Sufferers
General, 314 sufferers with newly identified AML had been included within the evaluation. The median age was 74 years (vary 25–90) and 111 (35%) had antecedent myeloid neoplasm. Most sufferers (75%) had been categorized as hostile danger by ELN 2022. In complete,166 (53%) had been handled with HMA + VEN, and 148 (47%) had been handled with HMA (127 [40%] with HMA monotherapy and 21 [7%] with HMA + non-VEN drug), Desk 1. There have been no vital variations in baseline traits between sufferers handled with HMA vs. HMA + VEN. When categorized by ontogeny, 111 (35%) had been within the TP53 group (60 with HMA + VEN, 51 with HMA); 115 (37%) within the secondary ontogeny group (68 with HMA + VEN, 47 with HMA); and 88 (28%) within the de novo ontogeny group (38 with HMA + VEN, 50 with HMA, Fig. 1 and Supplementary Fig. 1B). Decitabine was extra generally used in contrast with azacytidine (n = 208 [66%] vs. n = 106 [34%)], with 5 days of decitabine being the commonest HMA routine used (n = 166 [53%], Supplementary Desk 1).
Survival outcomes
Survival in the complete group
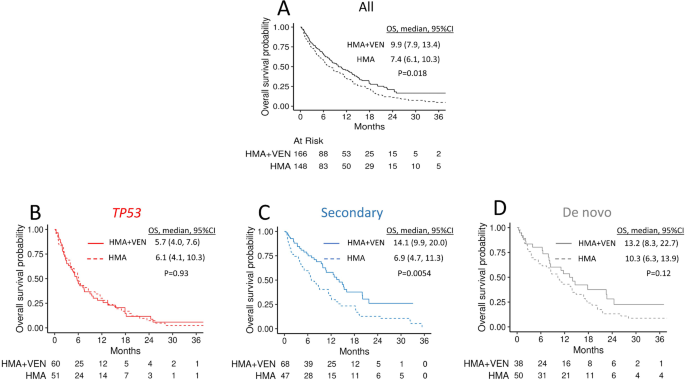
After a median follow-up of 29 months (95% CI 24–32), the OS was higher in sufferers handled with HMA + VEN in contrast with those that obtained HMA with out VEN (median OS 9.9 months [95% CI 7.9–13.4] vs. 7.4 months [95% CI 6.1–10.3], respectively, P = 0.018, Fig. 2A). The outcomes had been related in a sensitivity evaluation excluding sufferers who obtained HMA+ a further non-VEN drug (n = 21, Supplementary Fig. 2). In a multivariable evaluation, solely TP53 ontogeny (HR 1.88 [95% CI 1.37–2.59]) and consolidation with alloHCT (HR 0.23 [95% CI 0.14–0.40]) had been independently related to OS (Supplementary Desk 2).
Survival in every ontogeny group
To outline the ontogeny-specific exercise of VEN, we carried out a separate evaluation in every molecularly outlined ontogeny group. Amongst sufferers with TP53 mutated illness, there was no distinction in OS between HMA + VEN vs. HMA remedy teams (5.7 months [95% CI 4–7.6] vs. 6.1 months [95% CI 4.1–10.3], P = 0.93), Fig. 2B. Moreover, OS was comparable in each remedy teams amongst sufferers with TP53 mutations regardless of VAF cutoff, together with 10%, 20%, and 50% (Supplementary Desk 3). Within the de novo group, OS was not totally different between HMA + VEN vs. HMA (13.2 months [95% CI 8.3–22.7] vs. 10.3 months [95% CI 6.3–13.9], P = 0.12, Fig. 2C). Conversely, within the secondary group, sufferers handled with HMA + VEN had considerably higher OS in comparison with sufferers handled with HMA (14.1 months [9.9–20.0] vs. 6.9 months [95% CI 4.7–11.3], P = 0.0054, Fig. 2D). The outcomes had been retained in a sensitivity evaluation when RUNX1 was thought of as a secondary ontogeny-defining mutation (Supplementary Fig. 3).
To determine ontogeny-specific prognostic variables, we carried out Cox multivariable regression fashions inside every ontogeny group individually. Throughout all teams, alloHCT was related to improved OS (de novo: HR 0.14 [95% CI 0.03–0.58], P = 0.0067; secondary: HR 0.40 [95% CI 0.18–0.89], P = 0.026; TP53: HR 0.17 [95% CI 0.07–0.42], p < 0.001). Conversely, remedy modality (HMA + VEN vs. HMA) was related to improved OS solely within the secondary group (de novo: HR 0.67 [95% CI 0.40–1.12], P = 0.13; secondary: HR 0.59 [95% CI 0.38–0.94], P = 0.025; TP53: HR 1.19 [95% CI 0.78–1.81], P = 0.42).
Sensitivity evaluation with out TP53 group
Since TP53 mutations had been strongly related to worse outcomes and had been extremely prevalent within the cohort, their inclusion within the evaluation may obscure a significant impact of preliminary remedy on survival in sufferers with out TP53 mutations. We carried out a sensitivity evaluation and regression modeling for OS in sufferers with out TP53 mutations (n = 203). Amongst these sufferers, the median OS was 14.0 months (95% CI 10.6–17.8) with HMA + VEN vs. 8.7 months (95% CI 6.3–11.4) with HMA, P = 0.0028 (Supplementary Fig. 4). Within the multivariable evaluation, each alloHCT (HR 0.27 [95% CI 0.13–0.53, p < 0.001]) and remedy with HMA + VEN (vs. HMA) (HR 0.66 [95% CI 0.47–0.92], P = 0.015) had been related to improved OS (Supplementary Desk 4).
Survival in sufferers handled with HMA + VEN
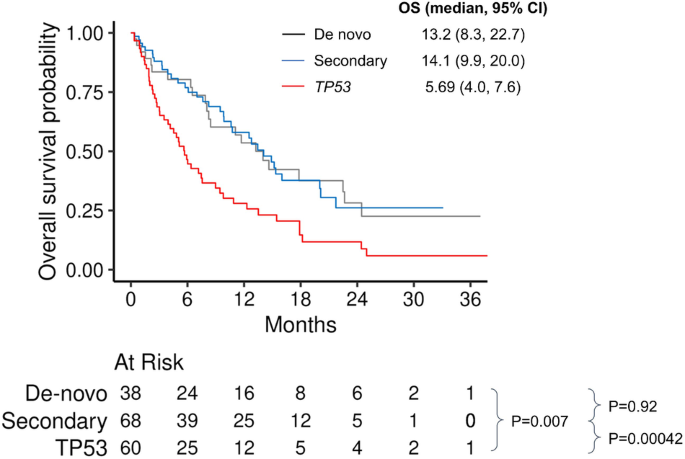
In sufferers handled with HMA + VEN the OS was related between de novo and secondary teams (median OS 13.2 months [95% CI 8.3–22.7] vs. 14.1 months [95% CI 9.9–20.0], respectively, P = 0.92); and every group had higher OS in comparison with TP53 group (median 5.7 months [95% CI 4.0–7.6], comparability vs. de novo P = 0.007; comparability vs. secondary p < 0.001), Fig. 3. Within the multivariable evaluation of sufferers handled with HMA + VEN, the comparable OS between secondary and de novo teams was retained (HR 1.07 [95% CI 0.63–1.84, P = 0.79]), in addition to the more serious survival within the TP53 group (in contrast with de novo, HR 2.57 [95% CI 1.53–4.33, P < 0.001]) and higher survival with alloHCT as a time-varying covariate (HR0.22 [95% CI 0.11–0.46], P < 0.001, Desk 2).
Response and transplant charges
To find out whether or not the distinct survival patterns in every ontogeny had been related to different measures of medical response, we evaluated cCR and alloHCT charges in the complete cohort and inside every ontogeny group individually. General, the cCR fee was higher in sufferers handled with HMA + VEN than in those that obtained HMA (49% vs. 28%, P = 0.001). When analyzing molecularly outlined ontogeny teams individually, the cCR charges had been greater in sufferers handled with HMA + VEN vs. HMA within the de novo group (54% vs. 29%, P = 0.034]) and secondary group (61% vs.18%, p < 0.001), however not totally different in sufferers with TP53 mutations (33% vs. 37%, P = 0.82, Desk 3).
As long-term survival amongst sufferers with high-risk AML subtypes depends upon alloHCT [12], we evaluated whether or not HMA + VEN could also be related to greater alloHCT charges in the complete cohort and in every ontogeny group. General, 41 (13%) sufferers had been consolidated with alloHCT, with sufferers handled with HMA + VEN extra regularly transplanted (17%, n = 29) than these handled with HMA (8%, n = 12), P = 0.018. Nevertheless, when analyzed inside every ontogeny group, the charges of alloHCT had been related inside the TP53 group (15% [n = 9] vs. 8% [n = 4], P = 0.38) and within the de novo group (11% [n = 4] vs.10% [n = 5], p > 0.99). Conversely, the charges had been greater amongst sufferers within the secondary group handled with HMA + VEN (24%, n = 16) vs. HMA (6%, n = 3), P = 0.02.
Subgroup evaluation of secondary ontogeny
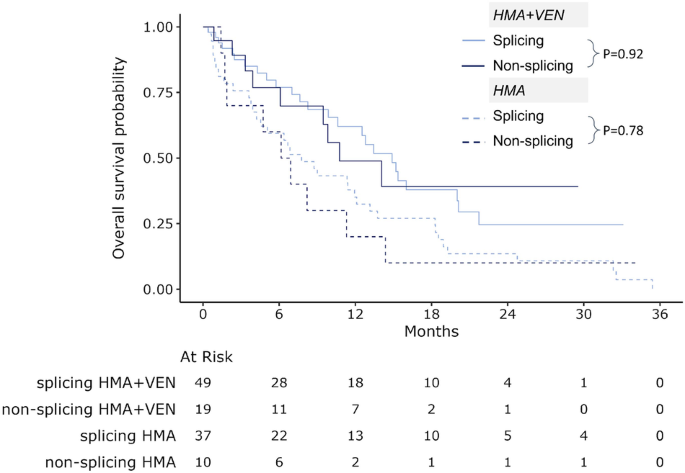
Mutations that have an effect on RNA splicing (SF3B1, SRSF2, U2AF1, ZRSR2) have been reported to be related to improved survival in sufferers handled with VEN-based regimens[13]. To find out whether or not the aforementioned response and survival profit with HMA + VEN vs. HMA within the secondary ontogeny group is attributable solely to splicing abnormalities in these mutations, we in contrast sufferers with (n = 86) vs. with out (n = 29) these mutations inside the secondary group. First, splicing vs. non-splicing subgroups had been comparable in age, prior remedy, or myeloid malignancy charges and ELN2022 danger standards (Supplementary Desk 5). The splicing vs. non-splicing subgroup had greater charges of NPM1 (16% vs. 0%, respectively P = 0.02) and decrease charges of ASXL1 (42% vs. 79%, respectively, P < 0.001). The cCR charges between splicing and non-splicing mutation subgroups had been comparable amongst sufferers handled with HMA (22 vs. 0%, P = 0.3) or HMA + VEN (61% VS. 60%, P > 0.99). Equally, OS was comparable between splicing and non-splicing subgroups handled with HMA (median OS 7.8 months [95% CI 4.2–12.1] vs. 6.5 months [95% CI 1.9-11.3], P = 0.78) or with HMA + VEN (median OS 14.9 months [95% CI 10.6–20.1] vs. 10.8 months [95% CI 6.1-NR], P = 0.92), Fig. 4.