDH exercise in U87 cells handled with 7 at 50 nM (Determine 4F), we speculate that 7 at low concentrations might induce the transition of GSCs into differentiated tumor cells with better proliferative exercise by suppressing their stemness, however this impact could also be counteracted by the cytotoxicity of 7 at greater concentrations (Lan et al., 2017). The noticed peculiarities within the impact of 7 on three-dimensional tumor progress recommend the necessity for additional detailed research on its pharmacokinetics.
Temozolomide (TMZ) is a first-line drug for glioblastoma that interferes with tumor cell proliferation by alkylating DNA guanines (Teraiya et al., 2023). Intrinsic and purchased resistance to TMZ stays a serious problem within the therapy of glioblastoma. One method to deal with this subject is to mix TMZ with cytotoxic compounds which have different mechanisms for inducing cell dying (Tomar et al., 2021). The outcomes present that compound 7 has the next cytotoxic potential and pro-apoptotic exercise towards glioblastoma cells in vitro than TMZ (Figures 5A, E, G), whereas within the murine U87 xenograft mannequin, these compounds exhibited comparable ranges of tumor progress inhibition (Figures 6B–D), accompanied by a marked discount within the variety of mitosis and Ki-67 optimistic cells (Determine 7B). The mixed utility of 7 and TMZ resulted in a synergistic discount of viability and induction of apoptosis in glioblastoma cells in vitro (Figures 5C, D), which was partially verified within the xenograft mannequin. Within the mid-term (13 days after tumor transplantation), co-administration of 7 and TMZ resulted in a better suppression of tumor progress in comparison with both agent administered alone (Determine 6B). Nonetheless, on the finish of the experiment, the tumor inhibitory impact of the mixture remedy was corresponding to that of TMZ, suggesting the necessity for extra pharmacokinetic and distribution research to find out the suitable pharmacological kind, route, and schedule of administration of 7 to extend its synergistic impact with TMZ in vivo.
Reactive oxygen species (ROS) play a twin function in glioblastoma. Whereas ROS promote tumor cell proliferation, their extreme accumulation can injury cell lipids, proteins, and DNA, resulting in apoptosis (Campos-Sandoval et al., 2021; Chien et al., 2021). Due to this double-edged impact, each antioxidant (McConnell et al., 2018; Tai et al., 2021) and prooxidant (Yuan et al., 2012; Grogan et al., 2014; Yin et al., 2014) compounds have been proven to sensitize glioblastoma cells to TMZ. With respect to PTs, Barbarisi et al. discovered that boswellic acid had a synergistic impact with TMZ whereas lowering ROS, lipid peroxides, and nitric oxide in glioblastoma cells (Barbarisi et al., 2019). In distinction to this research, the cytotoxicity of 7, used each alone and together with TMZ, was extremely depending on the induction of ROS (Figures 5E, F), as ROS depletion clearly decreased this exercise (Determine 5E), and, furthermore, decreased the pro-apoptotic impact of the mixture routine (Determine 5G). These outcomes are per our earlier in-depth investigation of the mechanisms of cell dying induced by soloxolone amides (Markov et al., 2022b). It’s noteworthy that some glioblastoma cells endure necrosis after incubation with 7 (Figures 5A, G). Lu et al. beforehand demonstrated that ursolic acid induces necrosis in TMZ-resistant glioblastoma cells by opening the mitochondrial permeability transition pore (MPTP) in a ROS-dependent method, leading to decreased ATP ranges and prevention of the apoptotic course of (Lu et al., 2014). On condition that necrosis is concerned in glioblastoma development (Markwell et al., 2022), additional analysis is required to research the therapeutic function and mechanism of compound 7-induced necrosis.
Evaluating the outcomes described above with the outcomes of our latest research on the anti-glioblastoma potential of compound 9 (also called soloxolone tryptamide) (Markov et al., 2022b; Markov et al., 2023), it may be concluded that compound 7, regardless of a comparable impact on glioblastoma cell morphology and adhesion as compound 9 (Figures 2B, C), displays extra pronounced antitumor properties as a result of:
• 7 is extra cytotoxic (Determine 2A);
• 7 reveals a stronger suppressive impact on spheroid progress (7 brought on a large-scale discount of spheroids at 1 µM (Determine 4D), whereas 9 confirmed an identical impact solely at 4 µM (Markov et al., 2023));
• 7 has a extra pronounced anti-glioblastoma impact in vivo (on the finish of the experiment, 7 decreased tumor weight 8-fold in comparison with the vehicle-treated group (Determine 6C), whereas 9, administered at an identical dose and schedule, decreased this parameter solely 3.4-fold (Markov et al., 2022a)).
As well as, our findings considerably develop our data of the anti-glioblastoma potential of cyanoenone-containing triterpenoids, as this research is the primary to display their skill to inhibit the TGF-β-induced aggressive phenotype of glioblastoma cells.
Regardless of the proof for potent anti-glioblastoma properties of compound 7 in vitro and in vivo, the current research has a number of limitations. First, the direct interplay of 7 with TβRI/II requires additional experimental verification by floor plasmon resonance or affinity labeling competitors assays. Second, for a extra full understanding of the inhibitory impact of 7 on the TGF-β/Smad signaling axis, it’s essential to moreover consider its impact on nuclear translocation not solely of SMAD2/3 but in addition of SMAD4, as a result of in keeping with Fink et al. SMAD4, though not essentially required for SMAD2/3 translocation to the nucleus, is important for the TGF-β-associated transcriptional response (Fink et al., 2003). Third, given the significance of oxidative stress in compound 7-induced glioblastoma cell dying, the impact of 7 on ROS technology at early time factors needs to be evaluated. Fourth, as talked about above, an in depth research of the pharmacokinetics and biodistribution of 7 is required to extra precisely assess the efficacy of mixed remedy of glioblastoma with 7 and TMZ in vivo.
5 Conclusion
Our research demonstrated that soloxolone para-methylanilide 7 successfully inhibited the viability, invasiveness, and stemness of glioblastoma cells. Importantly, 7 reversed GMT via the TGF-β1/SMAD2/3 pathway and enhanced the cytotoxicity of TMZ by inducing ROS-dependent apoptosis. These results make 7 a possible candidate for inclusion together therapy regimens for glioblastoma.
Information availability assertion
The unique contributions offered within the research are included within the article/Supplementary Materials, additional inquiries may be directed to the corresponding creator.
Ethics assertion
Moral approval was not required for the research on people in accordance with the native laws and institutional necessities as a result of solely commercially out there established cell strains had been used. The animal research was permitted by the Committee on the Ethics of Animal Experiments on the Institute of Cytology and Genetics SB RAS. The research was carried out in accordance with the native laws and institutional necessities.
Writer contributions
KO: Information curation, Formal Evaluation, Investigation, Methodology, Software program, Validation, Visualization, Writing–authentic draft, AS’okay: Formal Evaluation, Investigation, Methodology, Writing–authentic draft. OS: Sources, Writing–assessment and modifying. OM: Formal Evaluation, Investigation, Methodology, Writing–assessment and modifying. NS: Supervision, Writing–assessment and modifying. MZ: Funding acquisition, Sources, Supervision, Writing–assessment and modifying. AM: Conceptualization, Funding acquisition, Methodology, Mission administration, Sources, Supervision, Writing–authentic draft, Writing–assessment and modifying.
Funding
The creator(s) declare that monetary assist was obtained for the analysis, authorship, and/or publication of this text. This work was supported by the Russian Science Basis (Grant No. 23-14-00374) (in silico, in vitro and in vivo research) and partly by the Russian state-funded funds challenge of ICBFM SB RAS No. 121031300044-5 (cell upkeep and use of apparatus).
Acknowledgments
The authors gratefully thank Albina V. Vladimirova (Institute of Chemical Biology and Basic Drugs SB RAS, Novosibirsk, Russia) for cell upkeep.
Battle of curiosity
The authors declare that the analysis was carried out within the absence of any industrial or monetary relationships that might be construed as a possible battle of curiosity.
Writer’s observe
All claims expressed on this article are solely these of the authors and don’t essentially symbolize these of their affiliated organizations, or these of the writer, the editors and the reviewers. Any product that could be evaluated on this article, or declare that could be made by its producer, shouldn’t be assured or endorsed by the writer.
Supplementary materials
The Supplementary Materials for this text may be discovered on-line at: https://www.frontiersin.org/articles/10.3389/fphar.2024.1428924/full#supplementary-material
References
Ali, M. Y., Oliva, C. R., Noman, A. S. M., Allen, B. G., Goswami, P. C., Zakharia, Y., et al. (2020). Radioresistance in glioblastoma and the event of radiosensitizers. Cancers (Basel) 12, 2511. doi:10.3390/cancers12092511
PubMed Summary | CrossRef Full Textual content | Google Scholar
Alper, P., Salomatina, O. V., Salakhutdinov, N. F., Ulukaya, E., and Ari, F. (2021). Soloxolone methyl, as a 18βH-glycyrrhetinic acid derivate, might end in endoplasmic reticulum stress to induce apoptosis in breast most cancers cells. Bioorg. Med. Chem. 30, 115963. doi:10.1016/j.bmc.2020.115963
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bache, M., Bernhardt, S., Passin, S., Wichmann, H., Hein, A., Zschornak, M., et al. (2014). Betulinic acid derivatives NVX-207 and B10 for therapy of glioblastoma—an in vitro research of cytotoxicity and radiosensitization. Int. J. Mol. Sci. 15, 19777–19790. doi:10.3390/ijms151119777
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bakhtiyari-Ramezani, M., Nohekhan, M., Akbari, M. E., Abbasvandi, F., Bayat, M., Akbari, A., et al. (2024). Comparative evaluation of direct and oblique chilly atmospheric plasma results, based mostly on helium and argon, on human glioblastoma: an in vitro and in vivo research. Sci. Rep. 14, 3578. doi:10.1038/s41598-024-54070-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
Barbarisi, M., Barbarisi, A., De Sena, G., Armenia, E., Aurilio, C., Libutti, M., et al. (2019). Boswellic acid has anti-inflammatory results and enhances the anticancer actions of Temozolomide and Afatinib, an irreversible ErbB household blocker, in human glioblastoma cells. Phyther. Res. 33, 1670–1682. doi:10.1002/ptr.6354
PubMed Summary | CrossRef Full Textual content | Google Scholar
Becker, A. P., Sells, B. E., Jaharul Haque, S., and Chakravarti, A. (2021). Tumor heterogeneity in glioblastomas: from gentle microscopy to molecular pathology. Cancers (Basel) 13, 761. doi:10.3390/cancers13040761
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bergamin, L. S., Figueiró, F., Dietrich, F., Manica, F. D. M., Filippi-Chiela, E. C., Mendes, F. B., et al. (2017). Interference of ursolic acid therapy with glioma progress: an in vitro and in vivo research. Eur. J. Pharmacol. 811, 268–275. doi:10.1016/j.ejphar.2017.06.030
PubMed Summary | CrossRef Full Textual content | Google Scholar
Biserova, Okay., Jakovlevs, A., Uljanovs, R., and Strumfa, I. (2021). Most cancers stem cells: significance in origin, pathogenesis and therapy of glioblastoma. Cells 10, 621. doi:10.3390/cells10030621
PubMed Summary | CrossRef Full Textual content | Google Scholar
Borella, R., Forti, L., Gibellini, L., De Gaetano, A., De Biasi, S., Nasi, M., et al. (2019). Synthesis and anticancer exercise of CDDO and CDDO-me, two derivatives of pure triterpenoids. Molecules 24, 4097. doi:10.3390/molecules24224097
PubMed Summary | CrossRef Full Textual content | Google Scholar
Boridy, S., Le, P. U., Petrecca, Okay., and Maysinger, D. (2014). Celastrol targets proteostasis and acts synergistically with a heat-shock protein 90 inhibitor to kill human glioblastoma cells. Cell Dying Dis. 5, e1216. doi:10.1038/cddis.2014.182
PubMed Summary | CrossRef Full Textual content | Google Scholar
Bowman, R. L., Wang, Q., Carro, A., Verhaak, R. G. W., and Squatrito, M. (2017). GlioVis information portal for visualization and evaluation of mind tumor expression datasets. Neuro. Oncol. 19, 139–141. doi:10.1093/neuonc/now247
PubMed Summary | CrossRef Full Textual content | Google Scholar
Campos-Sandoval, J. A., Gómez-García, M. C., Santos-Jiménez, J. D. L., Matés, J. M., Alonso, F. J., and Márquez, J. (2021). Antioxidant responses associated to temozolomide resistance in glioblastoma. Neurochem. Int. 149, 105136. doi:10.1016/j.neuint.2021.105136
PubMed Summary | CrossRef Full Textual content | Google Scholar
Cao, J., Zhao, E., Zhu, Q., Ji, J., Wei, Z., Xu, B., et al. (2019). Tubeimoside-1 inhibits glioblastoma progress, migration, and invasion by way of inducing ubiquitylation of MET. Cells 8, 774. doi:10.3390/cells8080774
PubMed Summary | CrossRef Full Textual content | Google Scholar
Cha, Z., Cheng, J., Xiang, H., Qin, J., He, Y., Peng, Z., et al. (2019). Celastrol enhances TRAIL-induced apoptosis in human glioblastoma by way of the dying receptor pathway. Most cancers Chemother. Pharmacol. 84, 719–728. doi:10.1007/s00280-019-03900-8
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chaikuad, A., and Bullock, A. N. (2016). Structural foundation of intracellular TGF-β signaling: receptors and smads. Chilly Spring Harb. Perspect. Biol. 8, a022111. doi:10.1101/cshperspect.a022111
PubMed Summary | CrossRef Full Textual content | Google Scholar
Chien, C.-H., Hsueh, W.-T., Chuang, J.-Y., and Chang, Okay.-Y. (2021). Dissecting the mechanism of temozolomide resistance and its affiliation with the regulatory roles of intracellular reactive oxygen species in glioblastoma. J. Biomed. Sci. 28, 18. doi:10.1186/s12929-021-00717-7
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ciftci, H. I., Radwan, M. O., Sever, B., Hamdy, A. Okay., Emirdağ, S., Ulusoy, N. G., et al. (2021). EGFR-targeted pentacyclic triterpene analogues for glioma remedy. Int. J. Mol. Sci. 22, 10945. doi:10.3390/ijms222010945
PubMed Summary | CrossRef Full Textual content | Google Scholar
Dico, A. L., Salvatore, D., Martelli, C., Ronchi, D., Diceglie, C., Lucignani, G., et al. (2019). Intracellular redox-balance involvement in temozolomide resistance-related molecular mechanisms in glioblastoma. Cells 8, 1315. doi:10.3390/cells8111315
PubMed Summary | CrossRef Full Textual content | Google Scholar
Duzan, A., Reinken, D., McGomery, T. L., Ferencz, N. M., Plummer, J. M., and Basti, M. M. (2023). Endocannabinoids are potential inhibitors of glioblastoma multiforme proliferation. J. Integr. Med. 21, 120–129. doi:10.1016/j.joim.2023.01.005
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fink, S. P., Mikkola, D., Willson, J. Okay. V., and Markowitz, S. (2003). TGF-beta-induced nuclear localization of Smad2 and Smad3 in Smad4 null most cancers cell strains. Oncogene 22, 1317–1323. doi:10.1038/sj.onc.1206128
PubMed Summary | CrossRef Full Textual content | Google Scholar
Fujiwara, Y., Komohara, Y., Kudo, R., Tsurushima, Okay., Ohnishi, Okay., Ikeda, T., et al. (2011). Oleanolic acid inhibits macrophage differentiation into the M2 phenotype and glioblastoma cell proliferation by suppressing the activation of STAT3. Oncol. Rep. 26, 1533–1537. doi:10.3892/or.2011.1454
PubMed Summary | CrossRef Full Textual content | Google Scholar
Grochans, S., Cybulska, A. M., Simińska, D., Korbecki, J., Kojder, Okay., Chlubek, D., et al. (2022). Epidemiology of glioblastoma multiforme–literature assessment. Cancers (Basel) 14, 2412. doi:10.3390/cancers14102412
PubMed Summary | CrossRef Full Textual content | Google Scholar
Grogan, P. T., Sarkaria, J. N., Timmermann, B. N., and Cohen, M. S. (2014). Oxidative cytotoxic agent withaferin A resensitizes temozolomide-resistant glioblastomas by way of MGMT depletion and induces apoptosis via Akt/mTOR pathway inhibitory modulation. Make investments. New Medication 32, 604–617. doi:10.1007/s10637-014-0084-7
PubMed Summary | CrossRef Full Textual content | Google Scholar
Guan, F., Wang, X., and He, F. (2015). Promotion of cell migration by neural cell adhesion molecule (NCAM) is enhanced by PSA in a polysialyltransferase-specific method. PLoS One 10, e0124237. doi:10.1371/journal.pone.0124237
PubMed Summary | CrossRef Full Textual content | Google Scholar
Guimarães, L. P. T. P., Rocha, G. D. G., De Queiroz, R. M., De Araujo Martins, C., Takiya, C. M., and Gattass, C. R. (2017). Pomolic acid induces apoptosis and inhibits multidrug resistance protein MRP1 and migration in glioblastoma cells. Oncol. Rep. 38, 2525–2534. doi:10.3892/or.2017.5895
PubMed Summary | CrossRef Full Textual content | Google Scholar
Han, Y. H., Kim, S. H., Kim, S. Z., and Park, W. H. (2008). Apoptosis in arsenic trioxide-treated Calu-6 lung cells is correlated with the depletion of GSH ranges relatively than the modifications of ROS ranges. J. Cell. Biochem. 104, 862–878. doi:10.1002/jcb.21673
PubMed Summary | CrossRef Full Textual content | Google Scholar
Harford-Wright, E., Bidère, N., and Gavard, J. (2016). β-escin selectively targets the glioblastoma-initiating cell inhabitants and reduces cell viability. Oncotarget 7, 66865–66879. doi:10.18632/oncotarget.11784
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hersh, A. M., Gaitsch, H., Alomari, S., Lubelski, D., and Tyler, B. M. (2022). Molecular pathways and genomic panorama of glioblastoma stem cells: alternatives for focused remedy. Cancers (Basel) 14, 3743. doi:10.3390/cancers14153743
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hsu, C.-Y., Rajabi, S., Hamzeloo-Moghadam, M., Kumar, A., Maresca, M., and Ghildiyal, P. (2024). Sesquiterpene lactones as rising biomolecules to stop most cancers by concentrating on apoptosis. Entrance. Pharmacol. 15, 1371002. doi:10.3389/fphar.2024.1371002
PubMed Summary | CrossRef Full Textual content | Google Scholar
Hu, Y., Wu, F., Liu, Y., Zhao, Q., and Tang, H. (2019). DNMT1 recruited by EZH2-mediated silencing of miR-484 contributes to the malignancy of cervical most cancers cells via MMP14 and HNF1A. Clin. Epigenetics 11, 186. doi:10.1186/s13148-019-0786-y
PubMed Summary | CrossRef Full Textual content | Google Scholar
Iser, I. C., Ceschini, S. M., Onzi, G. R., Bertoni, A. P. S., Lenz, G., and Wink, M. R. (2016). Conditioned medium from adipose-derived stem cells (ADSCs) promotes epithelial-to-mesenchymal-like transition (EMT-Like) in glioma cells in vitro. Mol. Neurobiol. 53, 7184–7199. doi:10.1007/s12035-015-9585-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
Jin, S., Li, X., Dai, Y., Li, C., and Wang, D. (2020). NF-κB-mediated miR-650 performs oncogenic roles and prompts AKT/ERK/NF-κB pathways by concentrating on RERG in glioma cells. Cell. Oncol. 43, 1035–1048. doi:10.1007/s13402-020-00533-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
Joseph, J. V., Conroy, S., Tomar, T., Eggens-Meijer, E., Bhat, Okay., Copray, S., et al. (2014). TGF-β is an inducer of ZEB1-dependent mesenchymal transdifferentiation in glioblastoma that’s related to tumor invasion. Cell Dying Dis. 5, e1443. doi:10.1038/cddis.2014.395
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kang, Y. H., Han, S. R., Jeon, H., Lee, S., Lee, J., Yoo, S.-M., et al. (2019). Nogo receptor–vimentin interplay: a novel mechanism for the invasive exercise of glioblastoma multiforme. Exp. Mol. Med. 51, 1–15. doi:10.1038/s12276-019-0332-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kavitha, C. V., Jain, A. Okay., Agarwal, C., Pierce, A., Keating, A., Huber, Okay. M., et al. (2015). Asiatic acid induces endoplasmic reticulum stress and apoptotic dying in glioblastoma multiforme cells each in vitro and in vivo. Mol. Carcinog. 54, 1417–1429. doi:10.1002/mc.22220
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kim, H. J., Suh, S. S., Park, J., Shin, M. J., Koo, M. H., Lee, S. J., et al. (2022). 7β-22 dihydroxyhopane, remoted from the sub-antarctic lichen, inhibits the viability and stemness in glioma stem like cells. Onco. Targets. Ther. 15, 1375–1383. doi:10.2147/OTT.S371042
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kim, Y., Varn, F. S., Park, S.-H. H., Yoon, B. W., Park, H. R., Lee, C., et al. (2021). Perspective of mesenchymal transformation in glioblastoma. Acta Neuropathol. Commun. 9, 50. doi:10.1186/s40478-021-01151-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
Kopsida, M., Liu, N., Kotti, A., Wang, J., Jensen, L., Jothimani, G., et al. (2024). RhoB expression related to chemotherapy response and prognosis in colorectal most cancers. Most cancers Cell Int. 24, 75. doi:10.1186/s12935-024-03236-1
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lai, Y., Lu, X., Liao, Y., Ouyang, P., Wang, H., Zhang, X., et al. (2024). Crosstalk between glioblastoma and tumor microenvironment drives proneural–mesenchymal transition via ligand-receptor interactions. Genes Dis. 11, 874–889. doi:10.1016/j.gendis.2023.05.025
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lan, X., Jörg, D. J., Cavalli, F. M. G., Richards, L. M., Nguyen, L. V., Vanner, R. J., et al. (2017). Destiny mapping of human glioblastoma reveals an invariant stem cell hierarchy. Nature 549, 227–232. doi:10.1038/nature23666
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lee, C. H., Yu, C. C., Wang, B. Y., and Chang, W. W. (2016). Tumorsphere as an efficient in vitro platform for screening anticancer stem cell medication. Oncotarget 7, 1215–1226. doi:10.18632/oncotarget.6261
PubMed Summary | CrossRef Full Textual content | Google Scholar
Li, W., Liu, J., Fu, W., Zheng, X., Ren, L., Liu, S., et al. (2018). 3-O-acetyl-11-keto-β-boswellic acid exerts anti-tumor results in glioblastoma by arresting cell cycle at G2/M section. J. Exp. Clin. Most cancers Res. 37, 132. doi:10.1186/s13046-018-0805-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
Liu, J., Cao, J., and Zhao, X. (2015). miR-221 facilitates the TGFbeta1-induced epithelial-mesenchymal transition in human bladder most cancers cells by concentrating on STMN1. BMC Urol. 15, 36. doi:10.1186/s12894-015-0028-3
PubMed Summary | CrossRef Full Textual content | Google Scholar
Liu, Y., Zhou, Z., and Solar, S. (2024). Prospects of marine-derived compounds as potential therapeutic brokers for glioma. Pharm. Biol. 62, 513–526. doi:10.1080/13880209.2024.2359659
PubMed Summary | CrossRef Full Textual content | Google Scholar
Logashenko, E. B., Salomatina, O. V., Markov, A. V., Korchagina, D. V., Salakhutdinov, N. F., Tolstikov, G. A., et al. (2011). Synthesis and pro-apoptotic exercise of novel glycyrrhetinic acid derivatives. ChemBioChem 12, 784–794. doi:10.1002/cbic.201000618
PubMed Summary | CrossRef Full Textual content | Google Scholar
Lu, C.-C., Huang, B.-R., Liao, P.-J., and Yen, G.-C. (2014). Ursolic acid triggers nonprogrammed dying (necrosis) in human glioblastoma multiforme DBTRG-05MG cells via MPT pore opening and ATP decline. Mol. Nutr. Meals Res. 58, 2146–2156. doi:10.1002/mnfr.201400051
PubMed Summary | CrossRef Full Textual content | Google Scholar
Majchrzak-Celińska, A., and Studzińska-Sroka, E. (2024). New avenues and main achievements in phytocompounds analysis for glioblastoma remedy. Molecules 29, 1682. doi:10.3390/molecules29071682
PubMed Summary | CrossRef Full Textual content | Google Scholar
Markov, A. V., Ilyina, A. A., Salomatina, O. V., Sen’kova, A. V., Okhina, A. A., Rogachev, A. D., et al. (2022b). Novel soloxolone amides as potent anti-glioblastoma candidates: design, synthesis, in silico evaluation and organic actions in vitro and in vivo. Prescription drugs 15, 603. doi:10.3390/ph15050603
PubMed Summary | CrossRef Full Textual content | Google Scholar
Markov, A. V., Kel, A. E., Salomatina, O. V., Salakhutdinov, N. F., Zenkova, M. A., and Logashenko, E. B. (2019). Deep insights into the response of human cervical carcinoma cells to a brand new cyano enone-bearing triterpenoid soloxolone methyl: a transcriptome evaluation. Oncotarget 10, 5267–5297. doi:10.18632/oncotarget.27085
PubMed Summary | CrossRef Full Textual content | Google Scholar
Markov, A. V., Odarenko, Okay. V., Ilyina, A. A., and Zenkova, M. A. (2022a). Uncovering the anti-angiogenic impact of semisynthetic triterpenoid CDDO-Im on HUVECs by an built-in community pharmacology method. Comput. Biol. Med. 141, 105034. doi:10.1016/J.COMPBIOMED.2021.105034
PubMed Summary | CrossRef Full Textual content | Google Scholar
Markov, A. V., Odarenko, Okay. V., Sen’kova, A. V., Ilyina, A. A., and Zenkova, M. A. (2023). Analysis of the antitumor potential of soloxolone tryptamide towards glioblastoma multiforme utilizing in silico, in vitro, and in vivo approaches. Biochem. 88, 1008–1021. doi:10.1134/S000629792307012X
Markov, A. V., Odarenko, Okay. V., Sen’kova, A. V., Salomatina, O. V., Salakhutdinov, N. F., Zenkova, M. A., et al. (2020). Cyano enone-bearing triterpenoid soloxolone methyl inhibits epithelial-mesenchymal transition of human lung adenocarcinoma cells in vitro and metastasis of murine melanoma in vivo. Molecules 25, 5925. doi:10.3390/MOLECULES25245925
PubMed Summary | CrossRef Full Textual content | Google Scholar
Markwell, S. M., Ross, J. L., Olson, C. L., and Brat, D. J. (2022). Necrotic reshaping of the glioma microenvironment drives illness development. Acta Neuropathol. 143, 291–310. doi:10.1007/s00401-021-02401-4
PubMed Summary | CrossRef Full Textual content | Google Scholar
Mazuryk, O., Suzenet, F., Kieda, C., and Brindell, M. (2015). The organic impact of the nitroimidazole spinoff of a polypyridyl ruthenium complicated on most cancers and endothelial cells†. Metallomics 7, 553–566. doi:10.1039/c5mt00037h
PubMed Summary | CrossRef Full Textual content | Google Scholar
McConnell, D., McGreevy, J., Williams, M., and Litofsky, N. (2018). Do anti-oxidants vitamin D3, melatonin, and alpha-lipoic acid have synergistic results with temozolomide on cultured glioblastoma cells? Medicines 5, 58. doi:10.3390/medicines5020058
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ouanouki, A., Lamy, S., and Annabi, B. (2018). Periostin, a sign transduction intermediate in TGF-β-induced EMT in U-87MG human glioblastoma cells, and its inhibition by anthocyanidins. Oncotarget 9, 22023–22037. doi:10.18632/oncotarget.25153
PubMed Summary | CrossRef Full Textual content | Google Scholar
Popova, S. N., Bergqvist, M., Dimberg, A., Edqvist, P.-H., Ekman, S., Hesselager, G., et al. (2014). Subtyping of gliomas of assorted WHO grades by the appliance of immunohistochemistry. Histopathology 64, 365–379. doi:10.1111/his.12252
PubMed Summary | CrossRef Full Textual content | Google Scholar
Redlich, J.-P., Feuerhake, F., Weis, J., Schaadt, N. S., Teuber-Hanselmann, S., Buck, C., et al. (2024). Functions of synthetic intelligence within the evaluation of histopathology photos of gliomas: a assessment. npj Imaging 2, 16. doi:10.1038/s44303-024-00020-8
Rodríguez-Camacho, A., Flores-Vázquez, J. G., Moscardini-Martelli, J., Torres-Ríos, J. A., Olmos-Guzmán, A., Ortiz-Arce, C. S., et al. (2022). Glioblastoma therapy: state-of-the-art and future views. Int. J. Mol. Sci. 23, 7207. doi:10.3390/ijms23137207
PubMed Summary | CrossRef Full Textual content | Google Scholar
Rui, W., Zhang, S., Shi, H., Sheng, Y., Zhu, F., Yao, Y., et al. (2023). Deep learning-assisted quantitative susceptibility mapping as a device for grading and molecular subtyping of gliomas. Phenomics 3, 243–254. doi:10.1007/s43657-022-00087-6
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sahoo, O. S., Mitra, R., and Nagaiah, N. Okay. H. (2024). The hidden architects of glioblastoma multiforme: glioma stem cells. MedComm – Oncol. 3, e66. doi:10.1002/mog2.66
Samandari-Bahraseman, M. R., Khorsand, B., Zareei, S., Amanlou, M., and Rostamabadi, H. (2023). Varied concentrations of hesperetin induce various kinds of programmed cell dying in human breast cancerous and regular cell strains in a ROS-dependent method. Chem. Biol. Work together. 382, 110642. doi:10.1016/j.cbi.2023.110642
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sava, G., Frausin, F., Cocchietto, M., Vita, F., Podda, E., Spessotto, P., et al. (2004). Actin-dependent tumour cell adhesion after short-term publicity to the antimetastasis ruthenium complicated NAMI-A. Eur. J. Most cancers 40, 1383–1396. doi:10.1016/j.ejca.2004.01.034
PubMed Summary | CrossRef Full Textual content | Google Scholar
Seker-Polat, F., Pinarbasi Degirmenci, N., Solaroglu, I., and Bagci-Onder, T. (2022). Tumor cell infiltration into the mind in glioblastoma: from mechanisms to scientific views. Cancers (Basel) 14, 443. doi:10.3390/cancers14020443
PubMed Summary | CrossRef Full Textual content | Google Scholar
Sprowls, S. A., Arsiwala, T. A., Bumgarner, J. R., Shah, N., Lateef, S. S., Kielkowski, B. N., et al. (2019). Bettering CNS supply to mind metastases by blood–tumor barrier disruption. Traits Most cancers 5, 495–505. doi:10.1016/j.trecan.2019.06.003
PubMed Summary | CrossRef Full Textual content | Google Scholar
Stack, C., Ho, D., Wille, E., Calingasan, N. Y., Williams, C., Liby, Okay., et al. (2010). Triterpenoids CDDO-ethyl amide and CDDO-trifluoroethyl amide enhance the behavioral phenotype and mind pathology in a transgenic mouse mannequin of Huntington’s illness. Free Radic. Biol. Med. 49, 147–158. doi:10.1016/j.freeradbiomed.2010.03.017
PubMed Summary | CrossRef Full Textual content | Google Scholar
Solar, L. W., Kao, S. H., Yang, S. F., Jhang, S. W., Lin, Y. C., Chen, C. M., et al. (2021). Corosolic acid attenuates the invasiveness of glioblastoma cells by selling CHIP-mediated AXL degradation and inhibiting GAS6/AXL/JAK axis. Cells 10, 2919. doi:10.3390/cells10112919
PubMed Summary | CrossRef Full Textual content | Google Scholar
Szu, J. I., Tsigelny, I. F., Wojcinski, A., and Kesari, S. (2023). Organic capabilities of the Olig gene household in mind most cancers and therapeutic concentrating on. Entrance. Neurosci. 17, 1129434. doi:10.3389/fnins.2023.1129434
PubMed Summary | CrossRef Full Textual content | Google Scholar
Tai, S. H., Lin, Y. W., Huang, T. Y., Chang, C. C., Chao, L. C., Wu, T. S., et al. (2021). Cinnamophilin enhances temozolomide-induced cytotoxicity towards malignant glioma: the roles of ROS and cell cycle arrest. Transl. Most cancers Res. 10, 3906–3920. doi:10.21037/tcr-20-3426
PubMed Summary | CrossRef Full Textual content | Google Scholar
Teraiya, M., Perreault, H., and Chen, V. C. (2023). An summary of glioblastoma multiforme and temozolomide resistance: can LC-MS-based proteomics reveal the basic mechanism of temozolomide resistance? Entrance. Oncol. 13, 1166207. doi:10.3389/fonc.2023.1166207
PubMed Summary | CrossRef Full Textual content | Google Scholar
Thakor, F. Okay., Wan, Okay.-W., Welsby, P. J., and Welsby, G. (2017). Pharmacological results of asiatic acid in glioblastoma cells below hypoxia. Mol. Cell. Biochem. 430, 179–190. doi:10.1007/s11010-017-2965-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
Tomar, M. S., Kumar, A., Srivastava, C., and Shrivastava, A. (2021). Elucidating the mechanisms of Temozolomide resistance in gliomas and the methods to beat the resistance. Biochim. Biophys. Acta – Rev. Most cancers 1876, 188616. doi:10.1016/j.bbcan.2021.188616
PubMed Summary | CrossRef Full Textual content | Google Scholar
Tsai, T.-H., Lieu, A.-S., Wang, Y.-W., Yang, S.-F., Hsu, Y.-C., and Lin, C.-L. (2021). Therapeutic potential of RTA 404 in human mind malignant glioma cell strains by way of cell cycle arrest by way of p21/AKT signaling. Biomed. Res. Int. 2021, 5552226. doi:10.1155/2021/5552226
PubMed Summary | CrossRef Full Textual content | Google Scholar
Tsai, T. H., Su, Y. F., Tsai, C. Y., Wu, C. H., Lee, Okay. T., and Hsu, Y. C. (2023). RTA dh404 induces cell cycle arrest, apoptosis, and autophagy in glioblastoma cells. Int. J. Mol. Sci. 24, 4006. doi:10.3390/ijms24044006
PubMed Summary | CrossRef Full Textual content | Google Scholar
Uceda-Castro, R., van Asperen, J. V., Vennin, C., Sluijs, J. A., van Bodegraven, E. J., Margarido, A. S., et al. (2022). GFAP splice variants fine-tune glioma cell invasion and tumour dynamics by modulating migration persistence. Sci. Rep. 12, 424. doi:10.1038/s41598-021-04127-5
PubMed Summary | CrossRef Full Textual content | Google Scholar
van Dijk, I. A., Laura Ferrando, M., van der Wijk, A.-E., Hoebe, R. A., Nazmi, Okay., de Jonge, W. J., et al. (2017). Human salivary peptide histatin-1 stimulates epithelial and endothelial cell adhesion and barrier operate. FASEB J. 31, 3922–3933. doi:10.1096/fj.201700180R
PubMed Summary | CrossRef Full Textual content | Google Scholar
Verdura, S., Encinar, J. A., Teixidor, E., Segura-Carretero, A., Micol, V., Cuyàs, E., et al. (2022). Silibinin overcomes EMT-driven lung most cancers resistance to new-generation ALK inhibitors. Cancers (Basel) 14, 6101. doi:10.3390/cancers14246101
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wang, J., Li, Y., Wang, X., and Jiang, C. (2012). Ursolic acid inhibits proliferation and induces apoptosis in human glioblastoma cell strains U251 by suppressing TGF-β1/miR-21/PDCD4 pathway. Fundamental Clin. Pharmacol. Toxicol. 111, 106–112. doi:10.1111/j.1742-7843.2012.00870.x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wang, Y.-F., and Hu, J.-Y. (2023). Pure and artificial compounds for glioma therapy based mostly on ROS-mediated technique. Eur. J. Pharmacol. 953, 175537. doi:10.1016/j.ejphar.2023.175537
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wang, Z., Zhang, H., Xu, S., Liu, Z., and Cheng, Q. (2021). The adaptive transition of glioblastoma stem cells and its implications on therapies. Sign Transduct. Goal. Ther. 6, 124. doi:10.1038/s41392-021-00491-w
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wu, B., Zhu, J., Dai, X., Ye, L., Wang, B., Cheng, H., et al. (2021a). Raddeanin A inhibited epithelial-mesenchymal transition (EMT) and angiogenesis in glioblastoma by downregulating β-catenin expression. Int. J. Med. Sci. 18, 1609–1617. doi:10.7150/ijms.52206
PubMed Summary | CrossRef Full Textual content | Google Scholar
Wu, W., Klockow, J. L., Zhang, M., Lafortune, F., Chang, E., Jin, L., et al. (2021b). Glioblastoma multiforme (GBM): an summary of present therapies and mechanisms of resistance. Pharmacol. Res. 171, 105780. doi:10.1016/j.phrs.2021.105780
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xia, J., Li, S., Liu, S., and Zhang, L. (2023). Aldehyde dehydrogenase in stable tumors and different ailments: potential biomarkers and therapeutic targets. MedComm 4, e195. doi:10.1002/mco2.195
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xie, X. P., Laks, D. R., Solar, D., Ganbold, M., Wang, Z., Pedraza, A. M., et al. (2022). Quiescent human glioblastoma most cancers stem cells drive tumor initiation, enlargement, and recurrence following chemotherapy. Dev. Cell 57, 32–46.e8. doi:10.1016/j.devcel.2021.12.007
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xu, H., Miao, H., Chen, G., Zhang, G., Hua, Y., Wu, Y., et al. (2023). 20(S)-ginsenoside Rg3 exerts anti-fibrotic impact after myocardial infarction by alleviation of fibroblasts proliferation and collagen deposition via TGFBR1 signaling pathways. J. Ginseng Res. 47, 743–754. doi:10.1016/j.jgr.2023.06.007
PubMed Summary | CrossRef Full Textual content | Google Scholar
Xu, M., Cui, Q., Su, W., Zhang, D., Pan, J., Liu, X., et al. (2022). Excessive-content screening of energetic parts of Conventional Chinese language Drugs inhibiting TGF-β-induced cell EMT. Heliyon 8, e10238. doi:10.1016/j.heliyon.2022.e10238
PubMed Summary | CrossRef Full Textual content | Google Scholar
Ye, G., Jiao, Y., Deng, L., Cheng, M., Wang, S., Zhang, J., et al. (2023). Beauvericin suppresses the proliferation and pulmonary metastasis of osteosarcoma by selectively inhibiting TGFBR2 pathway. Int. J. Biol. Sci. 19, 4376–4392. doi:10.7150/ijbs.86214
PubMed Summary | CrossRef Full Textual content | Google Scholar
Yin, H., Zhou, Y., Wen, C., Zhou, C., Zhang, W., Hu, X., et al. (2014). Curcumin sensitizes glioblastoma to temozolomide by concurrently producing ROS and disrupting AKT/mTOR signaling. Oncol. Rep. 32, 1610–1616. doi:10.3892/or.2014.3342
PubMed Summary | CrossRef Full Textual content | Google Scholar
Yoshimura, H., Sugawara, Okay., Saito, M., Saito, S., Murakami, S., Miyata, N., et al. (2003). In vitro TGF-beta1 antagonistic exercise of ursolic and oleanolic acids remoted from Clerodendranthus spicatus. Planta Med. 69, 673–675. doi:10.1055/s-2003-41110
PubMed Summary | CrossRef Full Textual content | Google Scholar
Yuan, Y., Xue, X., Guo, R.-B., Solar, X.-L., and Hu, G. (2012). Resveratrol enhances the antitumor results of temozolomide in glioblastoma by way of ROS-dependent AMPK-TSC-mTOR signaling pathway. CNS Neurosci. Ther. 18, 536–546. doi:10.1111/j.1755-5949.2012.00319.x
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, J., Zhang, Z., Huang, Z., Li, M., Yang, F., Wu, Z., et al. (2023). Isotoosendanin exerts inhibition on triple-negative breast most cancers via abrogating TGF-β-induced epithelial–mesenchymal transition by way of straight concentrating on TGFβR1. Acta Pharm. Sin. B 13, 2990–3007. doi:10.1016/j.apsb.2023.05.006
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, X., Liu, Y., Xu, S., Yang, R., Jiang, C., Zhu, L., et al. (2022). Asiatic acid from Cyclocarya paliurus regulates the autophagy–lysosome system by way of straight inhibiting TGF-β kind I receptor and ameliorates diabetic nephropathy fibrosis. Meals Funct. 13, 5536–5546. doi:10.1039/D1FO02445K
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhang, X., Zheng, Okay., Li, C., Zhao, Y., Li, H., Liu, X., et al. (2017). Nobiletin inhibits invasion by way of inhibiting AKT/GSK3β/β-catenin signaling pathway in Slug-expressing glioma cells. Oncol. Rep. 37, 2847–2856. doi:10.3892/or.2017.5522
PubMed Summary | CrossRef Full Textual content | Google Scholar
Zhitnyak, I. Y., Rubtsova, S. N., Litovka, N. I., and Gloushankova, N. A. (2020). Early occasions in actin cytoskeleton dynamics and E-cadherin-mediated cell-cell adhesion throughout epithelial-mesenchymal transition. Cells 9, 578. doi:10.3390/cells9030578
PubMed Summary | CrossRef Full Textual content | Google Scholar